Scalable Pd-Catalyzed Synthesis of 4-Phenyl-6H-1,3-Oxazin-6-One Derivatives for Commercial Applications
The pharmaceutical and optoelectronic industries are constantly seeking robust synthetic pathways for complex heterocyclic scaffolds that offer both biological activity and advanced material properties. Patent CN111892550B introduces a significant breakthrough in the preparation of 4-phenyl-6H-1,3-oxazin-6-one derivatives, a class of compounds known for their presence in bioactive natural products like Discoipyrroles and their emerging potential as Aggregation-Induced Emission (AIE) materials. This technology addresses critical bottlenecks in traditional synthesis by utilizing a palladium-catalyzed ring-expansion strategy that operates under remarkably mild conditions. For R&D directors and procurement specialists, this patent represents a shift towards safer, more efficient manufacturing protocols that eliminate hazardous reagents while maintaining high purity standards exceeding 95%. The ability to access these novel skeletons through a one-pot procedure not only streamlines the workflow but also opens new avenues for developing next-generation optoelectronic components and therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,3-oxazin-6-one core has relied heavily on methodologies that pose significant safety and environmental challenges. Traditional routes often necessitate the use of phosphorus oxychloride (POCl3), a highly toxic and corrosive reagent that generates substantial hazardous waste, complicating disposal and increasing operational costs for any reliable functional heterocyclic intermediates supplier. Furthermore, alternative strategies have depended on diazo compounds, which are inherently unstable and explosive, requiring specialized equipment and rigorous safety protocols that slow down production throughput. Beyond safety concerns, these conventional methods frequently suffer from limited substrate scope, often failing to accommodate diverse aromatic or heteroaromatic substituents, and typically deliver only moderate yields that are insufficient for commercial scale-up of complex heterocycles. The cumbersome post-processing steps associated with removing phosphorus byproducts further degrade the overall atom economy, making these legacy processes economically unviable for large-scale manufacturing.
The Novel Approach
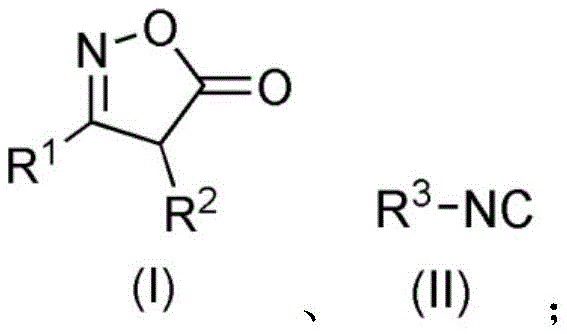
In stark contrast, the methodology disclosed in CN111892550B utilizes a transition-metal catalyzed insertion of isonitriles into isoxazolone substrates, fundamentally changing the risk profile and efficiency of the synthesis. This innovative route employs readily available starting materials, specifically isoxazolones and isonitriles, which react in the presence of a palladium catalyst such as PdCl2 and a benign additive like sodium benzoate. The reaction proceeds smoothly at temperatures ranging from 40-85°C, with optimal results observed at 85°C, eliminating the need for cryogenic conditions or extreme thermal stress. By avoiding toxic chlorinating agents and explosive precursors, this approach significantly simplifies the workup procedure, often requiring only standard extraction and column chromatography to achieve high-purity oxazinone derivatives. The broad tolerance for various R groups, including substituted phenyls, thienyls, and pyridyls, ensures that this method is versatile enough to support the rapid development of diverse libraries for both drug discovery and material science applications.
Mechanistic Insights into Palladium-Catalyzed Isonitrile Insertion
Understanding the catalytic cycle is crucial for optimizing this process for industrial applications. The reaction initiates with the formation of an active palladium species, likely Pd(PhCOO)2 generated in situ from the palladium salt and the benzoate additive. This active catalyst coordinates with the enamine tautomer of the isoxazolone substrate to form a key organometallic intermediate. Subsequent coordination of the isonitrile followed by migratory insertion creates a new carbon-nitrogen bond, expanding the ring system. The cycle continues through protonolysis and a subsequent ring-opening and ring-closing sequence that ultimately releases the final 4-phenyl-6H-1,3-oxazin-6-one product while regenerating the palladium catalyst for the next turnover. This mechanistic pathway is highly efficient, minimizing side reactions and ensuring that the catalyst loading can be kept relatively low, typically between 5-15 mol%, which is vital for cost reduction in optoelectronic material manufacturing where precious metal residues must be strictly controlled.
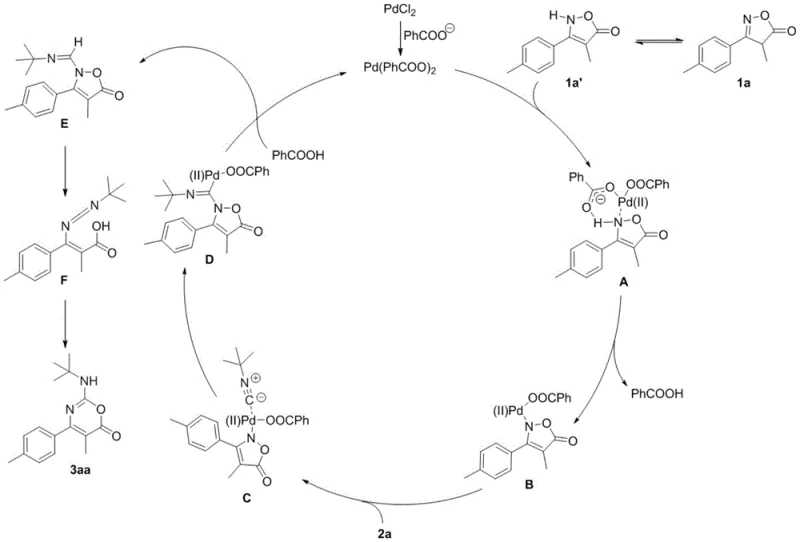
From an impurity control perspective, the mildness of this catalytic system is a distinct advantage. Harsh acidic or basic conditions often lead to the hydrolysis of sensitive functional groups or the formation of polymeric byproducts, but the neutral to slightly basic environment provided by the carboxylate additive preserves the integrity of the substrate. The specific choice of solvent, such as 1,1,2-trichloroethane, further stabilizes the intermediates and facilitates the necessary conformational changes for ring expansion. By carefully tuning the ratio of isonitrile to isoxazolone, preferably at 1:2, the reaction drives towards completion, minimizing the presence of unreacted starting materials in the crude mixture. This high level of selectivity translates directly to simpler purification processes and higher overall yields, with specific examples in the patent demonstrating isolated yields as high as 98% for certain substrates, thereby ensuring a consistent supply of high-quality material for downstream applications.
How to Synthesize 4-Phenyl-6H-1,3-Oxazin-6-One Efficiently
To implement this synthesis effectively, operators should adhere to the optimized parameters defined in the patent examples to ensure reproducibility and safety. The process involves dissolving the isoxazolone and palladium catalyst in the chosen solvent, followed by the addition of the sodium benzoate additive and the isonitrile component. The mixture is then heated to the target temperature of 85°C for approximately 6 hours, although reaction times can vary between 2-12 hours depending on the specific electronic nature of the substituents. Detailed standardized synthesis steps see the guide below.
- Combine isoxazolone compound (Formula I) and isonitrile compound (Formula II) in an organic solvent such as 1,1,2-trichloroethane.
- Add palladium catalyst (e.g., PdCl2, 5-15 mol%) and alkali metal salt additive (e.g., sodium benzoate, 10-150 mol%).
- Heat the mixture to 40-85°C for 2-12 hours, then purify via extraction and column chromatography to obtain the target oxazinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple chemical transformation. The elimination of hazardous reagents like POCl3 removes the need for expensive corrosion-resistant reactors and specialized waste treatment facilities, leading to substantial cost savings in capital expenditure and operational overhead. Furthermore, the use of stable, commercially available isonitriles and isoxazolones ensures a robust supply chain that is less susceptible to the regulatory restrictions often placed on controlled precursors. This stability allows for better inventory planning and reduces the risk of production stoppages due to raw material shortages, enhancing supply chain reliability for long-term projects.
- Cost Reduction in Manufacturing: The removal of toxic phosphorus reagents drastically simplifies the effluent treatment process, reducing the environmental compliance burden and associated disposal fees. Additionally, the high atom economy and excellent yields mean that less raw material is wasted per kilogram of product, directly lowering the cost of goods sold. The ability to use lower catalyst loadings without sacrificing performance further contributes to economic efficiency, making this route highly competitive for large-volume production.
- Enhanced Supply Chain Reliability: Since the starting materials are stable solids or liquids that do not require special storage conditions like refrigeration or inert atmospheres, logistics are simplified. This ease of handling reduces lead time for high-purity AIE materials, allowing manufacturers to respond more quickly to market demands. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in utility supply, ensuring consistent output quality.
- Scalability and Environmental Compliance: The mild thermal requirements (40-85°C) make this process easily scalable from gram to ton quantities without the need for complex engineering controls. The absence of explosive diazo compounds removes a major safety barrier to scale-up, facilitating faster technology transfer from the lab to the plant. Moreover, the greener profile of the synthesis aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing process against tighter emission standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for potential partners.
Q: What are the advantages of this Pd-catalyzed method over traditional POCl3 routes?
A: This method eliminates the use of toxic phosphorus oxychloride (POCl3) and explosive diazo compounds, offering milder conditions (40-85°C), higher safety, and easier post-processing.
Q: What is the substrate scope for the isonitrile component?
A: The process tolerates a wide range of isonitriles including tert-butyl, adamantyl, cyclohexyl, and functionalized aryl isonitriles, allowing for diverse derivative synthesis.
Q: Do these derivatives exhibit specific optical properties?
A: Yes, the synthesized 4-phenyl-6H-1,3-oxazin-6-one derivatives demonstrate Aggregation-Induced Emission (AIE) characteristics, making them valuable for optoelectronic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Phenyl-6H-1,3-Oxazin-6-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Pd-catalyzed synthesis for producing advanced functional materials and pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-phenyl-6H-1,3-oxazin-6-one derivative meets the exacting standards required for optoelectronic and medicinal applications. Our commitment to quality and safety makes us the ideal partner for organizations looking to leverage this innovative chemistry.
We invite you to discuss how we can optimize your supply chain with these high-value intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to help you evaluate the integration of this technology into your portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
