Scalable Organocatalytic Synthesis of Crizotinib Intermediates for Global Pharmaceutical Manufacturing
Scalable Organocatalytic Synthesis of Crizotinib Intermediates for Global Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for the production of high-value oncology therapeutics. Patent CN102584795B introduces a transformative preparation method for Crizotinib, specifically targeting the synthesis of the critical chiral intermediate (R)-3-(1-(2,6-dichloro-3-fluorophenyl)ethoxy)-5-(1-(piperidin-4-yl)-1H-pyrazol-4-yl)pyridin-2-amine. This technology represents a significant paradigm shift from traditional biocatalytic or chemical resolution methods to a streamlined organocatalytic asymmetric synthesis. By leveraging small molecule catalysts such as (S)-diphenylprolinol in conjunction with metal borohydrides, the process achieves exceptional stereocontrol and yield. For R&D directors and supply chain leaders, this innovation addresses the chronic bottlenecks of enzyme stability and resolution inefficiency, offering a reliable pathway for the commercial scale-up of complex pharmaceutical intermediates.
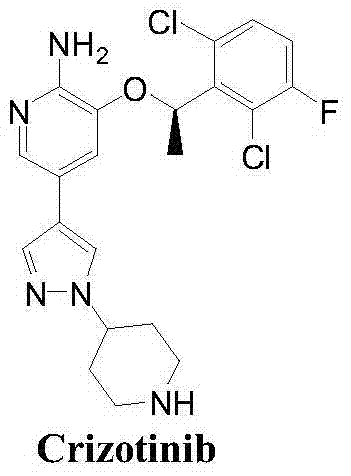
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key chiral alcohol intermediate for Crizotinib has relied heavily on two primary strategies, both of which present substantial drawbacks for modern manufacturing. The first approach, disclosed in earlier patents like WO2006021881A2, utilizes an esterase-catalyzed hydrolysis of racemic acetates. While effective on a small scale, this enzymatic route is plagued by long reaction cycles, severe reaction conditions, and the high cost of biocatalysts. Furthermore, enzymatic processes often suffer from poor reproducibility and difficulty in large-scale industrial application due to the sensitivity of enzymes to process parameters. The second conventional method involves chemical resolution of racemic phenylethyl alcohol using chiral resolving agents. As illustrated in the prior art reaction schemes, this approach is inherently inefficient because the maximum theoretical yield is capped at 50%, necessitating the disposal or recycling of the unwanted enantiomer, which drastically inflates production costs and waste generation.
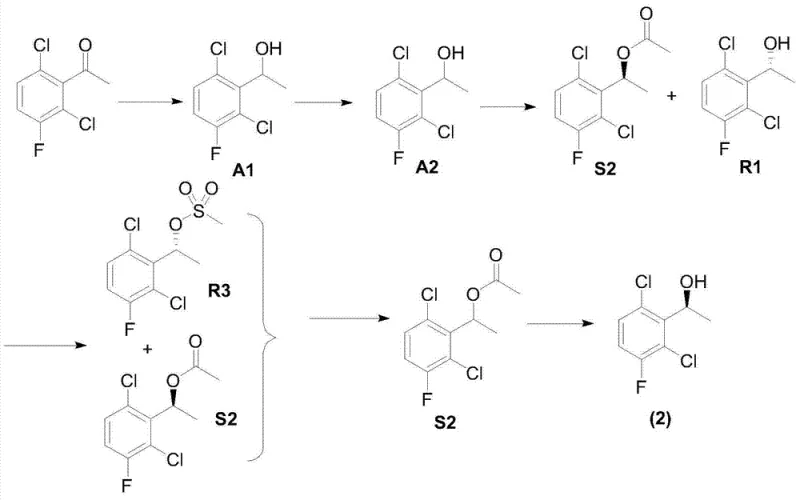
The Novel Approach
In stark contrast to these legacy methods, the novel organocatalytic strategy described in CN102584795B bypasses the need for expensive enzymes and the yield limitations of resolution. The core innovation lies in the direct asymmetric reduction of substituted acetophenone (Formula 1) to the chiral phenylethyl alcohol (Formula 2) in a single step. This is achieved using an organic micromolecule catalyst, specifically (S)-diphenylprolinol, activated by a silane and a borohydride reducing agent. This method not only simplifies the synthetic route by eliminating the acetylation and hydrolysis steps required in the enzymatic path but also ensures high optical purity from the outset. The reaction conditions are mild, typically ranging from 20°C to 80°C, and utilize inexpensive, commercially available reagents. This transition from biocatalysis to organocatalysis fundamentally alters the economic model of production, enabling cost reduction in pharmaceutical intermediates manufacturing through simplified operations and superior atom economy.
Mechanistic Insights into Organocatalytic Asymmetric Reduction
The heart of this technological advancement is the stereoselective reduction mechanism employed in the first step of the synthesis. The reaction utilizes a chiral organic catalyst, (S)-diphenylprolinol, which interacts with the ketone substrate and the reducing system comprising a metal borohydride (such as sodium borohydride) and a trialkylchlorosilane (such as trimethylchlorosilane). The silane likely activates the borohydride in situ to generate a more reactive hydride species, while the chiral catalyst creates a sterically defined environment around the carbonyl group. This environment directs the hydride attack to a specific face of the ketone, resulting in the preferential formation of the (S)-configured alcohol intermediate with high enantiomeric excess (ee values reported up to 96-99%). This level of control is critical for downstream processing, as high initial optical purity minimizes the burden on subsequent purification steps and ensures the final API meets stringent regulatory standards for chiral drugs.
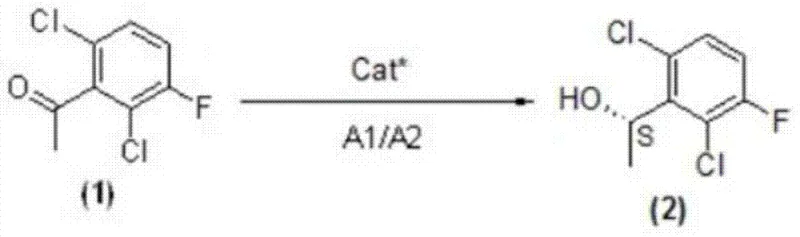
Beyond the primary reduction, the process incorporates robust mechanisms for impurity control throughout the six-step sequence. The subsequent Mitsunobu coupling (Step 2) utilizes triphenylphosphine and DIAD/DEAD to link the chiral alcohol with 2-nitro-3-pyridone under mild conditions (-20°C to 30°C), minimizing side reactions such as elimination or racemization. The reduction of the nitro group to the amine (Step 3) employs iron or zinc powder in protic solvents, a classic and highly selective method that avoids the over-reduction of other sensitive functional groups on the pyridine ring. Finally, the Suzuki-Miyaura coupling (Step 5) connects the pyridine fragment with the pyrazole-piperidine moiety using palladium catalysis. Each step is optimized to prevent the formation of difficult-to-remove impurities, ensuring a clean impurity profile that facilitates easier isolation and crystallization of the final product, thereby enhancing the overall process robustness for GMP manufacturing.
How to Synthesize Crizotinib Efficiently
The synthesis of Crizotinib via this organocatalytic route is designed for operational simplicity and high throughput. The process begins with the preparation of the chiral alcohol, followed by etherification, nitro reduction, bromination, cross-coupling, and final deprotection. The detailed standardized synthesis steps, including specific reagent ratios, solvent choices, and temperature profiles for each stage, are outlined below to guide process development teams in replicating this high-efficiency pathway.
- Perform asymmetric reduction of substituted acetophenone using an organic micromolecule catalyst (S-diphenylprolinol) with metal borohydride and trialkylchlorosilane to obtain chiral phenylethyl alcohol.
- Execute a Mitsunobu reaction between the chiral alcohol and 2-nitro-3-pyridone using triphenylphosphine and DIAD/DEAD to form the nitro-compound intermediate.
- Reduce the nitro group to an aromatic amine using iron or zinc powder, followed by bromination, Suzuki coupling with a boronic ester, and final acid deprotection to yield the target API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic synthesis offers profound strategic advantages beyond mere technical feasibility. The shift away from biological enzymes eliminates the supply chain volatility associated with specialized biocatalysts, which can be subject to batch-to-batch variability and long lead times. Instead, the process relies on commodity chemicals like sodium borohydride and silanes, which are produced globally in massive quantities, ensuring a stable and continuous supply of raw materials. This stability is crucial for maintaining uninterrupted production schedules for life-saving oncology medications. Furthermore, the simplification of the process flow—removing multiple protection and deprotection steps inherent in the enzymatic route—drastically reduces the number of unit operations, leading to shorter manufacturing cycles and lower utility consumption.
- Cost Reduction in Manufacturing: The economic impact of replacing enzymatic resolution with organocatalysis is substantial. By eliminating the need for expensive enzyme preparations and the associated buffer systems, the direct material costs are significantly lowered. Additionally, the avoidance of the 50% yield ceiling inherent in resolution methods means that nearly all starting material is converted into the desired chiral intermediate, effectively doubling the theoretical output per kilogram of raw material compared to resolution routes. This dramatic improvement in atom economy translates directly into lower cost of goods sold (COGS), allowing for more competitive pricing in the generic and contract manufacturing markets without compromising margin.
- Enhanced Supply Chain Reliability: The reliance on widely available organic small molecules and standard inorganic reagents mitigates the risk of supply disruptions. Unlike specialized enzymes that may have single-source suppliers, reagents like triphenylphosphine, borohydrides, and palladium catalysts are sourced from a broad global network of chemical manufacturers. This diversification of the supply base enhances resilience against geopolitical or logistical shocks. Moreover, the robustness of the reaction conditions, which tolerate a wider range of temperatures and do not require strict sterile environments like fermentation, allows for manufacturing in a broader range of facilities, further securing the supply chain against localized production issues.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is markedly superior. The reduction in step count and the elimination of aqueous enzymatic waste streams simplify wastewater treatment protocols. The use of standard organic solvents like THF and ethyl acetate allows for efficient solvent recovery and recycling systems, aligning with green chemistry principles. The process is explicitly designed for scale operation, with exotherms and reaction kinetics that are manageable in large reactors. This ease of scale-up ensures that production can be rapidly ramped from pilot plant quantities to multi-ton commercial campaigns to meet market demand, all while maintaining a reduced environmental footprint compared to the resource-intensive enzymatic alternatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this organocatalytic technology. These insights are derived directly from the patent data to assist technical teams in evaluating the feasibility of adopting this route for their specific manufacturing contexts. Understanding these nuances is essential for making informed decisions about process transfer and vendor qualification.
Q: How does this organocatalytic method improve upon traditional enzymatic resolution?
A: Unlike traditional enzymatic methods which suffer from long reaction cycles, severe conditions, and expensive catalysts, this organocatalytic approach utilizes small molecule catalysts like (S)-diphenylprolinol. This results in a shorter reaction cycle, higher yields, simpler operation, and significantly lower raw material costs, making it ideal for large-scale industrial production.
Q: What represents the key advantage regarding optical purity in this synthesis?
A: The process achieves high optical purity (ee value up to 99%) through a highly stereoselective asymmetric reduction step. By avoiding the 50% theoretical yield limit of chemical resolution methods, this route ensures that the final Crizotinib intermediate maintains rigorous chiral specifications required for oncology therapeutics without the waste associated with discarding the unwanted enantiomer.
Q: Is this synthetic route suitable for commercial scale-up?
A: Yes, the patent explicitly highlights the method's suitability for scale operation. The reaction conditions are mild (often room temperature to 87°C), the reagents are widely available and inexpensive, and the process avoids the reproducibility issues common with biological enzymes, ensuring consistent quality and supply continuity for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Crizotinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the delivery of high-quality oncology therapeutics. Our team of expert chemists has extensively analyzed the organocatalytic pathways described in CN102584795B and possesses the technical capability to implement this advanced methodology at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to commercial reactor is seamless. Our facilities are equipped with rigorous QC labs and state-of-the-art analytical instrumentation to verify stringent purity specifications, including chiral HPLC for monitoring enantiomeric excess at every critical stage of the synthesis.
We invite global pharmaceutical partners to collaborate with us on optimizing this cost-effective Crizotinib synthesis. By leveraging our expertise in organocatalysis and process intensification, we can help you achieve significant efficiency gains in your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term supply goals for this vital cancer treatment.
