Advanced Two-Step Synthesis of Bifenazate Intermediates for Commercial Scale-Up
The global demand for high-efficiency acaricides continues to drive innovation in the synthesis of key agrochemical intermediates. Patent CN115417797A introduces a groundbreaking preparation method for bifenazate, a potent agent widely used for controlling spider mites in agriculture. This technical disclosure marks a significant departure from traditional synthetic routes that have long plagued the industry with safety hazards and economic inefficiencies. By leveraging a strategic combination of selective electrophilic substitution and transition metal catalysis, the inventors have established a robust pathway that transforms 4-methoxybiphenyl into the target hydrazinoformate derivative with exceptional precision. For R&D directors and procurement strategists, this patent represents a pivotal opportunity to optimize supply chains for agrochemical intermediates, offering a route that is not only chemically elegant but also commercially viable for large-scale manufacturing.
The core innovation lies in the avoidance of complex, multi-step sequences involving hazardous nitration and reduction protocols. Instead, the methodology focuses on a concise two-step transformation that maximizes atom economy while minimizing waste generation. The initial step involves the precise introduction of a bromine atom at the ortho-position relative to the methoxy group, setting the stage for a subsequent coupling reaction. This approach effectively bypasses the need for dangerous diazotization processes found in older patents, thereby reducing the operational risk profile associated with the production of this critical pest control agent. As we delve deeper into the mechanistic and commercial implications, it becomes clear that this technology addresses the triple bottom line of modern chemical manufacturing: safety, cost, and sustainability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing bifenazate have been fraught with significant technical and economic bottlenecks that hinder efficient commercial production. For instance, earlier disclosures such as U.S. Pat. No. 6,938,43 rely on the use of boron trifluoride diethyl etherate, a highly corrosive and hazardous reagent that necessitates specialized equipment and rigorous safety protocols. Furthermore, these legacy processes often involve hydrolysis and decarboxylation steps that can extend reaction times to as long as four days, severely limiting throughput and increasing energy consumption. Another prominent route, described in US6706895, utilizes palladium acetate as a catalyst; while effective, the reliance on precious metals introduces substantial cost volatility and supply chain fragility, making it less attractive for commodity-scale agrochemical intermediate manufacturing. Additionally, processes outlined in US6166243 involve a perilous sequence of nitration, reduction, and diazotization, each step carrying inherent risks of thermal runaway and the formation of unstable intermediates, which are detrimental to safe industrial operation.
The Novel Approach
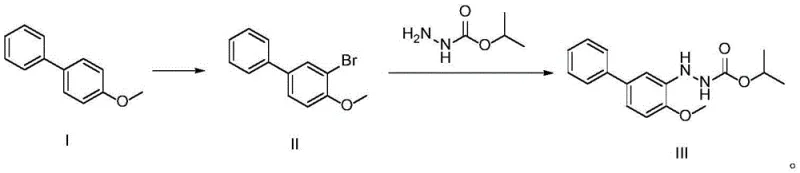
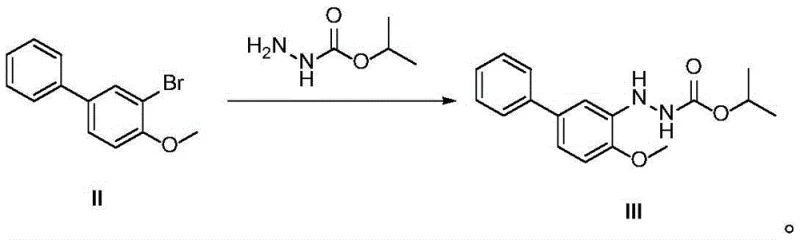
In stark contrast to these cumbersome legacy methods, the novel approach detailed in CN115417797A streamlines the synthesis into a highly efficient two-step sequence that prioritizes both safety and yield. The process initiates with the selective bromination of 4-methoxybiphenyl (Compound I) to generate 3-bromo-4-methoxybiphenyl (Compound II), utilizing mild brominating agents such as N-bromosuccinimide (NBS) or dibromohydantoin. This is followed by a copper-catalyzed coupling reaction with isopropyl hydrazinoformate to directly afford the target bifenazate (Compound III). The elimination of precious metal catalysts and hazardous nitration steps results in a drastically simplified workflow that is inherently safer and more cost-effective. The reaction conditions are notably mild, with the first step proceeding at temperatures between 0°C and 40°C, and the coupling step occurring in a manageable range of 50°C to 95°C. This technological leap facilitates the commercial scale-up of complex agrochemical intermediates by ensuring high reproducibility and minimizing the formation of difficult-to-remove impurities.
Mechanistic Insights into Copper-Catalyzed Coupling and Selective Bromination
The success of this synthetic route hinges on the precise control of regioselectivity during the initial functionalization and the efficiency of the subsequent cross-coupling. In the first step, the methoxy group on the biphenyl ring acts as a strong ortho/para-directing activator. However, due to steric hindrance and the electronic environment of the biphenyl system, the reaction conditions are tuned to favor substitution at the ortho-position relative to the methoxy group (position 3 of the biphenyl system). The use of N-bromosuccinimide (NBS) in solvents like dichloromethane allows for a controlled electrophilic aromatic substitution. The mechanism likely involves the generation of a bromonium ion species which attacks the electron-rich aromatic ring, followed by deprotonation to restore aromaticity. This step is critical because any formation of para-brominated byproducts or poly-brominated species would complicate downstream purification and reduce the overall yield of the final acaricide.
The second step represents the heart of the innovation: a copper-mediated C-N bond formation. Unlike palladium-catalyzed Buchwald-Hartwig couplings which require expensive ligands and inert atmospheres, this protocol utilizes simple divalent copper salts (such as copper sulfate, chloride, or bromide) in conjunction with a phase transfer catalyst (PTC) like tetrabutylammonium bromide. The reaction occurs in a biphasic toluene-water system, where the PTC facilitates the transport of the hydrazinoformate nucleophile into the organic phase containing the aryl bromide. The copper catalyst likely undergoes a redox cycle, facilitating the oxidative addition of the aryl bromide and the subsequent reductive elimination to form the C-N bond. This mechanism is robust and tolerant of various functional groups, ensuring that the hydrazinoformate moiety remains intact without undergoing hydrolysis or decomposition under the reaction conditions.
How to Synthesize Bifenazate Efficiently
Implementing this synthesis requires careful attention to stoichiometry and phase management to maximize the benefits of the biphasic system. The process begins with the dissolution of 4-methoxybiphenyl in a chlorinated solvent, followed by the slow addition of the brominating agent to manage exotherms and ensure high selectivity. Once the intermediate is isolated, typically with yields exceeding 95% and purity greater than 99%, it is subjected to the coupling conditions. The key to success in the second step is the maintenance of the toluene-water interface and the correct loading of the phase transfer catalyst, which ensures efficient mass transfer between the inorganic copper species and the organic substrates. Detailed standardized operating procedures regarding temperature ramps, stirring rates, and work-up protocols are essential for transferring this laboratory success to pilot and production scales.
- Perform selective ortho-bromination of 4-methoxybiphenyl using N-bromosuccinimide (NBS) or dibromohydantoin in a solvent like dichloromethane at 0-40°C to obtain 3-bromo-4-methoxybiphenyl.
- Execute a copper-catalyzed coupling reaction between the brominated intermediate and isopropyl hydrazinoformate in a toluene-water biphasic system with a phase transfer catalyst at 50-95°C.
- Isolate the final bifenazate product through liquid separation, concentration of the organic phase, and crystallization using ethyl acetate or isopropyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers transformative advantages that directly impact the bottom line and operational resilience. By shifting away from palladium-based catalysis and hazardous nitration chemistry, manufacturers can achieve significant cost reductions in agrochemical intermediate manufacturing. The replacement of expensive precious metals with abundant copper salts eliminates a major variable cost driver, while the simplified two-step process reduces the number of unit operations, thereby lowering labor, energy, and waste disposal expenses. Furthermore, the use of readily available starting materials like 4-methoxybiphenyl ensures a stable supply base, mitigating the risks associated with sourcing specialized or regulated precursors. This stability is crucial for maintaining continuous production schedules and meeting the fluctuating demands of the global agricultural market.
- Cost Reduction in Manufacturing: The elimination of palladium catalysts and hazardous reagents like boron trifluoride leads to substantial savings in raw material procurement and waste treatment. The shorter reaction times and milder conditions also translate to lower energy consumption per kilogram of product, enhancing overall process economics without compromising quality.
- Enhanced Supply Chain Reliability: Utilizing common industrial solvents such as dichloromethane, toluene, and ethyl acetate simplifies logistics and inventory management. The robustness of the copper-catalyzed coupling ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed reactions or difficult purifications, thus securing a reliable flow of high-purity intermediates.
- Scalability and Environmental Compliance: The process avoids the generation of heavy metal waste streams associated with palladium and minimizes the use of corrosive acids, aligning with increasingly stringent environmental regulations. The high selectivity and yield demonstrated in the patent examples suggest that the process can be scaled from kilogram to multi-ton quantities with minimal re-optimization, facilitating rapid market entry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bifenazate synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits for potential licensees and manufacturing partners. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production facilities.
Q: Why is the copper-catalyzed route preferred over palladium methods for bifenazate?
A: The copper-catalyzed route eliminates the need for expensive palladium catalysts (like palladium acetate), significantly reducing raw material costs. Furthermore, copper salts are more abundant and easier to handle in large-scale industrial settings compared to precious metals, enhancing supply chain reliability.
Q: What are the safety advantages of this new synthesis method?
A: This method avoids hazardous reagents such as boron trifluoride diethyl etherate and eliminates dangerous unit operations like nitration, diazotization, and long-duration hydrolysis (up to 4 days). The mild reaction conditions (0-40°C for step 1, 50-95°C for step 2) ensure safer operation and lower energy consumption.
Q: How does this process improve scalability for agrochemical production?
A: The process utilizes a simple biphasic toluene-water system and common solvents like dichloromethane, which are standard in chemical manufacturing. The high selectivity and short reaction times (0.5-12 hours per step) allow for rapid throughput, making it highly suitable for commercial scale-up from pilot plants to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bifenazate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN115417797A for securing a competitive edge in the agrochemical sector. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity bifenazate and its key intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to adapt and optimize copper-catalyzed processes allows us to offer clients a supply solution that is both economically attractive and technically superior.
We invite global partners to collaborate with us to leverage this innovative technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional regulatory landscape. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your production of this vital acaricide intermediate is built on a foundation of safety, efficiency, and reliability.
