Scalable Visible Light Mediated Synthesis of Indole Allyl Alcohol Compounds for Advanced Pharmaceutical Manufacturing
Scalable Visible Light Mediated Synthesis of Indole Allyl Alcohol Compounds for Advanced Pharmaceutical Manufacturing
The landscape of organic synthesis is undergoing a profound transformation driven by the urgent need for greener, more sustainable manufacturing processes, particularly within the high-value sector of pharmaceutical intermediates. A pivotal advancement in this domain is detailed in Chinese Patent CN112778191A, which discloses a highly efficient, visible light-mediated method for synthesizing allyl alcohol compounds containing an indole skeleton. This technology represents a significant leap forward from traditional thermal methods, leveraging the power of photoredox catalysis to construct complex molecular architectures under exceptionally mild conditions. For R&D directors and process chemists, this patent offers a robust pathway to access critical building blocks that are essential for the development of novel bioactive molecules, while simultaneously addressing the growing regulatory pressure to reduce hazardous waste and energy consumption in chemical production facilities globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of allyl alcohol compounds, which serve as versatile intermediates for allylation reactions and the preparation of amino compounds, has relied heavily on classical nucleophilic addition reactions or the selective reduction of alpha,beta-unsaturated carbonyl compounds. These conventional strategies, while established, are fraught with significant inefficiencies that hinder modern large-scale manufacturing. They typically necessitate the use of stoichiometric amounts of harsh reagents and additives, leading to poor atom economy and the generation of substantial quantities of chemical waste that require costly disposal. Furthermore, these methods often demand rigorous reaction conditions, such as extreme temperatures or pressures, which not only increase energy costs but also pose safety risks in a commercial plant setting. The reliance on equivalent reagents also complicates the purification process, often requiring extensive chromatographic separations that reduce overall throughput and increase the cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
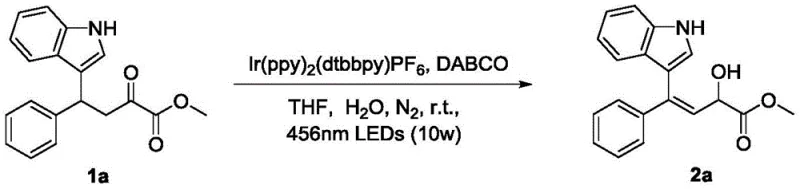
In stark contrast to these legacy methods, the technology described in Patent CN112778191A introduces a revolutionary visible light-mediated protocol that fundamentally alters the reaction landscape. By utilizing a photocatalytic system driven by low-energy 456nm LEDs, this method achieves the transformation of beta-indole-carbonyl compounds into valuable allyl alcohol derivatives with remarkable efficiency. The process operates under inert gas atmospheres at mild temperatures ranging from 0°C to 60°C, eliminating the need for thermal activation that can degrade sensitive functional groups. This approach not only ensures high yields and excellent atom economy but also drastically simplifies the operational workflow, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates. The use of catalytic amounts of reagents rather than stoichiometric equivalents marks a paradigm shift towards sustainable chemistry, aligning perfectly with the goals of modern green manufacturing initiatives.

Mechanistic Insights into Visible Light Photocatalytic Dehydrogenation
The core of this innovative synthesis lies in the sophisticated interplay between the photocatalyst, the base, and the visible light source, which together facilitate a dehydrogenative transformation that would otherwise be thermodynamically challenging. The reaction employs potent iridium-based photocatalysts, such as Ir(ppy)2(dtbbpy)PF6, which absorb photons from the 456nm LED source to reach an excited state capable of single-electron transfer processes. In the presence of a base like DABCO (1,4-Diazabicyclo[2.2.2]octane) and a protic additive such as water, the system generates reactive radical intermediates from the beta-indole-carbonyl substrate. This radical pathway allows for the formation of the carbon-carbon double bond with high stereoselectivity, often favoring the Z-isomer as evidenced by Z/E ratios reaching up to 15.00:1 in specific examples. The inclusion of water as an additive is particularly intriguing, as it likely plays a crucial role in proton shuttling or stabilizing transition states, thereby enhancing the overall reaction rate and selectivity without the need for toxic organic co-solvents in large excess.
From an impurity control perspective, this photocatalytic mechanism offers distinct advantages over thermal radical initiators which often produce a broad spectrum of side products due to non-selective bond homolysis. The precise energy input provided by the monochromatic 456nm LEDs ensures that only the photocatalyst is activated, minimizing background thermal reactions that lead to decomposition or polymerization of the sensitive indole scaffold. This high level of control translates directly into a cleaner crude reaction profile, reducing the burden on downstream purification units. For quality assurance teams, this means a more consistent impurity profile and a higher likelihood of meeting stringent pharmacopeial standards for residual solvents and heavy metals, given that the catalyst loading is kept extremely low, typically between 0.5 mol% and 2 mol%. The ability to tune the reaction outcome by simply adjusting the irradiation time or the nature of the photocatalyst provides process chemists with a powerful toolkit for optimizing the synthesis of diverse analogues.
How to Synthesize Indole Allyl Alcohol Efficiently
Implementing this visible light-mediated synthesis in a laboratory or pilot plant setting requires careful attention to the specific parameters outlined in the patent to ensure reproducibility and maximum yield. The procedure is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic cooling systems that are often associated with sensitive organometallic transformations. By adhering to the optimized molar ratios of substrate, photocatalyst, base, and additive, manufacturers can achieve consistent results across different batches, which is critical for maintaining supply chain reliability. The following guide summarizes the critical operational steps derived from the patent examples, providing a clear roadmap for technical teams looking to adopt this methodology for the production of high-purity pharmaceutical intermediates.
- Under an inert gas atmosphere, sequentially add the beta-indole-carbonyl compound, a photocatalyst such as Ir(ppy)2(dtbbpy)PF6, a base like DABCO, an additive such as water, and a solvent like THF into a reactor.
- Stir the reaction mixture under the irradiation of a 456nm LED light source (10w) at a temperature between 0°C and 60°C for a duration of 6 to 48 hours until completion.
- Concentrate the filtrate using a rotary evaporator to obtain a crude product, and perform column chromatography separation using a mixed solvent of petroleum ether and ethyl acetate to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this visible light-mediated technology presents a compelling value proposition that extends far beyond mere technical novelty. The shift from stoichiometric reagents to a catalytic system fundamentally reshapes the cost structure of the manufacturing process, offering opportunities for significant margin improvement in a highly competitive market. By eliminating the need for expensive, hazardous, or difficult-to-source stoichiometric additives, the raw material costs are substantially reduced, while the simplified workup procedure decreases the consumption of solvents and silica gel during purification. This efficiency gain is further amplified by the mild reaction conditions, which lower the energy footprint of the production facility and reduce the wear and tear on reactor vessels, leading to lower capital expenditure requirements for plant maintenance and upgrades over the long term.
- Cost Reduction in Manufacturing: The transition to a photocatalytic regime eliminates the massive waste streams associated with traditional nucleophilic additions, where equivalent amounts of reagents end up as salt waste. By utilizing catalytic amounts of iridium complexes and inexpensive organic bases like DABCO, the direct material cost per kilogram of product is drastically lowered. Furthermore, the high atom economy of this transformation means that a greater proportion of the starting material mass is incorporated into the final product, maximizing the utility of every gram of raw material purchased and minimizing the cost of waste disposal which is a major hidden expense in fine chemical manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes significantly to supply chain stability, as it relies on readily available starting materials such as beta-indole-carbonyl compounds which can be sourced from multiple global suppliers. The mild reaction conditions reduce the risk of batch failures due to thermal runaways or equipment malfunctions, ensuring a consistent and predictable output of high-purity intermediates. This reliability is crucial for pharmaceutical clients who require uninterrupted supply to meet their own production schedules, and the scalability of the LED-based reactor setup allows for seamless transition from gram-scale R&D to multi-ton commercial production without the need for process re-engineering.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent, the ability to demonstrate a green manufacturing process is a key competitive advantage. This method operates at near-ambient temperatures and uses water as a benign additive, significantly reducing the volatile organic compound (VOC) emissions associated with high-temperature reflux processes. The simplified post-treatment, involving standard concentration and chromatography, avoids the generation of complex aqueous waste streams that are difficult to treat, thereby facilitating compliance with local environmental protection laws and reducing the liability associated with hazardous waste management in the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light-mediated synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring that stakeholders have access to accurate and verified information. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing lines and for assessing its potential impact on product quality and cost structures.
Q: What are the optimal reaction conditions for this visible light mediated synthesis?
A: The optimal conditions involve using Ir(ppy)2(dtbbpy)PF6 as the photocatalyst and DABCO as the base in a THF and water solvent system. The reaction proceeds efficiently at room temperature under 456nm LED irradiation for approximately 12 to 36 hours, yielding products with high Z/E selectivity.
Q: What is the substrate scope for this indole allyl alcohol synthesis method?
A: The method demonstrates wide applicability, accommodating various substituents on the indole ring including halogens, alkyl groups, and esters. It also tolerates different aryl and heteroaryl groups at the R1 position, such as phenyl and thienyl groups, making it versatile for diverse pharmaceutical intermediate synthesis.
Q: How does this photocatalytic method compare to traditional nucleophilic addition strategies?
A: Unlike traditional methods that often require equivalent amounts of additives and suffer from poor atom economy, this visible light mediated approach utilizes catalytic amounts of reagents. It offers superior atom economy, milder reaction conditions, and simplified post-treatment processes, significantly reducing chemical waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Allyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light photocatalysis in the synthesis of complex pharmaceutical intermediates like indole allyl alcohols. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in laboratory patents can be successfully translated into robust industrial processes. We are committed to delivering high-purity intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our infrastructure is designed to handle sensitive photochemical reactions safely and efficiently, providing our partners with a secure and reliable source for their critical raw materials.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced synthetic technology for their next-generation drug candidates. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how this green chemistry approach can optimize your specific supply chain. We encourage you to contact our technical procurement team today to request specific COA data for our indole derivatives and to discuss route feasibility assessments tailored to your project's unique requirements, ensuring a seamless path from discovery to commercial success.
