Scalable Silver-Catalyzed Synthesis of 3-Isocoumarin-Based Chromones for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking efficient pathways to access complex heterocyclic scaffolds that serve as critical building blocks for drug discovery; in this context, patent CN115504953A introduces a groundbreaking preparation method for 3-isocoumarin-based chromone compounds that addresses many longstanding synthetic challenges. Chromone derivatives are renowned for their diverse biological activities, ranging from anti-inflammatory and anticancer properties to antiviral effects, making them highly coveted targets for medicinal chemistry programs worldwide. The disclosed technology leverages a tandem cyclization strategy involving 2-hydroxy-N,N-dimethylenaminone compounds and 2-alkynylbenzaldehyde compounds, facilitated by a silver catalyst under aerobic conditions. This approach not only streamlines the synthetic sequence by avoiding multiple protection and deprotection steps but also operates under remarkably mild thermal conditions, typically ranging between 40°C and 120°C, which significantly lowers the energy consumption profile compared to traditional high-temperature reflux protocols often required for heterocyclic construction. By integrating this innovative methodology into your supply chain, organizations can secure a reliable pharmaceutical intermediates supplier capable of delivering high-purity scaffolds with improved process economics and reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-substituted chromones has relied heavily on strategies that involve the transition metal-catalyzed coupling of 3-halochromones, a route that imposes significant logistical and financial burdens on manufacturing operations. These conventional pathways necessitate the pre-functionalization of the chromone core, requiring additional synthetic steps to introduce halogen atoms before the final coupling can occur, which inherently reduces the overall atom economy and increases waste generation. Furthermore, the reliance on precious metal catalysts such as palladium often introduces complications regarding residual metal removal, a critical quality attribute for pharmaceutical intermediates that demands rigorous purification processes to meet stringent regulatory standards. The reaction conditions for these traditional methods are frequently harsh, involving strong bases or high pressures that can compromise the integrity of sensitive functional groups present on the molecular scaffold, thereby limiting the scope of accessible derivatives. Consequently, procurement teams often face inflated costs and extended lead times due to the complexity of sourcing specialized halogenated precursors and managing the hazardous waste streams associated with these inefficient processes.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a direct tandem cyclization reaction that constructs the 3-isocoumarin-based chromone skeleton from simple, commercially available starting materials without the need for pre-halogenation. The process employs a silver-based catalytic system, such as silver oxide or silver carbonate, which is generally more cost-effective and easier to handle than palladium counterparts, operating efficiently under an air atmosphere rather than requiring inert gas shielding. As illustrated in the specific reaction conditions below, the transformation proceeds smoothly in common solvents like acetone at moderate temperatures, demonstrating exceptional functional group tolerance and high yields across a broad range of substrates. 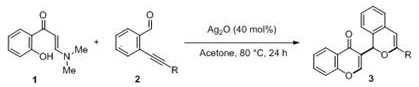 This shift towards oxidative cyclization represents a paradigm shift in cost reduction in fine chemical manufacturing, as it eliminates expensive ligand systems and simplifies the downstream workup procedure to basic extraction and chromatography. For supply chain heads, this translates to enhanced supply chain reliability because the raw materials are commodity chemicals with stable market availability, reducing the risk of production bottlenecks caused by specialized reagent shortages.
This shift towards oxidative cyclization represents a paradigm shift in cost reduction in fine chemical manufacturing, as it eliminates expensive ligand systems and simplifies the downstream workup procedure to basic extraction and chromatography. For supply chain heads, this translates to enhanced supply chain reliability because the raw materials are commodity chemicals with stable market availability, reducing the risk of production bottlenecks caused by specialized reagent shortages.
Mechanistic Insights into Silver-Catalyzed Tandem Cyclization
To fully appreciate the robustness of this synthetic route for R&D directors evaluating process feasibility, it is essential to understand the underlying mechanistic pathway that drives the formation of the 3-isocoumarin-based chromone core. The reaction initiates with the silver-catalyzed self-cyclization of the 2-ethynylbenzaldehyde component, which undergoes an intramolecular nucleophilic attack to form an isochromene intermediate species in situ. This activated intermediate then serves as an electrophile that is susceptible to nucleophilic attack by the 2-hydroxy-N,N-dimethylenaminone compound, forging the critical carbon-carbon bond that links the two heterocyclic systems together. Following this addition step, the intermediate undergoes a subsequent cyclization event accompanied by the elimination of the metal ligand and a dehydrogenation process that restores aromaticity and establishes the final conjugated system.  This cascade sequence is highly efficient because it combines multiple bond-forming events into a single operational step, minimizing the exposure of reactive intermediates to degradation and ensuring a clean impurity profile. The ability to control the regioselectivity and stereochemistry through the choice of silver salt and solvent system allows chemists to fine-tune the reaction for optimal purity, which is paramount when producing high-purity pharmaceutical intermediates for clinical trial materials.
This cascade sequence is highly efficient because it combines multiple bond-forming events into a single operational step, minimizing the exposure of reactive intermediates to degradation and ensuring a clean impurity profile. The ability to control the regioselectivity and stereochemistry through the choice of silver salt and solvent system allows chemists to fine-tune the reaction for optimal purity, which is paramount when producing high-purity pharmaceutical intermediates for clinical trial materials.
Regarding impurity control mechanisms, the mild oxidative conditions employed in this protocol significantly reduce the formation of side products that are commonly observed in harsher transition metal-catalyzed couplings. The use of air as the terminal oxidant avoids the introduction of exogenous oxidizing agents that could lead to over-oxidation of sensitive moieties such as sulfides or amines present on the substrate aryl rings. Moreover, the silver catalyst demonstrates high chemoselectivity, preferentially activating the alkyne moiety for cyclization without affecting other reducible groups like nitro or halo substituents, which expands the chemical space available for derivative synthesis. This selectivity ensures that the crude reaction mixture contains fewer structurally related impurities, thereby simplifying the purification burden and increasing the overall recovery rate of the desired product. For quality assurance teams, this means that achieving stringent purity specifications becomes more predictable and less resource-intensive, facilitating faster release of batches for downstream processing.
How to Synthesize 3-Isocoumarin-Based Chromone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and reproducibility while maintaining safety standards. The general procedure involves charging a reaction vessel with the 2-hydroxy-N,N-dimethylenaminone and 2-alkynylbenzaldehyde substrates in a molar ratio that favors complete conversion, typically using a slight excess of the alkyne component to drive the equilibrium forward. A silver catalyst is added along with a polar aprotic or ketonic solvent, and the mixture is heated with stirring under ambient air pressure until thin-layer chromatography indicates the disappearance of the starting materials. Detailed standardized synthesis steps see the guide below.
- Combine 2-hydroxy-N,N-dimethylenaminone and 2-alkynylbenzaldehyde with a silver catalyst such as silver oxide in a suitable solvent like acetone.
- Heat the reaction mixture to a temperature between 40°C and 120°C under an air atmosphere while monitoring progress via thin-layer chromatography.
- Upon completion, extract the product with ethyl acetate, dry the organic phase, concentrate under reduced pressure, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, adopting this silver-catalyzed methodology offers substantial cost savings and operational efficiencies that directly impact the bottom line of pharmaceutical manufacturing projects. The elimination of pre-functionalized halogenated starting materials removes a significant cost driver, as these specialized reagents often command premium pricing and suffer from volatile supply dynamics in the global chemical market. Furthermore, the replacement of expensive palladium catalysts with abundant silver salts results in drastic simplification of the catalyst recovery process and reduces the financial risk associated with precious metal price fluctuations.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this reaction reduces the number of unit operations required, leading to lower labor costs and decreased solvent consumption throughout the production lifecycle. By avoiding the need for inert atmosphere equipment and high-pressure reactors, capital expenditure for facility upgrades is minimized, allowing existing infrastructure to be utilized for the commercial scale-up of complex pharmaceutical intermediates. The high atom economy of the tandem cyclization ensures that raw material costs are optimized, providing a competitive edge in pricing negotiations for bulk active pharmaceutical ingredient contracts.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetone, ethyl acetate, and simple aromatic aldehydes ensures that raw material procurement is not subject to the geopolitical risks often associated with specialized fine chemical imports. This stability allows for more accurate demand forecasting and inventory management, reducing the lead time for high-purity pharmaceutical intermediates and ensuring consistent delivery schedules to downstream clients. The robustness of the reaction against moisture and oxygen further enhances reliability by reducing the incidence of batch failures due to environmental excursions during transport or storage.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of toxic heavy metals align perfectly with modern green chemistry principles, facilitating easier regulatory approval and reducing the cost of waste treatment and disposal. Scaling this process from gram to kilogram quantities is straightforward because the heat transfer and mixing requirements are not demanding, enabling rapid technology transfer from R&D to production sites. This environmental compatibility supports corporate sustainability goals and helps manufacturers meet increasingly strict emissions standards without compromising on production throughput or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for 3-isocoumarin-based chromones. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation to provide clarity for potential partners.
Q: What are the primary advantages of this silver-catalyzed method over traditional chromone synthesis?
A: This method eliminates the need for pre-functionalized halogenated substrates and expensive transition metal catalysts like palladium, utilizing readily available starting materials and mild oxidative conditions instead.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process operates under air atmosphere at moderate temperatures without stringent inert gas requirements, significantly simplifying reactor setup and reducing operational costs for commercial scale-up.
Q: What is the substrate scope for this 3-isocoumarin-based chromone preparation?
A: The reaction demonstrates broad adaptability, tolerating various substituents including halogens, alkyl groups, and nitro groups on both the enaminone and alkynylbenzaldehyde components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Isocoumarin-Based Chromone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this silver-catalyzed synthesis route for accelerating the development of novel therapeutic agents based on the chromone scaffold. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-isocoumarin-based chromone meets the exacting standards required for global pharmaceutical registration. We understand that time-to-market is critical, and our optimized processes are designed to deliver high-quality intermediates with consistent reproducibility.
We invite you to engage with our technical procurement team to discuss how this innovative technology can be integrated into your specific drug development pipeline. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this greener, more efficient synthetic route for your target molecules. Please contact us today to obtain specific COA data and route feasibility assessments tailored to your unique chemical requirements, and let us demonstrate our commitment to being your trusted partner in chemical innovation.
