Scalable Synthesis of Beta-Phenyl-Gamma-Butyrolactone Using Recyclable Ionic Cobalt Catalysts
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient routes for synthesizing chiral lactones, which serve as critical building blocks for bioactive molecules. Patent CN115181081A introduces a groundbreaking methodology for the synthesis of beta-phenyl-gamma-butyrolactone, utilizing a novel ionized metal cobalt complex as a chiral catalyst. This technology represents a significant leap forward in asymmetric Baeyer-Villiger oxidation, specifically addressing the longstanding challenge of balancing high enantioselectivity with catalyst recyclability. By employing hydrogen peroxide as a green oxidant in conjunction with this specialized ionic catalyst, the process achieves superior conversion rates while eliminating the need for hazardous organic peroxyacids. The innovation lies not just in the chemical transformation itself, but in the engineering of the catalyst's physicochemical properties to facilitate seamless phase separation, thereby offering a robust solution for the scalable production of high-purity pharmaceutical intermediates.
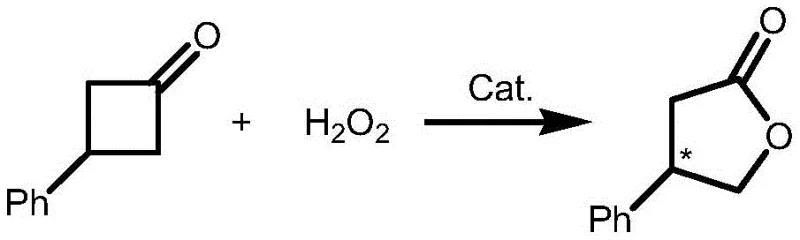
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing chiral lactones often rely on stoichiometric oxidants such as meta-chloroperoxybenzoic acid (mCPBA) or other organic peroxyacids, which present severe drawbacks for large-scale manufacturing. These reagents are inherently unstable, posing significant safety risks regarding explosion and thermal runaway during storage and handling. Furthermore, the atom economy of such reactions is poor, generating substantial quantities of acidic waste byproducts that require complex and costly neutralization and disposal procedures. From a catalytic perspective, conventional homogeneous metal complexes used in asymmetric oxidation are typically lipophilic, meaning they dissolve in the same organic phase as the product. This creates a major downstream processing bottleneck, as separating trace amounts of toxic heavy metals from the final API intermediate often necessitates expensive chromatography or multiple recrystallization steps, drastically inflating production costs and extending lead times.
The Novel Approach
The methodology disclosed in the patent circumvents these issues by integrating green chemistry principles with advanced catalyst design. Instead of dangerous peroxyacids, the process utilizes aqueous hydrogen peroxide, which is inexpensive, safe to handle, and produces water as the sole byproduct, perfectly aligning with modern environmental regulations. The core innovation is the deployment of an ionized cobalt complex that functions as a phase-transfer catalyst. This unique structural feature allows the reaction to proceed efficiently at the interface of an organic-water biphasic system without the addition of external surfactants. Consequently, upon completion of the reaction, the catalyst naturally partitions into the aqueous layer while the beta-phenyl-gamma-butyrolactone product resides in the organic layer. This intrinsic phase separation capability simplifies the workup procedure to a simple extraction, enabling the direct recovery and reuse of the valuable cobalt catalyst, thus solving the critical trade-off between high enantioselectivity and catalyst recyclability.
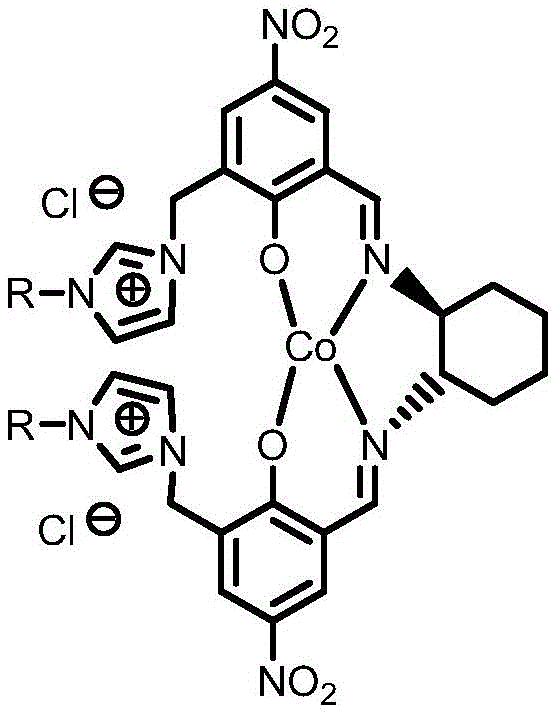
Mechanistic Insights into Ionic Cobalt-Catalyzed Asymmetric Oxidation
The efficacy of this synthesis relies on the sophisticated molecular architecture of the ionized cobalt complex, which acts as both the chiral inducer and the phase-transfer agent. The catalyst is constructed from a chiral Schiff base ligand derived from 5-nitrosalicylaldehyde and 1,2-cyclohexanediamine, coordinated to a central cobalt ion. Crucially, the ligand framework is functionalized with N-alkylimidazolium groups, imparting a permanent positive charge to the complex. This ionic character dramatically alters the solubility profile of the catalyst, rendering it hydrophilic enough to reside in the aqueous phase yet sufficiently amphiphilic to interact with the organic substrate at the phase boundary. During the catalytic cycle, the cobalt center activates the hydrogen peroxide to form a reactive peroxo-species, which then attacks the carbonyl carbon of the 3-phenylcyclobutanone. The chiral environment provided by the cyclohexanediamine backbone dictates the facial selectivity of this oxygen insertion, ensuring the formation of the desired enantiomer of the lactone with high fidelity.
Impurity control in this system is inherently managed through the biphasic reaction design and the specific choice of ligands. The use of 5-nitro substituents on the salicylaldehyde moiety enhances the electron-withdrawing character of the ligand, which stabilizes the metal center against oxidative degradation during the harsh conditions of peroxide oxidation. This stability prevents the leaching of free cobalt ions into the product stream, a common source of contamination in metal-catalyzed reactions. Furthermore, because the catalyst is confined to the aqueous phase, organic-soluble side products or unreacted starting materials remain in the organic phase, facilitating a cleaner separation. The absence of added surfactants is particularly beneficial for purity; traditional surfactants often form emulsions that trap impurities and are notoriously difficult to remove completely, whereas this surfactant-free ionic system allows for sharp phase boundaries and high-purity isolation of the beta-phenyl-gamma-butyrolactone product after simple drying and concentration.
How to Synthesize Beta-Phenyl-Gamma-Butyrolactone Efficiently
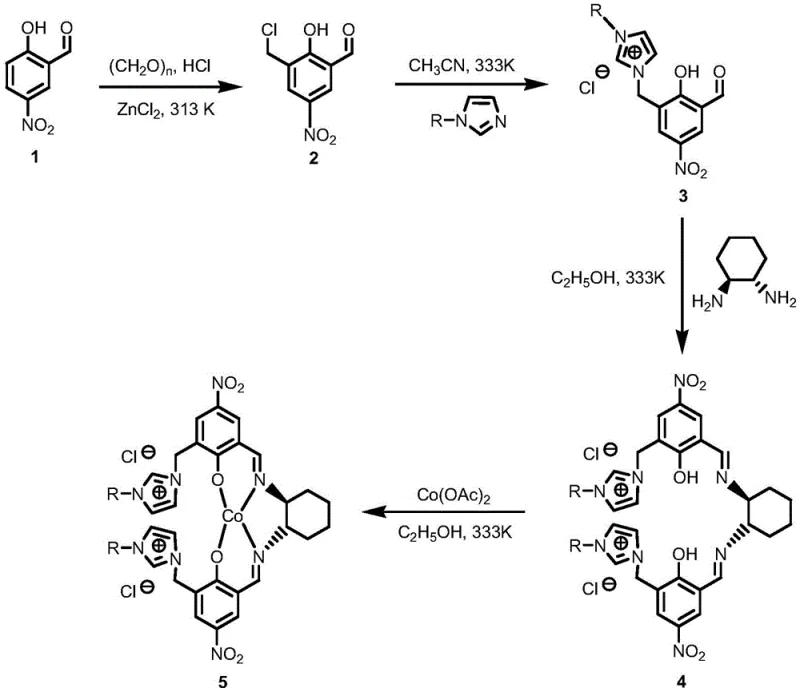
The practical implementation of this synthesis involves a streamlined multi-step preparation of the catalyst followed by a straightforward oxidation protocol. The catalyst synthesis begins with the chloromethylation of 5-nitrosalicylaldehyde, followed by quaternization with an N-alkylimidazole to create the ionic liquid precursor. This precursor is then condensed with 1,2-cyclohexanediamine to form the chiral ligand, which is finally metallated with cobalt acetate to yield the active ionic complex. For the oxidation reaction, the substrate 3-phenylcyclobutanone is suspended in deionized water along with the catalyst, and 30wt% hydrogen peroxide is added dropwise under controlled temperature conditions ranging from 10°C to 40°C. The detailed standardized synthesis steps, including precise molar ratios, stirring speeds, and workup parameters, are outlined in the guide below.
- Preparation of the ionic cobalt catalyst involving chloromethylation of 5-nitrosalicylaldehyde, quaternization with N-alkylimidazole, complexation with 1,2-cyclohexanediamine, and final coordination with cobalt acetate.
- Suspension of 3-phenylcyclobutanone and the ionic cobalt catalyst in deionized water within a three-necked flask equipped with a reflux condenser and thermometer.
- Dropwise addition of 30wt% hydrogen peroxide solution at controlled temperatures (10-40°C), followed by stirring for 4-8 hours, extraction with ethyl acetate, and recovery of the aqueous catalyst phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic cobalt-catalyzed process offers transformative economic and logistical benefits over legacy manufacturing technologies. The shift from stoichiometric organic oxidants to catalytic hydrogen peroxide fundamentally alters the cost structure of the synthesis by reducing raw material expenses and minimizing waste disposal fees. The elimination of hazardous peroxyacids also lowers insurance premiums and safety compliance costs associated with storing and transporting explosive reagents. Moreover, the inherent recyclability of the catalyst means that the effective cost per kilogram of the precious metal catalyst is amortized over multiple batches, leading to substantial long-term savings. The simplified downstream processing, which avoids complex chromatographic purification or surfactant removal, significantly reduces solvent consumption and energy usage during concentration and drying, further driving down the overall cost of goods sold (COGS) for this high-value intermediate.
- Cost Reduction in Manufacturing: The transition to this catalytic system eliminates the need for purchasing expensive stoichiometric oxidants like mCPBA, which are not only costly but also generate equivalent amounts of acidic waste that require neutralization. By using cheap hydrogen peroxide and a reusable catalyst, the direct material costs are significantly reduced. Additionally, the removal of surfactants from the process formulation saves on auxiliary chemical costs and reduces the complexity of the workup, which translates to lower labor and utility costs per batch. The ability to recover the catalyst from the aqueous phase via simple evaporation avoids the loss of expensive chiral ligands, ensuring that the capital invested in catalyst preparation yields maximum return over extended production campaigns.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as hydrogen peroxide and cobalt salts mitigates the supply risk associated with specialized, custom-synthesized oxidants that may have limited suppliers or long lead times. The robustness of the catalyst, demonstrated by its ability to maintain performance over multiple cycles, ensures consistent production throughput without frequent interruptions for catalyst replenishment. This stability allows for more accurate production planning and inventory management, reducing the risk of stockouts for downstream API manufacturers. Furthermore, the mild reaction conditions (10-40°C) reduce the dependency on specialized high-pressure or cryogenic equipment, making the process adaptable to a wider range of manufacturing facilities and enhancing overall supply chain flexibility.
- Scalability and Environmental Compliance: The green chemistry profile of this method, characterized by water as the only byproduct and the absence of halogenated waste from oxidants, greatly simplifies environmental permitting and wastewater treatment. Scaling up exothermic oxidation reactions is traditionally risky, but the biphasic nature of this system provides a thermal buffer, as the aqueous phase helps dissipate heat, making the transition from pilot plant to commercial scale safer and more predictable. The ease of separating the catalyst into the aqueous stream means that heavy metal discharge can be tightly controlled and treated centrally, ensuring strict adherence to increasingly stringent global environmental regulations regarding heavy metal residues in pharmaceutical effluents.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this asymmetric oxidation technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing workflows and for quality assurance teams establishing specifications for the final intermediate.
Q: What are the primary advantages of using the ionic cobalt catalyst described in CN115181081A over traditional methods?
A: The primary advantage lies in the catalyst's ionic nature, which enables effective phase-transfer catalysis without additional surfactants. Unlike traditional oil-soluble metal complexes that are difficult to separate from the product, this ionic catalyst remains in the aqueous phase, allowing for simple decantation, recovery, and reuse while maintaining high enantioselectivity and yield.
Q: How does this method address environmental concerns in Baeyer-Villiger oxidation?
A: This method replaces hazardous organic peroxyacids, which generate stoichiometric amounts of acidic waste, with hydrogen peroxide (H2O2). The only byproduct of the oxidation is water, aligning with green chemistry principles. Furthermore, the ability to recycle the catalyst multiple times significantly reduces heavy metal waste discharge.
Q: Is the catalyst stable enough for multiple reaction cycles?
A: Yes, experimental data indicates robust stability. The patent demonstrates that after three consecutive recycling runs, the catalyst maintains a conversion rate of approximately 94% and an enantioselectivity of around 62%, proving its suitability for continuous or batch-wise industrial processes without significant loss of activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Phenyl-Gamma-Butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the ionic cobalt-catalyzed pathway described in CN115181081A and possesses the technical expertise to optimize this route for your specific production needs. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of beta-phenyl-gamma-butyrolactone delivered meets the highest standards of enantiomeric excess and chemical purity required for drug substance synthesis.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this green catalytic process for your specific volume requirements. We encourage you to contact us today to discuss your project specifics,索取 specific COA data for our reference standards, and receive comprehensive route feasibility assessments tailored to your timeline and budget constraints. Let us help you secure a sustainable and cost-effective supply of this vital chiral intermediate.
