Advanced Synthetic Route for (5-Fluoropyrimidin-4-yl) Methanol: Scalable Manufacturing for Global Pharma
Introduction to Novel Fluorinated Pyrimidine Synthesis
The pharmaceutical industry continuously demands efficient and scalable routes for complex heterocyclic building blocks, particularly those containing fluorine atoms which are crucial for metabolic stability and bioavailability in modern drug design. Patent CN111675659B, published in March 2022, introduces a groundbreaking synthetic methodology for producing (5-fluoropyrimidin-4-yl) methanol, a valuable intermediate in the construction of kinase inhibitors and antiviral agents. This technology represents a significant leap forward by establishing a concise two-step pathway that transforms readily available chloro-fluoro-methylpyrimidine precursors into the target alcohol with high selectivity. For R&D directors and process chemists, this patent offers a robust alternative to traditional methods that often suffer from harsh conditions or excessive step counts. By leveraging a combination of radical oxidation and catalytic hydrogenolysis, the inventors have created a process that is not only chemically elegant but also inherently safer and more controllable for industrial application.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated pyrimidine alcohols has been plagued by significant operational challenges that hinder large-scale production and increase overall manufacturing costs. Traditional routes often rely on multi-step sequences involving protecting group strategies, cryogenic conditions, or the use of highly toxic and expensive reagents that complicate waste management and safety protocols. Furthermore, many existing methods struggle with regioselectivity, leading to difficult-to-separate impurities that compromise the purity profile required for pharmaceutical grade intermediates. The reliance on unstable intermediates or sensitive organometallic reagents in conventional approaches frequently results in batch-to-batch variability, making it difficult for supply chain managers to guarantee consistent delivery timelines. These inefficiencies create bottlenecks in the development of new APIs, forcing companies to seek more reliable and streamlined synthetic solutions that can withstand the rigors of commercial scale-up without sacrificing quality.
The Novel Approach
In stark contrast to these legacy issues, the methodology disclosed in CN111675659B utilizes a direct and rational design that minimizes chemical manipulation while maximizing yield potential. The novel approach initiates with a selective oxidation of the methyl group on the pyrimidine ring, bypassing the need for pre-functionalization or complex activation steps. This is followed by a highly specific dechlorination reaction that cleanly removes the chlorine atom without affecting the sensitive fluorine substituent or the newly formed hydroxymethyl group. This strategic simplification of the synthetic tree reduces the number of unit operations, thereby lowering the cumulative loss of material and energy consumption throughout the process. For procurement teams, this translates to a reduction in the variety of raw materials needed, simplifying inventory management and reducing the risk of supply disruptions associated with niche reagents. The operational simplicity allows for easier control of reaction parameters, ensuring that the process remains robust even when transferred from laboratory glassware to multi-ton reactor systems.
Mechanistic Insights into Radical Oxidation and Catalytic Hydrogenolysis
The core of this synthetic innovation lies in the precise orchestration of two distinct chemical transformations that work in harmony to construct the target molecule. The first step involves a radical-mediated oxidation where dibenzoyl peroxide acts as a potent initiator to abstract hydrogen atoms from the methyl group of the starting pyrimidine. In the presence of a strong acid promoter such as trifluoromethanesulfonic acid or acetic acid, this radical species is trapped by methanol or oxygen equivalents to form the hydroxymethyl intermediate, Compound B. This mechanism is particularly advantageous because it operates under relatively mild thermal conditions, typically around 68°C, which prevents the degradation of the fluorinated heterocyclic core. The second stage employs a heterogeneous catalytic hydrogenolysis using palladium-based catalysts like Pd/C or Lindlar catalyst. Under a hydrogen pressure of approximately 0.6 MPa and moderate temperatures of 45°C, the catalyst facilitates the cleavage of the carbon-chlorine bond. The presence of sodium acetate serves as a crucial acid scavenger, neutralizing the hydrochloric acid byproduct and preventing acid-catalyzed decomposition of the product, thus ensuring high purity levels exceeding 96%.
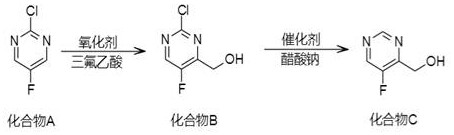
Understanding the impurity profile is critical for R&D directors aiming to replicate this process, and the patent provides clear guidance on how side reactions are mitigated. The use of specific acid promoters in the oxidation step helps to direct the reaction pathway towards the desired alcohol rather than over-oxidation to the carboxylic acid or aldehyde. Similarly, the choice of catalyst and the addition of sodium acetate in the second step are vital for suppressing potential defluorination, a common side reaction in fluorinated aromatic systems under hydrogenation conditions. By maintaining strict control over the pH and pressure parameters, the process ensures that the fluorine atom remains intact, preserving the electronic properties essential for the biological activity of downstream drug candidates. This level of mechanistic control demonstrates a deep understanding of heterocyclic chemistry, offering a reliable blueprint for synthesizing high-purity intermediates that meet stringent regulatory standards for pharmaceutical applications.
How to Synthesize (5-Fluoropyrimidin-4-yl) Methanol Efficiently
Implementing this synthesis requires adherence to specific operational protocols to ensure safety and reproducibility, starting with the preparation of the oxidation reaction mixture under inert atmosphere. The detailed standardized synthesis steps involve precise mass ratios of the starting material, oxidant, and acid, followed by a controlled heating period to drive the conversion to the intermediate alcohol. Once the intermediate is isolated, the subsequent hydrogenation step must be conducted in a pressure-rated vessel with careful monitoring of hydrogen uptake to determine reaction completion.
- Oxidize Compound A (2-chloro-5-fluoro-4-methylpyrimidine) using dibenzoyl peroxide and acid in methanol at 68°C to form Compound B.
- Subject Compound B to catalytic hydrogenolysis using Pd/C or Lindlar catalyst with sodium acetate in alcohol solvent at 45°C and 0.6MPa pressure.
- Purify the final reaction mixture via filtration and column chromatography to isolate (5-fluoropyrimidin-4-yl) methanol with >96% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits that directly address the pain points of cost and supply chain reliability in the fine chemical sector. The elimination of complex multi-step sequences significantly reduces the overall processing time and labor costs associated with manufacturing, allowing for faster turnaround times from order to delivery. By utilizing common and commercially available reagents such as dibenzoyl peroxide and standard palladium catalysts, the process avoids the volatility and high costs associated with exotic or custom-synthesized reagents. This accessibility of raw materials ensures a stable supply chain, reducing the risk of production halts due to material shortages which is a critical concern for supply chain heads managing global API projects. Furthermore, the mild reaction conditions reduce the energy load on the facility, contributing to lower utility costs and a smaller environmental footprint, aligning with modern sustainability goals.
- Cost Reduction in Manufacturing: The streamlined nature of this two-step process inherently lowers the cost of goods sold by minimizing the number of isolation and purification stages required. Eliminating the need for cryogenic cooling or high-pressure specialized equipment beyond standard autoclaves reduces capital expenditure and maintenance costs for manufacturing partners. The high selectivity of the reactions means less material is lost to byproducts, improving the overall mass balance and reducing the cost per kilogram of the final active intermediate. Additionally, the ability to use standard solvents like methanol and ethanol simplifies solvent recovery and recycling processes, further driving down operational expenses without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for reagents and solvents ensures that the supply chain is resilient against market fluctuations and geopolitical disruptions. Since the process does not depend on single-source suppliers for niche catalysts or reagents, procurement managers have the flexibility to source materials from multiple vendors, securing better pricing and delivery terms. The robustness of the reaction conditions also means that the process can be easily transferred between different manufacturing sites or CDMO partners without extensive re-validation, providing strategic flexibility in production planning. This reliability is paramount for maintaining continuous supply to downstream pharmaceutical customers who depend on just-in-time delivery models for their own clinical or commercial programs.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily manageable in large-scale reactors without significant exothermic risks or safety hazards. The use of heterogeneous catalysts allows for easy filtration and recovery, minimizing heavy metal contamination in the waste stream and simplifying compliance with environmental regulations. The reduction in step count naturally leads to a decrease in the total volume of waste generated per unit of product, supporting greener chemistry initiatives and reducing waste disposal costs. This combination of safety, scalability, and environmental friendliness makes the technology highly attractive for long-term commercial partnerships focused on sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route based on the patent data. These insights are derived directly from the experimental examples and technical disclosures to provide clarity for process development teams.
Q: What are the critical reaction conditions for the oxidation step?
A: The oxidation step requires precise temperature control at 68°C under nitrogen protection, utilizing dibenzoyl peroxide as the oxidant and trifluoromethanesulfonic acid or acetic acid as the promoter in a methanol solvent system.
Q: Which catalysts are suitable for the dechlorination process?
A: The patent specifies the use of palladium carbon (Pd/C) or Lindlar catalysts, which effectively facilitate the removal of the chlorine atom at the 2-position while preserving the fluorine substituent and the newly formed hydroxymethyl group.
Q: How does this method improve upon existing synthetic routes?
A: This method offers a significantly shorter synthetic route with easily available raw materials and mild reaction conditions, eliminating the need for complex multi-step sequences often associated with fluorinated pyrimidine derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (5-Fluoropyrimidin-4-yl) Methanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development pipeline, and we are fully equipped to bring this advanced synthetic technology to life. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (5-fluoropyrimidin-4-yl) methanol meets the exacting standards required for pharmaceutical applications. Our commitment to technical excellence means we can navigate the complexities of fluorinated chemistry with precision, delivering materials that accelerate your timeline to market.
We invite you to collaborate with us to leverage this innovative synthesis for your specific project needs, unlocking new efficiencies in your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic goals for cost-effective and reliable intermediate sourcing.
