Advanced Green Manufacturing of 2-Chloro-3-cyanopyridine for Global Pharmaceutical Supply Chains
Advanced Green Manufacturing of 2-Chloro-3-cyanopyridine for Global Pharmaceutical Supply Chains
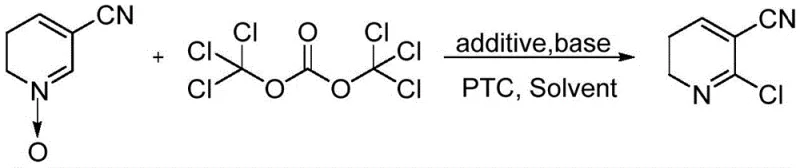
The pharmaceutical and agrochemical industries are increasingly demanding sustainable manufacturing pathways that do not compromise on yield or purity, a challenge effectively addressed by the technology disclosed in patent CN109369519B. This pivotal intellectual property outlines a robust green preparation method for 2-chloro-3-cyanopyridine, a critical intermediate utilized in the synthesis of high-value active pharmaceutical ingredients such as the antidepressant mirtazapine and the anti-AIDS medication nevirapine. By shifting away from traditional, hazardous chlorinating agents toward a safer bis(trichloromethyl) carbonate system enhanced by a novel additive cocktail, this process represents a significant leap forward in fine chemical synthesis. For procurement leaders and technical directors alike, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with a reduced environmental footprint. The methodology described herein not only optimizes reaction kinetics but also fundamentally alters the waste profile of the production cycle, aligning perfectly with modern green chemistry principles and stringent global regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-chloro-3-cyanopyridine has relied heavily on aggressive chlorinating reagents such as phosphorus oxychloride (POCl3), phosphorus pentachloride (PCl5), thionyl chloride (SOCl2), or sulfuryl chloride (SO2Cl2), all of which present severe operational and environmental drawbacks. When phosphorus-based reagents are employed, the process typically requires reflux conditions and a substantial molar excess of the chlorinating agent, often exceeding three times the molar amount of the starting material, which inevitably leads to the generation of massive quantities of phosphorus-containing wastewater and waste acid that are notoriously difficult and expensive to treat. Alternatively, the use of thionyl chloride or sulfuryl chloride introduces different complications, including the evolution of toxic sulfur dioxide gas and the formation of undesirable 2-position hydroxyl substituted byproducts, which drastically lower reaction selectivity and overall yield. These legacy methods impose a heavy burden on facility infrastructure regarding corrosion resistance, gas scrubbing capabilities, and effluent treatment, ultimately inflating the cost reduction in pharmaceutical intermediates manufacturing and complicating supply chain continuity due to environmental compliance risks.
The Novel Approach
In stark contrast to these archaic techniques, the novel approach detailed in the patent utilizes bis(trichloromethyl) carbonate (BTC) as a superior chlorinating reagent, effectively replacing hazardous liquid or gaseous alternatives with a stable solid that offers enhanced safety and handling characteristics. While earlier attempts to use BTC, such as those reported in patent JP8217753 or CN101659637(A), struggled with mediocre yields ranging from 43% to 85%, the current innovation achieves a breakthrough by incorporating a specific system of additives, organic bases, and phase transfer catalysts. This optimized protocol allows the reaction to proceed under significantly milder conditions, avoiding the extreme thermal stress of reflux while simultaneously driving the conversion efficiency to stably exceed 90%. By eliminating the formation of phosphorus waste and sulfur dioxide emissions, this method not only simplifies the post-reaction workup but also drastically reduces the environmental pollution associated with industrial production, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates in regulated markets.
Mechanistic Insights into Additive-Enhanced BTC Chlorination
The core of this technological advancement lies in the sophisticated interplay between the chlorinating agent and the newly introduced additive system, which fundamentally changes the reaction landscape compared to standard nucleophilic substitution mechanisms. Bis(trichloromethyl) carbonate serves as a safe source of phosgene in situ, reacting with the 3-cyanopyridine N-oxide to form a reactive intermediate that is susceptible to chloride attack, yet without the proper stabilization, this intermediate can degrade or revert, leading to poor yields. The introduction of inorganic salts like sodium chloride and sodium sulfite, in conjunction with a phase transfer catalyst such as benzyltriethylammonium chloride, creates a micro-environment that facilitates the transport of chloride ions into the organic phase while simultaneously scavenging potential oxidative byproducts or stabilizing transition states. This synergistic effect ensures that the chlorination occurs selectively at the 2-position of the pyridine ring, minimizing the formation of regio-isomers or hydrolyzed impurities that typically plague N-oxide chlorination reactions. Furthermore, the use of an organic base like triethylamine acts as an acid scavenger, neutralizing the HCl generated during the transformation and driving the equilibrium toward the desired product without promoting decomposition.
From an impurity control perspective, the mechanism is designed to suppress the formation of the aforementioned 2-hydroxy byproducts and polymeric tars that often result from harsh acidic conditions. The phase transfer catalyst plays a dual role by not only enhancing the reaction rate at lower temperatures, typically between -15°C and 60°C, but also by ensuring a homogeneous distribution of the ionic additives within the organic solvent matrix, likely 1,2-dichloroethane. This homogeneity prevents localized hot spots of high acidity or concentration that could trigger side reactions, thereby ensuring a clean reaction profile that simplifies downstream purification. The result is a crude product that requires minimal processing—often just a simple aqueous wash and decolorization—to achieve high purity specifications, demonstrating a level of process robustness that is critical for GMP manufacturing environments. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate or license this technology for their own high-purity API intermediate production lines.
How to Synthesize 2-Chloro-3-cyanopyridine Efficiently
Implementing this synthesis route requires precise control over reaction parameters to fully realize the yield and purity benefits described in the patent literature. The process begins with the dissolution of the starting material, 3-cyanopyridine N-oxide, in a suitable organic solvent, followed by the sequential addition of the base, additives, and catalyst under strict temperature control to prevent exothermic runaway. Once the system is stabilized at sub-zero temperatures, the bis(trichloromethyl) carbonate solution is introduced slowly to maintain reaction selectivity, after which the mixture is allowed to warm gradually to complete the conversion. The detailed standardized synthetic steps, including specific molar ratios, stirring rates, and quenching procedures necessary to achieve the reported >90% yield, are outlined in the technical guide below for immediate reference by process engineers.
- Dissolve 3-cyanopyridine N-oxide in an organic solvent like 1,2-dichloroethane and cool the mixture to between -5°C and 0°C.
- Add organic base, specific additives such as sodium chloride and sodium sulfite, and a phase transfer catalyst to the reaction system.
- Slowly dropwise add a solution of bis(trichloromethyl) carbonate while maintaining low temperature, then react and purify via extraction and decolorization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this green synthesis route offers profound strategic advantages that extend far beyond simple chemical conversion, impacting the total cost of ownership and risk profile of the supply chain. By transitioning to a process that eliminates heavy metals and corrosive phosphorus waste, manufacturers can significantly reduce the operational expenditures associated with waste disposal, effluent treatment, and regulatory compliance audits, leading to substantial cost savings over the lifecycle of the product. The use of solid bis(trichloromethyl) carbonate instead of gaseous or liquid chlorinating agents enhances onsite safety, reducing the need for specialized containment infrastructure and lowering insurance premiums, which contributes to a more resilient and cost-effective supply network. Furthermore, the simplified workup procedure, which avoids complex distillation or chromatography steps, shortens the production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations without compromising quality.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like phosphorus oxychloride removes the need for costly neutralization and disposal of phosphorus-laden wastewater, directly lowering the variable cost per kilogram of the final intermediate. Additionally, the high reaction yield exceeding 90% means that less raw material is wasted, maximizing the atom economy and ensuring that every dollar spent on starting materials translates efficiently into saleable product inventory. The reduction in energy consumption, achieved by operating at milder temperatures compared to traditional reflux methods, further compounds these savings, making the overall manufacturing process economically superior to legacy routes.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is straightforward, as bis(trichloromethyl) carbonate and common organic solvents are widely available commodities with stable global supply chains, reducing the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions ensures consistent batch-to-batch quality, minimizing the likelihood of failed batches that could disrupt delivery schedules to downstream API manufacturers. This reliability is crucial for long-term supply agreements, providing partners with the confidence that their production timelines will be met consistently without unexpected delays caused by process instability or environmental shutdowns.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind, utilizing standard reactor equipment and avoiding the engineering challenges associated with handling toxic gases on a multi-ton scale. The green nature of the synthesis aligns with increasingly stringent environmental regulations worldwide, future-proofing the supply chain against tightening emission standards and ensuring continuous operation without regulatory interruptions. This compliance advantage is a key differentiator for suppliers looking to partner with multinational corporations that have strict sustainability mandates and carbon footprint reduction goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this green chlorination technology, derived directly from the experimental data and comparative analysis found in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this method for large-scale production of pyridine derivatives. By understanding the specific advantages in yield, safety, and waste reduction, stakeholders can make informed decisions about integrating this pathway into their existing manufacturing portfolios.
Q: Why is bis(trichloromethyl) carbonate preferred over phosphorus oxychloride for this synthesis?
A: Bis(trichloromethyl) carbonate acts as a solid phosgene equivalent, eliminating the generation of difficult-to-treat phosphorus-containing wastewater and acidic waste associated with POCl3, thereby significantly reducing environmental compliance costs.
Q: What role do the additives play in improving the reaction yield?
A: The specific combination of additives like sodium chloride and sodium sulfite, alongside a phase transfer catalyst, stabilizes the reaction intermediates and suppresses side reactions, allowing yields to consistently exceed 90% compared to previous methods.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions, common organic solvents, and avoids hazardous gaseous reagents, making it highly scalable and safer for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-3-cyanopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced, environmentally responsible synthesis routes to meet the evolving needs of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-chloro-3-cyanopyridine meets the highest standards required for API synthesis, thereby supporting our partners in delivering safe and effective medicines to patients worldwide.
We invite you to engage with our technical procurement team to discuss how this green manufacturing technology can be tailored to your specific volume requirements and quality expectations. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this superior process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of innovation, reliability, and sustainable growth.
