Revolutionizing 2-Arylmalonamide Production: A Strategic Guide for Commercial Scale-up
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of key intermediates, particularly within the agrochemical sector. A pivotal development in this domain is documented in patent CN108689874B, which discloses a groundbreaking method for preparing 2-arylmalonamide compounds. These compounds serve as critical precursors for advanced herbicides such as pinoxaden, a widely used post-emergence graminicide. The traditional synthetic pathways for these molecules have long been plagued by complexity, high costs associated with precious metal catalysts, and significant environmental burdens due to hazardous waste generation. This new technology represents a paradigm shift, introducing a direct, one-pot aromatization-hydrolysis strategy that converts readily available 2-(cyclohexenylene)malononitrile derivatives directly into the target arylmalonamides. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for optimizing supply chains and reducing the cost of goods sold (COGS) in competitive agrochemical markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-arylmalonamide compounds, especially those with significant steric hindrance like 2-(2,6-disubstituted aryl)malonamides, has relied on convoluted multi-step sequences. The conventional route typically begins with aromatic amines, which must undergo a dangerous diazotization followed by a Sandmeyer reaction to introduce a halogen atom. This step not only requires hazardous reagents but also generates substantial quantities of acidic wastewater and nitrogen oxide emissions, posing severe environmental compliance challenges. Subsequently, the resulting sterically hindered aryl halide must be coupled with malonic acid derivatives via transition metal-catalyzed C-C bond formation, often utilizing expensive palladium or nickel catalysts. These metal catalysts are not only costly to procure but are also difficult to recover and recycle, leading to potential heavy metal contamination in the final API or intermediate, which necessitates expensive purification steps to meet stringent regulatory limits.
The Novel Approach
In stark contrast, the methodology described in patent CN108689874B bypasses these bottlenecks entirely by employing a biomimetic-inspired oxidative aromatization strategy. Instead of building the aromatic ring through coupling, this approach starts with a partially saturated cyclohexenyl precursor that already contains the necessary carbon skeleton. Under the influence of a mild oxidant and aqueous acid, the cyclohexene ring undergoes dehydrogenation to form the stable aromatic system, while the nitrile groups are simultaneously hydrolyzed to amides. This telescoped process eliminates the need for isolating unstable aryl halide intermediates and completely removes the requirement for precious metal catalysts. The result is a streamlined workflow that drastically reduces the number of unit operations, minimizes solvent consumption, and simplifies the overall process safety profile by avoiding explosive diazonium salts.
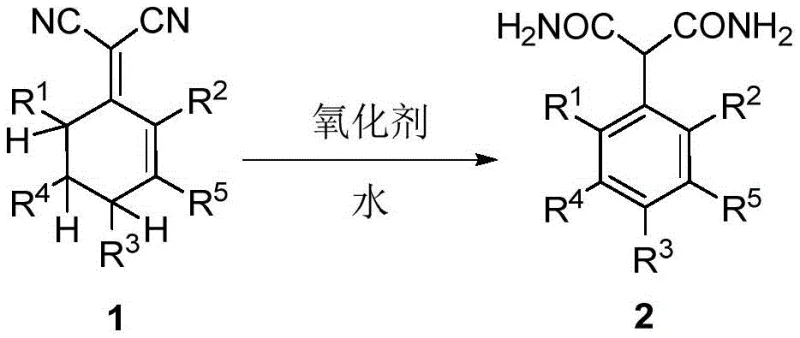
Mechanistic Insights into Oxidative Aromatization-Hydrolysis
The core chemical transformation relies on a synergistic interplay between oxidative dehydrogenation and acid-catalyzed hydration. The reaction initiates with the activation of the cyclohexenyl double bond by the oxidant species, such as the persulfate radical or peroxide oxygen. This triggers a cascade of electron transfers that ultimately remove hydrogen atoms from the ring system, restoring aromaticity—a thermodynamically favorable driving force that propels the reaction forward. Concurrently, the acidic medium, typically concentrated sulfuric acid, protonates the nitrile nitrogen atoms, rendering the cyano carbons highly electrophilic and susceptible to nucleophilic attack by water molecules. This dual-activation mechanism ensures that the aromatization and hydrolysis occur in a synchronized manner, preventing the accumulation of partially hydrolyzed byproducts or over-oxidized impurities that often plague stepwise syntheses.
From an impurity control perspective, this mechanism offers superior selectivity compared to metal-catalyzed couplings. In traditional Pd-catalyzed reactions, homocoupling of aryl halides and dehalogenation are common side reactions that generate difficult-to-remove impurities. In this oxidative protocol, the primary byproducts are inorganic salts derived from the oxidant and water, which are easily removed during the aqueous workup. The patent data indicates that reaction temperatures between 60-80°C provide the optimal kinetic balance, ensuring complete conversion of the starting material while minimizing thermal degradation of the sensitive amide functionality. This precise control over reaction parameters allows for the consistent production of high-purity intermediates, a critical factor for downstream formulation stability.
How to Synthesize 2-Arylmalonamide Efficiently
Implementing this synthesis route requires careful attention to the addition order and temperature profiling to manage the exothermic nature of the oxidation. The process generally involves suspending the 2-(cyclohexenylene)malononitrile substrate in water or an aqueous acid mixture, cooling the slurry to 0-5°C to suppress initial side reactions, and then slowly introducing the oxidant and acid catalyst. Maintaining this low temperature during the addition phase is crucial for safety and selectivity before the mixture is warmed to the reaction temperature of 60-80°C to drive the conversion to completion. For detailed operational parameters and specific stoichiometric ratios validated by experimental data, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by cooling 2-(cyclohexenylene)malononitrile, an oxidant such as potassium persulfate or hydrogen peroxide, and water to a temperature range of 0-5°C.
- Slowly add concentrated sulfuric acid to the cooled mixture to initiate the catalytic cycle while maintaining strict temperature control.
- Raise the reaction temperature to 60-80°C to drive the aromatization and hydrolysis to completion, followed by standard workup involving extraction and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere technical elegance. The most immediate impact is the drastic simplification of the raw material portfolio. By eliminating the need for specialized aryl amines, hazardous halogenating agents, and precious metal catalysts, companies can source commodities that are available in bulk from multiple suppliers, thereby reducing supply risk and volatility. Furthermore, the removal of heavy metal catalysts simplifies the regulatory approval process for the final product, as there is no need for extensive testing and validation of residual metal levels, which accelerates time-to-market for new agrochemical formulations.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling, primarily driven by the elimination of high-cost inputs. Traditional routes rely on palladium or nickel catalysts which represent a significant portion of the variable cost, alongside the expensive bromine or iodine reagents required for the Sandmeyer step. By replacing these with inexpensive inorganic oxidants like potassium persulfate or even air/oxygen, the direct material cost is significantly reduced. Additionally, the one-pot nature of the reaction reduces utility costs associated with heating, cooling, and stirring across multiple isolated steps, leading to substantial overall cost savings in agrochemical manufacturing without compromising yield.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reagents used. Unlike sensitive organometallic catalysts that require inert atmosphere storage and specialized handling, the oxidants and acids utilized in this process are stable, shelf-stable commodities. This reduces the logistical complexity and storage costs associated with hazardous material management. Moreover, the high yields reported in the patent examples (ranging from 80% to 89%) ensure that less raw material is wasted, improving the overall mass balance and reducing the volume of waste that needs to be transported and treated, further stabilizing the supply chain against environmental regulatory disruptions.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, particularly regarding heat transfer and mixing in metal-catalyzed reactions. This oxidative aromatization pathway is inherently safer and more scalable because it avoids the use of pyrophoric catalysts and explosive diazonium intermediates. The reaction generates benign inorganic byproducts, significantly lowering the E-factor (environmental factor) of the process. This aligns perfectly with modern green chemistry initiatives and helps manufacturers meet increasingly strict environmental discharge standards, avoiding potential fines and shutdowns that could disrupt supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term viability of the supply source.
Q: What are the primary advantages of this new synthesis route over traditional methods?
A: The primary advantage is the elimination of expensive transition metal catalysts and hazardous diazotization steps. Traditional routes require palladium-catalyzed coupling of aryl halides, which generates heavy metal waste and incurs high raw material costs. This new method utilizes simple oxidants and acid catalysis, significantly reducing both environmental impact and production costs.
Q: Can this process be scaled for industrial production of herbicide intermediates?
A: Yes, the process is highly scalable. The patent examples demonstrate successful reactions on molar scales up to 1.00 mol with yields reaching 80-89%. The use of common reagents like sulfuric acid and potassium persulfate, rather than sensitive organometallic catalysts, makes the process robust and suitable for large-scale manufacturing facilities.
Q: What specific oxidants are compatible with this aromatization-hydrolysis reaction?
A: The patent specifies a variety of effective oxidants including inorganic peroxides like potassium persulfate, hydrogen peroxide, and even molecular oxygen or air. This flexibility allows manufacturers to select the most cost-effective and logistically feasible oxidant based on their specific facility capabilities and safety protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Arylmalonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN108689874B can be reliably replicated on an industrial scale. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-arylmalonamide delivered meets the exacting standards required for high-performance agrochemical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this metal-free route can improve your margins. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this critical intermediate.
