Advanced Organocatalytic Synthesis of Chiral Bridged Polycyclic Compounds for Pharmaceutical Applications
Advanced Organocatalytic Synthesis of Chiral Bridged Polycyclic Compounds for Pharmaceutical Applications
The field of organic synthesis is constantly evolving to meet the rigorous demands of modern drug discovery, particularly in the construction of complex polycyclic scaffolds found in bioactive natural products. Patent CN113444108B introduces a groundbreaking methodology for the preparation of 1,4-sulfur bridge polycyclic compounds containing a dihydrobenzofuran structure, a motif prevalent in potent analgesics like morphine. This innovation addresses the critical challenge of asymmetric dearomatization of nitroheteroaromatic compounds, a transformation that has historically been difficult to control with high precision. By leveraging a novel cascade reaction sequence involving 2-nitrobenzofuran and 5H-thiazolone, this technology enables the rapid assembly of molecules with four consecutive chiral centers under remarkably mild conditions. For research and development teams, this represents a significant leap forward in accessing chemically diverse libraries for screening against various pathological targets.
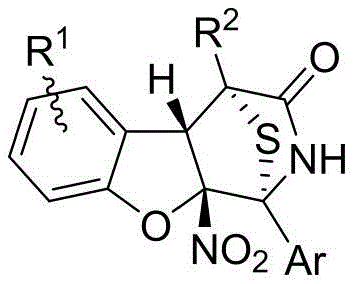
Furthermore, the strategic combination of dihydrobenzofuran and 1,4-thiobridge piperidone substructures within a single molecular framework offers unique pharmacological properties that are highly sought after in medicinal chemistry. The ability to functionalize these core structures easily allows for extensive derivatization, facilitating the optimization of lead compounds for improved efficacy and safety profiles. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such advanced synthetic routes is essential for maintaining a competitive edge in the global market. The implications of this patent extend beyond mere academic interest, providing a robust platform for the industrial-scale production of high-value intermediates required for next-generation antitumor agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of bridged polycyclic compounds has relied heavily on transition metal catalysis or harsh thermal conditions that often compromise the integrity of sensitive functional groups. Conventional asymmetric dearomatization strategies frequently utilize electron-rich aromatic systems like indoles or naphthols, which limits the structural diversity available to chemists exploring nitro-substituted heterocycles. When nitroheteroaromatic compounds are employed, existing methods often struggle to achieve the necessary levels of stereocontrol, resulting in complex mixtures of diastereomers that are costly and time-consuming to separate. Additionally, the use of heavy metal catalysts introduces significant regulatory hurdles regarding residual metal limits in active pharmaceutical ingredients, necessitating expensive purification steps that inflate overall manufacturing costs.
The Novel Approach
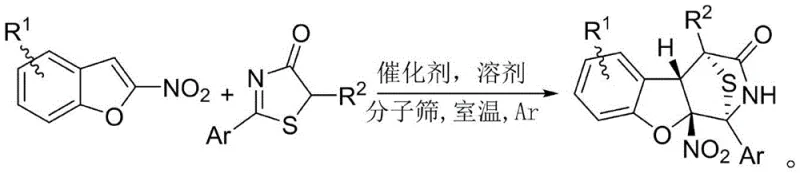
In stark contrast, the methodology disclosed in the patent utilizes an organocatalytic system that operates efficiently at room temperature, eliminating the need for energy-intensive heating or cooling protocols. The reaction proceeds through a highly selective cascade mechanism where 2-nitrobenzofuran acts as the electrophile and 5H-thiazolone serves as the nucleophile, driven by a chiral bifunctional catalyst. This approach not only simplifies the operational procedure but also ensures exceptional stereoselectivity, with diastereomeric ratios consistently exceeding 20:1 and enantiomeric excess values reaching up to 98 percent. The use of dichloromethane as a solvent further enhances the reaction kinetics, providing a balanced environment for the formation of the complex bridged architecture without side reactions.
Mechanistic Insights into Organocatalytic Asymmetric Dearomatization
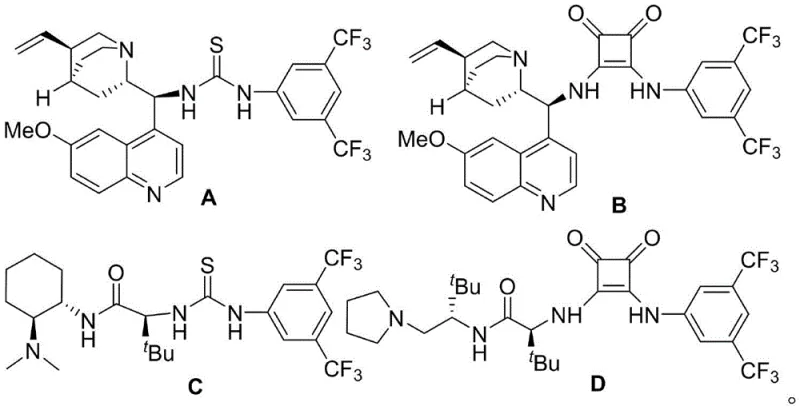
The success of this synthetic route hinges on the precise activation of substrates by a chiral tertiary amine-squaramide bifunctional catalyst, specifically Catalyst D as identified in the experimental data. The mechanism involves an initial Michael addition where the catalyst simultaneously activates the nucleophilic 5H-thiazolone and the electrophilic 2-nitrobenzofuran through a network of hydrogen bonding interactions. This dual activation lowers the energy barrier for the C-C bond formation while rigidly controlling the spatial orientation of the reacting species, thereby dictating the absolute configuration of the newly formed chiral centers. Following the Michael addition, an intramolecular Mannich reaction occurs spontaneously, closing the ring system to form the stable 1,4-sulfur bridge structure with high fidelity.
From an impurity control perspective, the high stereoselectivity of the catalyst minimizes the formation of unwanted diastereomers and enantiomers, which are often the most difficult impurities to remove during purification. The reaction conditions, including the use of molecular sieves to scavenge trace water, prevent hydrolysis of the sensitive thiazolone ring, ensuring a clean reaction profile. This level of control is paramount for pharmaceutical manufacturers who must adhere to strict quality standards regarding impurity profiles. The mechanistic understanding allows process chemists to fine-tune parameters such as substrate ratios and catalyst loading to maximize yield while maintaining the rigorous purity specifications required for clinical-grade materials.
How to Synthesize 1,4-Sulfur Bridge Polycyclic Compounds Efficiently
The practical implementation of this synthesis is designed to be straightforward and scalable, making it highly attractive for process development teams looking to transition from benchtop discovery to pilot plant operations. The protocol requires dissolving the starting materials in anhydrous dichloromethane, adding the chiral catalyst and molecular sieves, and stirring under an inert atmosphere. Detailed standard operating procedures for this specific transformation, including precise stoichiometric ratios and work-up instructions, are critical for reproducibility and are outlined in the technical guide below.
- Dissolve 2-nitrobenzofuran and 5H-thiazolone substrates in anhydrous dichloromethane solvent within a dry reaction vessel.
- Add activated 5A molecular sieves and 20 mol% of chiral catalyst D (squaramide derivative) to the mixture under argon protection.
- Stir the reaction at room temperature until completion, then purify the crude product via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic technology offers substantial strategic benefits that directly impact the bottom line and operational resilience. The elimination of transition metals from the catalytic cycle removes the necessity for specialized metal scavenging resins and extensive analytical testing for heavy metal residues, leading to significant cost reduction in API manufacturing. Furthermore, the mild reaction conditions reduce energy consumption and allow for the use of standard glass-lined reactors without the need for specialized high-pressure or cryogenic equipment, enhancing the flexibility of existing production facilities.
- Cost Reduction in Manufacturing: The use of commercially available organocatalysts and common solvents like dichloromethane drastically simplifies the raw material supply chain, reducing dependency on scarce or expensive metal complexes. By avoiding the complex downstream processing associated with metal removal, manufacturers can achieve substantial cost savings in both material usage and waste disposal. The high yields and selectivity reported in the patent minimize the loss of valuable starting materials, ensuring that the overall process mass intensity is optimized for economic efficiency.
- Enhanced Supply Chain Reliability: Since the reagents and catalysts required for this synthesis are readily accessible from multiple global suppliers, the risk of supply chain disruption is significantly mitigated compared to processes relying on proprietary or single-source metal catalysts. The robustness of the reaction at room temperature means that production can proceed without interruption due to utility failures related to heating or cooling systems. This reliability ensures consistent delivery schedules for high-purity pharmaceutical intermediates, allowing downstream drug developers to maintain their clinical trial timelines without delay.
- Scalability and Environmental Compliance: The simplicity of the reaction setup facilitates easy scale-up from gram to kilogram quantities without the need for extensive re-optimization, supporting the commercial scale-up of complex pharmaceutical intermediates. Additionally, the absence of toxic heavy metals aligns with increasingly stringent environmental regulations and green chemistry principles, reducing the environmental footprint of the manufacturing process. This compliance advantage not only safeguards the company against regulatory penalties but also enhances its reputation as a sustainable partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these novel bridged polycyclic compounds. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their pipelines. Understanding these details is crucial for making informed decisions about process adoption and resource allocation.
Q: What is the stereoselectivity of this novel synthesis method?
A: The method utilizing chiral catalyst D achieves exceptional stereoselectivity, consistently delivering diastereomeric ratios greater than 20:1 and enantiomeric excess values up to 98%.
Q: Why is this organocatalytic route preferred over transition metal catalysis?
A: This metal-free approach eliminates the risk of heavy metal contamination in the final product, significantly simplifying downstream purification and ensuring compliance with strict pharmaceutical regulatory standards.
Q: What are the biological applications of these compounds?
A: Preliminary studies indicate potent antitumor activity against human leukemia K562 and lung cancer A549 cell lines, suggesting strong potential as lead compounds for oncology drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Sulfur Bridge Polycyclic Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalytic methodologies in accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into industrial reality. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite pharmaceutical partners to collaborate with us to leverage this cutting-edge synthesis technology for your antitumor drug programs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals effectively.
