Revolutionizing Beta-Fluoroalkenyl Alcohol Production via Visible-Light Photocatalysis for Commercial Scale-Up
Introduction to Next-Generation Fluorination Technology
The integration of fluorine atoms into organic scaffolds remains a cornerstone strategy in modern medicinal chemistry, profoundly influencing the metabolic stability, lipophilicity, and bioavailability of drug candidates. Patent CN115448816A introduces a groundbreaking methodology for the synthesis of beta-fluoroalkenyl alcohols, a critical structural motif found in numerous bioactive molecules. This innovation leverages a visible-light-driven photocatalytic hydrogen atom transfer (HAT) strategy, marking a significant departure from conventional thermal processes. By utilizing an organic photocatalyst system comprising 4-CzIPN and quinuclidine, the technology enables the direct coupling of alcohols and gem-difluoroolefins under exceptionally mild conditions. For R&D directors and process chemists, this represents a paradigm shift towards greener, safer, and more efficient synthetic pathways that eliminate the need for hazardous reagents and extreme temperature controls.
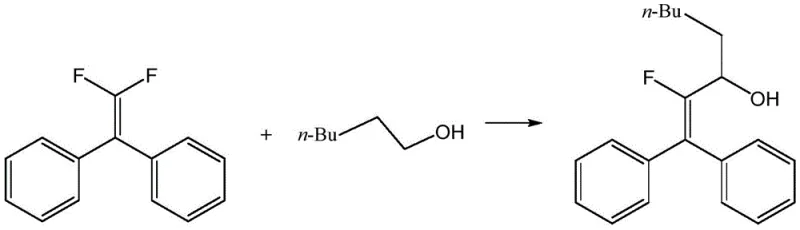
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of beta-fluoroalkenyl alcohol frameworks has relied on multi-step sequences involving highly reactive and dangerous reagents. One prominent prior art route, disclosed in WO 2009017190A1, employs a diaryl ketone reacted with triethyl 2-fluoro-2-phosphoryl acetate in the presence of sodium hydride, followed by a complex downstream transformation using boron tribromide and Grignard reagents. While this method can achieve yields around 85%, the operational hazards are severe, requiring strict anhydrous conditions and handling of pyrophoric materials. Furthermore, the necessity for strong bases limits the functional group tolerance, often leading to side reactions with sensitive moieties present in complex pharmaceutical intermediates.
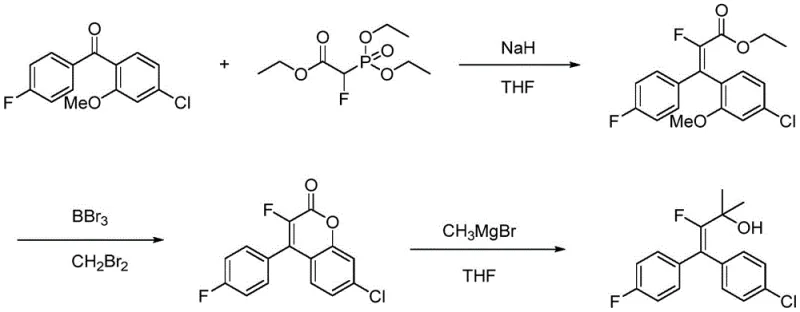
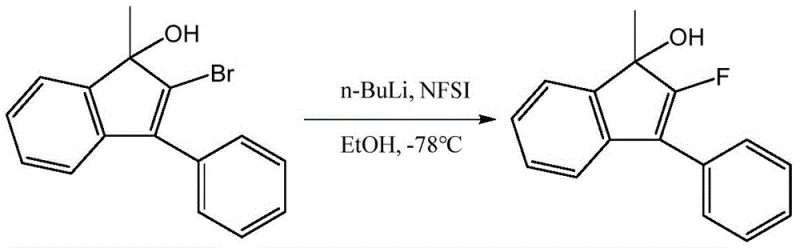
Another established approach, described in WO2008037604 A1, utilizes a cryogenic lithiation strategy involving tert-butyllithium and N-fluorobisbenzenesulfonamide (NFSI) at temperatures as low as -78°C. This pathway suffers from critically low yields, reported at merely 36%, and poses significant safety risks due to the use of organolithium reagents which are prone to thermal runaway if not meticulously controlled. The requirement for deep cryogenic cooling also imposes a heavy energy burden on manufacturing facilities, drastically increasing the cost of goods sold (COGS) and complicating supply chain logistics for large-scale production.
The Novel Approach
In stark contrast, the methodology outlined in CN115448816A circumvents these historical bottlenecks by employing a photoredox catalytic cycle that operates efficiently at room temperature (25°C). The process utilizes blue light irradiation (420-480nm) to activate the 4-CzIPN photocatalyst, initiating a radical cascade that couples readily available alcohols with gem-difluoroolefins. This single-step transformation eliminates the need for pre-functionalized precursors or stoichiometric organometallic reagents. The reaction demonstrates remarkable robustness, achieving isolated yields up to 93% in optimized examples, thereby offering a superior alternative for the reliable beta-fluoroalkenyl alcohol supplier seeking to enhance process safety and efficiency.
Mechanistic Insights into 4-CzIPN Catalyzed Hydrogen Atom Transfer
The efficacy of this synthesis relies on a sophisticated interplay between the organic photocatalyst 4-CzIPN, the hydrogen atom transfer (HAT) mediator quinuclidine, and a Lewis acid co-catalyst. Upon irradiation with blue LEDs, the 4-CzIPN enters an excited state capable of oxidizing quinuclidine to generate a radical cation species. This highly reactive intermediate abstracts a hydrogen atom from the alpha-position of the alcohol substrate, which is activated via coordination with the Lewis acid (such as zinc chloride or lithium chloride). This step generates a nucleophilic alpha-hydroxy carbon radical, a key transient species that drives the subsequent bond formation.
Simultaneously, the reduced form of the photocatalyst acts as a single-electron reductant for the gem-difluoroolefin substrate. This reduction triggers the expulsion of a fluoride ion, generating a stabilized alkenyl radical. The convergence of the alpha-hydroxy radical and the alkenyl radical results in the formation of the desired carbon-carbon bond, yielding the beta-fluoroalkenyl alcohol product. The inclusion of potassium phosphate serves a dual purpose: it acts as a base to neutralize the generated hydrogen fluoride byproduct and helps maintain the catalytic turnover. This mechanistic elegance ensures high atom economy and minimizes the generation of toxic waste streams.
How to Synthesize Beta-Fluoroalkenyl Alcohol Efficiently
Implementing this photocatalytic protocol requires careful attention to the exclusion of oxygen and moisture, as these environmental factors can quench the radical intermediates and deactivate the catalyst. The standard procedure involves charging a Schlenk bottle with the catalyst system and substrates in a polar aprotic solvent like DMSO, followed by rigorous degassing. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining 4-CzIPN, quinuclidine, a Lewis acid (such as zinc chloride), potassium phosphate, gem-difluoroalkene, and alcohol in a solvent like DMSO within a Schlenk bottle under inert atmosphere.
- Degas the solution thoroughly to remove oxygen and moisture, ensuring the stability of the radical intermediates generated during the photocatalytic cycle.
- Irradiate the mixture with blue LED light (420-480nm) at room temperature (approx. 25°C) for 24 hours, followed by aqueous workup and column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from cryogenic organometallic chemistry to ambient photocatalysis offers transformative economic and logistical benefits. The elimination of deep-freeze infrastructure (-78°C) and the associated energy costs for maintaining such temperatures represents a significant reduction in operational expenditure. Furthermore, the replacement of hazardous reagents like tert-butyllithium and Grignard reagents with stable, shelf-stable organic catalysts and simple salts drastically lowers the costs associated with hazardous waste disposal and safety compliance protocols.
- Cost Reduction in Manufacturing: The new process achieves substantial cost savings by removing the need for expensive stoichiometric fluorinating agents and organometallic reagents. By utilizing simple alcohols and gem-difluoroolefins as starting materials, the raw material costs are significantly lowered. Additionally, the high yields observed (up to 93%) minimize material loss and reduce the burden on downstream purification processes, leading to a more economical overall production cost structure for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents such as 4-CzIPN, quinuclidine, and common Lewis acids ensures a robust supply chain that is less susceptible to the volatility often seen with specialized organometallic suppliers. The mild reaction conditions allow for processing in standard glass-lined or stainless steel reactors without the need for specialized cryogenic jackets, thereby increasing the availability of suitable manufacturing capacity and reducing lead time for high-purity beta-fluoroalkenyl alcohols.
- Scalability and Environmental Compliance: The use of visible light and ambient temperature conditions aligns perfectly with green chemistry principles, facilitating easier regulatory approval and environmental compliance. The process generates minimal hazardous waste compared to traditional methods involving heavy metals or strong acids, simplifying effluent treatment. This scalability ensures that the commercial scale-up of complex fluorinated intermediates can be achieved rapidly to meet market demand without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this photocatalytic technology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent literature to assist technical teams in evaluating feasibility.
Q: What are the primary advantages of this photocatalytic method over traditional Grignard routes?
A: Unlike traditional methods requiring harsh reagents like sodium hydride and Grignard reagents at low temperatures, this photocatalytic approach operates at room temperature using visible light, significantly improving safety and reducing energy consumption associated with cryogenic cooling.
Q: Is the 4-CzIPN catalyst reusable or expensive to source?
A: While 4-CzIPN is a specialized organic photocatalyst, it is used in very low catalytic loadings (approx. 3 mol%), which mitigates cost impact. Furthermore, the elimination of stoichiometric amounts of expensive organometallic reagents offsets the catalyst cost.
Q: Can this method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the mild reaction conditions (room temperature, ambient pressure) and the use of standard LED light sources make this process highly amenable to flow chemistry and large-scale batch reactors, facilitating commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Fluoroalkenyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced fluorination technologies play in accelerating drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the 4-CzIPN photocatalytic system can be seamlessly translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of beta-fluoroalkenyl alcohol meets the exacting standards required by global pharmaceutical clients.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and reduce your overall development timeline.
