Scalable Synthesis of Trans-Biphenyl Liquid Crystal Monomers for Advanced Display Technologies
The rapid evolution of flat-panel display technologies, particularly TN, STN, and TFT modes, has created an insatiable demand for high-performance liquid crystal materials with superior electro-optical properties. Patent CN102010287B introduces a groundbreaking methodology for synthesizing (trans)-4-alkyl-3-alkenyl biphenyl derivative monomers, which are critical components known for their high k33/k11 ratios and excellent steepness characteristics. This technical breakthrough addresses the longstanding challenges of safety and scalability in the fine chemical industry by replacing hazardous reagents with robust, industrially viable alternatives. By leveraging a strategic combination of acetal protection, palladium-catalyzed cross-coupling, and stereoselective isomerization, this route ensures the production of high-purity intermediates essential for next-generation optoelectronic devices. For global procurement leaders, understanding this shift from laboratory-scale curiosity to industrial reality is paramount for securing a stable supply chain of advanced electronic chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these vital biphenyl derivatives relied heavily on methodologies that posed significant safety risks and economic inefficiencies, severely limiting their widespread industrial adoption. One prominent prior art route, disclosed by Merck, utilized n-butyllithium in the presence of TMEDA to deprotonate a methyl group, a process that requires handling extremely pyrophoric and explosive reagents at ambient temperatures. 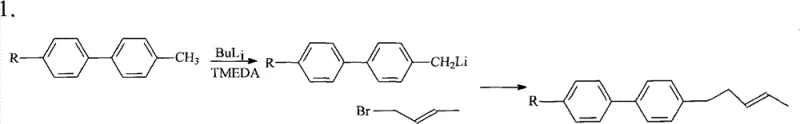 This approach not only demands specialized equipment to mitigate fire hazards but also results in difficult purification processes due to the formation of complex byproduct profiles. Furthermore, alternative conventional pathways often necessitated the use of expensive homogeneous catalysts like PdCl2·dppf, which cannot be recovered or recycled after the reaction, leading to exorbitant raw material costs and substantial heavy metal waste generation that complicates environmental compliance.
This approach not only demands specialized equipment to mitigate fire hazards but also results in difficult purification processes due to the formation of complex byproduct profiles. Furthermore, alternative conventional pathways often necessitated the use of expensive homogeneous catalysts like PdCl2·dppf, which cannot be recovered or recycled after the reaction, leading to exorbitant raw material costs and substantial heavy metal waste generation that complicates environmental compliance.
The Novel Approach
In stark contrast, the novel methodology outlined in the patent data employs a sophisticated yet practical sequence that prioritizes operator safety and process economics without compromising on product quality. The core innovation lies in the utilization of stable aldehyde precursors, such as p-bromocinnamaldehyde or p-bromophenylpropionaldehyde, which are first protected as acetals to prevent unwanted side reactions during the subsequent coupling steps. This strategic protection allows for the use of heterogeneous palladium catalysts, specifically palladium on carbon, which offers the distinct advantage of easy filtration and multiple reuse cycles. By eliminating the need for cryogenic conditions and pyrophoric organolithium reagents, the new process dramatically lowers the barrier to entry for large-scale manufacturing, enabling facilities to produce high-value liquid crystal monomers with reduced capital expenditure on safety infrastructure and waste treatment systems.
Mechanistic Insights into Suzuki Coupling and Stereoselective Isomerization
The heart of this synthetic strategy is a robust Suzuki-Miyaura cross-coupling reaction that efficiently constructs the biphenyl backbone, a structural motif essential for the mesogenic properties of the final liquid crystal material. In this step, the acetal-protected bromo-intermediate reacts with 4-alkylphenylboronic acid under alkaline conditions, facilitated by a palladium catalyst that mediates the oxidative addition and reductive elimination cycles. The choice of a heterogeneous catalyst system is mechanistically significant because it prevents the leaching of palladium into the product stream, thereby simplifying downstream purification and ensuring the final electronic material meets rigorous metal impurity specifications. Following the coupling, if the starting material contained an alkene moiety, a selective hydrogenation step is employed to saturate the double bond, ensuring the correct positioning for the subsequent chain extension via the Wittig reaction.
Following the construction of the biphenyl aldehyde intermediate, the process advances through a Wittig olefination to introduce the alkenyl side chain, initially yielding a mixture of cis and trans isomers. To achieve the desired high-performance characteristics, specifically the high k33/k11 value required for fast response times in displays, a critical catalytic isomerization step is performed. Using catalysts such as benzenesulfinic acid or naphthalene-2-sulfonic acid, the thermodynamic equilibrium is shifted to favor the trans-configuration, which possesses the linear geometry necessary for effective molecular alignment in the liquid crystal phase. This stereochemical control is further refined through repeated recrystallization, a physical purification technique that leverages the solubility differences between isomers to deliver a final product with exceptional optical purity and consistent batch-to-batch performance.
How to Synthesize Trans-4-Alkyl-3-Alkenyl Biphenyl Derivatives Efficiently
The synthesis of these advanced display materials involves a carefully orchestrated five-step sequence that transforms simple aromatic aldehydes into complex mesogenic monomers. The process begins with the protection of the aldehyde functionality, followed by the pivotal carbon-carbon bond formation, deprotection, chain extension, and finally, stereochemical refinement. Each stage is optimized for mild conditions and high yield, ensuring that the overall process remains economically viable for ton-scale production.
- Protect the aldehyde group of p-bromocinnamaldehyde or p-bromophenylpropionaldehyde via acetal formation with glycols.
- Perform Suzuki cross-coupling with 4-alkylphenylboronic acid using a recyclable palladium catalyst to form the biphenyl skeleton.
- Deprotect the acetal to regenerate the aldehyde, followed by a Wittig reaction and catalytic isomerization to achieve high trans-isomer purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this patented synthesis route represents a strategic opportunity to optimize total cost of ownership while mitigating supply risks associated with hazardous chemical logistics. The elimination of n-butyllithium removes the need for specialized storage and handling protocols, thereby reducing insurance premiums and operational downtime related to safety audits. Moreover, the ability to recycle the palladium catalyst multiple times fundamentally alters the cost structure of the manufacturing process, decoupling production volume from the volatile pricing of precious metals. This stability allows for more accurate long-term budgeting and protects margins against fluctuations in the global commodities market, ensuring a reliable supply of critical electronic chemicals.
- Cost Reduction in Manufacturing: The replacement of single-use homogeneous catalysts with recyclable heterogeneous systems significantly lowers the direct material cost per kilogram of finished product. By avoiding the loss of expensive palladium complexes in the waste stream, manufacturers can recover a substantial portion of the catalyst value, leading to drastic improvements in gross margin. Additionally, the milder reaction conditions reduce energy consumption for heating and cooling, further contributing to operational expenditure savings without the need for complex cryogenic infrastructure.
- Enhanced Supply Chain Reliability: Utilizing stable, commercially available starting materials like p-bromophenylpropionaldehyde ensures that production is not bottlenecked by the availability of exotic or highly regulated reagents. The robustness of the synthetic route means that production schedules are less likely to be disrupted by safety incidents or regulatory hurdles associated with transporting dangerous goods. This reliability is crucial for maintaining continuous supply to downstream panel manufacturers who operate on just-in-time inventory models and cannot afford interruptions in their raw material flow.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction steps that translate seamlessly from laboratory glassware to industrial reactors without losing efficiency or selectivity. The reduction in hazardous waste generation, particularly the avoidance of lithium salts and non-recyclable metal complexes, simplifies wastewater treatment and aligns with increasingly stringent global environmental regulations. This eco-friendly profile not only reduces disposal costs but also enhances the brand reputation of the supply chain partners as responsible stewards of sustainable chemistry practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these liquid crystal monomers, based on the detailed specifications provided in the patent documentation. Understanding these nuances helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios.
Q: Why is this synthesis method safer than conventional routes?
A: Unlike traditional methods that rely on highly flammable and explosive n-butyllithium at low temperatures, this patented process utilizes mild reaction conditions and stable reagents like palladium on carbon, significantly reducing operational hazards.
Q: How does this method improve cost efficiency for liquid crystal production?
A: The process replaces expensive, single-use homogeneous palladium catalysts (like PdCl2·dppf) with heterogeneous palladium on carbon, which can be filtered and recycled multiple times, drastically lowering catalyst consumption costs.
Q: What is the purity level achievable with this synthetic route?
A: Through optimized recrystallization and catalytic isomerization steps, the method consistently achieves gas chromatography purity levels exceeding 99.9%, meeting the stringent requirements for high-performance TFT and STN display applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-4-Alkyl-3-Alkenyl Biphenyl Derivative Supplier
As the demand for high-resolution and fast-response display panels continues to surge, the need for a dependable source of high-purity liquid crystal monomers has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to meet the exacting standards of the optoelectronics sector. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of trans-biphenyl derivatives delivers the consistent electro-optical performance required for premium TFT and STN applications. We understand that in the fast-paced world of electronic materials, consistency is key, and our commitment to quality assurance guarantees that your production lines run smoothly without interruption.
We invite you to collaborate with our technical team to explore how this advanced synthesis route can be tailored to your specific formulation needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to our optimized manufacturing process can impact your bottom line. We encourage potential partners to contact our technical procurement team directly to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your transition to these superior materials is seamless, cost-effective, and strategically sound for your long-term growth.
