Scalable Production of High-Purity 1,3-Indandione Compounds via Novel Condensation Route
Scalable Production of High-Purity 1,3-Indandione Compounds via Novel Condensation Route
The chemical industry continuously seeks robust methodologies for synthesizing critical intermediates that balance efficiency, safety, and cost-effectiveness. Patent CN103121887A introduces a transformative preparation method for 1,3-indandione compounds, addressing significant bottlenecks found in legacy manufacturing processes. These compounds serve as pivotal building blocks in the pharmaceutical sector, particularly for anticoagulant medications, as well as in the production of functional materials and azo dispersion dyes. The disclosed technology leverages a streamlined two-step sequence involving the condensation of phthalate derivatives with malonate esters, followed by a hydrolysis and decarboxylation phase. This approach not only simplifies the operational workflow but also drastically reduces the environmental footprint associated with traditional synthesis, positioning it as a superior choice for modern chemical supply chains seeking reliable high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3-indandione has been plagued by inefficient reaction pathways and hazardous solvent requirements that hinder large-scale adoption. One prevalent method involves the condensation of phthalic esters with ethyl acetate, a process notorious for generating numerous side reactions and complex impurity profiles. As illustrated in prior art, this route typically suffers from abysmal productivity, with reported yields hovering around merely 35 percent, necessitating extensive purification steps. Furthermore, the recrystallization of the crude product often relies on benzene, a known carcinogen with severe regulatory restrictions, making it untenable for modern green chemistry standards. Another existing route employs microwave catalysis with tetrahydrophthalic anhydride; however, this technique is inherently limited to small-scale laboratory preparations due to equipment constraints, failing to meet the demands of industrial volume production.
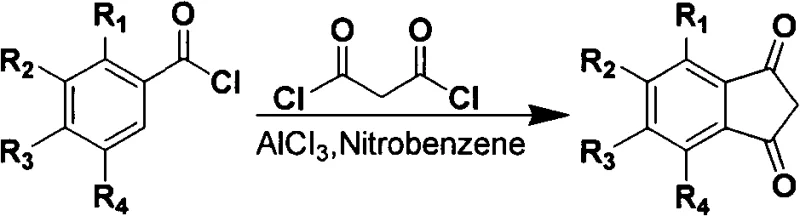
Additionally, alternative Friedel-Crafts acylation strategies utilizing aluminum trichloride catalysts introduce further complications regarding waste management and operator safety. These reactions frequently require nitrobenzene as a solvent, which is highly toxic and poses significant disposal challenges. The generation of substantial quantities of acidic gases during both the reaction and post-processing stages exacerbates corrosion issues in standard reactor vessels and demands sophisticated scrubbing systems. Consequently, these conventional pathways create substantial barriers to entry for manufacturers aiming to produce cost reduction in pharmaceutical intermediates manufacturing while adhering to strict environmental compliance protocols.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology outlined in CN103121887A offers a refined and economically viable alternative centered on the direct condensation of phthalate compounds with malonate esters. This innovative route eliminates the need for hazardous solvents like benzene or nitrobenzene, instead utilizing a versatile range of common organic solvents such as DMF, ethanol, or toluene. The reaction proceeds through a stable substituted malonate intermediate, which can be isolated and purified with relative ease before undergoing the final cyclization step. By avoiding the harsh conditions and toxic reagents associated with previous methods, this new approach ensures a cleaner reaction profile and significantly higher overall yields, as demonstrated by experimental data achieving over 80 percent efficiency in specific embodiments.
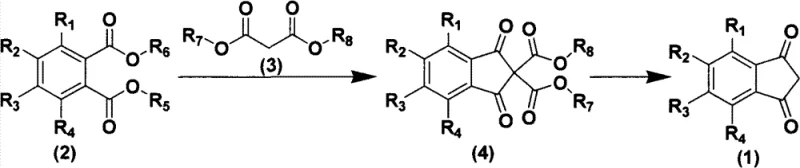
The versatility of this novel approach extends to its compatibility with a wide array of substituents on the aromatic ring, allowing for the synthesis of diverse derivatives without compromising reaction efficiency. Whether incorporating electron-withdrawing groups like halogens and nitro groups or electron-donating moieties such as methoxy and alkyl chains, the process maintains robust performance. This flexibility is crucial for R&D teams developing next-generation active pharmaceutical ingredients or specialized dye molecules. Furthermore, the operational simplicity of the method, which relies on standard heating and stirring rather than specialized microwave apparatus, facilitates seamless translation from pilot plant studies to full-scale commercial production, ensuring a consistent supply of high-purity 1,3-indandione compounds for global markets.
Mechanistic Insights into Base-Catalyzed Condensation and Cyclization
The core of this synthetic breakthrough lies in the precise orchestration of a base-catalyzed Claisen-type condensation followed by an acid-mediated hydrolysis and decarboxylation sequence. In the initial step, a strong base such as sodium ethoxide or potassium carbonate deprotonates the active methylene group of the malonate ester, generating a nucleophilic enolate species. This enolate then attacks the carbonyl carbon of the phthalate ester, initiating a cascade of elimination and cyclization events that construct the indane backbone. The choice of base and solvent is critical here; the patent specifies a broad spectrum of acceptable catalysts ranging from alkali metal hydroxides to alkoxides, allowing process chemists to optimize kinetics based on specific substrate electronic properties. This mechanistic pathway avoids the formation of stable chelates often seen with Lewis acid catalysts, thereby simplifying the downstream workup and minimizing metal contamination in the final product.
Following the formation of the substituted malonate intermediate, the second stage involves a rigorous hydrolysis and decarboxylation protocol to reveal the final 1,3-diketone functionality. Under acidic conditions, typically utilizing sulfuric acid or hydrochloric acid at elevated temperatures, the ester groups are hydrolyzed to carboxylic acids, which subsequently undergo thermal decarboxylation. This step is thermodynamically driven by the release of carbon dioxide gas, pushing the equilibrium towards the desired cyclic ketone product. The mechanism ensures that potential impurities arising from incomplete condensation are effectively removed or converted, resulting in a product with exceptional purity profiles. Understanding these mechanistic nuances allows manufacturers to fine-tune reaction parameters such as temperature ramps and acid concentrations to maximize throughput while maintaining stringent quality control standards required for pharmaceutical applications.
How to Synthesize 1,3-Indandione Efficiently
Implementing this synthesis strategy requires careful attention to reaction stoichiometry and thermal management to ensure optimal conversion rates and product quality. The process begins with the preparation of the reaction mixture containing the phthalate starting material and the malonate coupling partner in a suitable polar aprotic or protic solvent. Temperature control is paramount during the addition of reagents to prevent exothermic runaway, followed by a sustained heating period to drive the condensation to completion. Once the intermediate is formed and isolated, the subsequent hydrolysis step demands precise acid handling and gas venting capabilities to manage the evolution of carbon dioxide safely. For detailed operational parameters and specific stoichiometric ratios tailored to your production needs, please refer to the standardized synthesis guide below.
- Condense phthalate compounds with malonate compounds under basic catalysis at elevated temperatures to form substituted malonate intermediates.
- Isolate the intermediate crude product via filtration and washing, optionally purifying before the next step.
- Subject the intermediate to hydrolysis and decarboxylation using acid catalysis to yield the final 1,3-indandione product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this novel synthesis route offers compelling advantages that directly impact the bottom line and supply chain resilience. By shifting away from restricted solvents like benzene and nitrobenzene, manufacturers can significantly reduce the costs associated with hazardous waste disposal and regulatory compliance reporting. The elimination of these toxic materials also streamlines the logistics of raw material sourcing, as the required phthalates and malonates are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This diversification of the supply base enhances overall supply chain reliability, ensuring that production schedules remain uninterrupted even during market fluctuations for specific reagents.
- Cost Reduction in Manufacturing: The simplified workflow and higher yields inherent to this method translate into substantial cost savings per kilogram of finished product. By avoiding the low-yield pitfalls of traditional ethyl acetate condensation, which often wastes over half of the starting material, this process maximizes atom economy and reduces raw material consumption. Furthermore, the ability to use crude intermediates directly in the second step without extensive purification minimizes solvent usage and labor hours, driving down the overall cost of goods sold. These efficiencies allow suppliers to offer more competitive pricing structures while maintaining healthy margins, a critical factor for buyers managing tight budgets in the pharmaceutical and agrochemical sectors.
- Enhanced Supply Chain Reliability: The reliance on widely available, non-specialized raw materials ensures a stable and continuous supply flow for long-term contracts. Unlike methods requiring custom-synthesized precursors or specialized microwave equipment, this route can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure reduces the capital expenditure required for technology transfer and accelerates the time-to-market for new products. Consequently, partners can rely on consistent delivery timelines and robust inventory levels, safeguarding their own production lines against upstream disruptions.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, having been successfully demonstrated in multi-liter reactor setups without loss of efficiency. The absence of toxic solvents and the generation of manageable byproducts like carbon dioxide and alcohol simplify the environmental permitting process and reduce the burden on effluent treatment facilities. This alignment with green chemistry principles not only future-proofs the manufacturing operation against tightening environmental regulations but also enhances the corporate sustainability profile of the end-user brands. Such compliance is increasingly becoming a prerequisite for inclusion in the supply chains of major multinational corporations committed to ESG goals.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the process capabilities and limitations. These responses are derived directly from the experimental data and technical specifications detailed in the patent literature, providing a transparent view of what can be achieved. Understanding these technical facets is essential for integrating this synthesis route into your existing manufacturing portfolio and ensuring that quality targets are met consistently.
Q: What are the primary advantages of this new synthesis method over traditional routes?
A: The novel method avoids toxic solvents like benzene and nitrobenzene, operates under milder conditions, and achieves significantly higher yields compared to the 35% yield of conventional ethyl acetate condensation methods.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes readily available raw materials and standard reactor equipment without the need for specialized microwave catalysts, making it highly suitable for commercial scale-up from kilograms to metric tons.
Q: What types of substituents are compatible with this synthetic route?
A: The method demonstrates broad substrate scope, accommodating various substituents on the phthalate ring including alkyl groups, halogens, nitro groups, and alkoxy groups, allowing for diverse derivative synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Indandione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medicines and advanced functional materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of 1,3-indandione meets the exacting standards required by the global pharmaceutical industry. Our commitment to technical excellence allows us to navigate complex synthetic challenges, delivering products that empower our clients' innovation pipelines.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality criteria. We encourage potential partners to reach out for specific COA data and route feasibility assessments to determine how this efficient manufacturing process can enhance your supply chain stability and reduce overall production costs. Let us be your trusted partner in bringing high-performance chemical solutions to the market.
