Advanced Synthetic Route for 11-Aminoundecanoic Acid: Enabling Scalable Nylon 11 Production
The global demand for high-performance engineering plastics continues to drive innovation in monomer synthesis, particularly for Nylon 11, a material prized for its exceptional impact resistance and chemical stability. A pivotal advancement in this sector is detailed in patent CN109593045B, which discloses a novel preparation method for 11-aminoundecanoic acid, the critical monomer precursor. Unlike legacy processes that depend on the volatile supply chain of castor oil derivatives, this technology leverages a robust oxidative condensation strategy starting from 9-hydroxynonanoic acid esters. This approach represents a paradigm shift towards greener chemistry, eliminating the need for hazardous bromination reagents and energy-intensive thermal cracking units. For R&D directors and procurement strategists, understanding this pathway is essential for securing a reliable nylon 11 monomer supplier capable of delivering consistent quality without the geopolitical and agricultural risks associated with natural oil feedstocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 11-aminoundecanoic acid has been bottlenecked by the reliance on castor oil as the primary raw material. The conventional process involves the methanolysis of castor oil followed by high-temperature vacuum cracking to isolate undecylenic acid. This pyrolysis step is inherently inefficient, characterized by high energy consumption, incomplete conversion, and the formation of stubborn coke deposits that foul reactor vessels. Furthermore, the subsequent conversion of undecylenic acid to the amino acid typically requires an anti-Markovnikov addition of hydrobromic acid to generate 11-bromoundecanoic acid. This bromination stage introduces severe operational hazards, including the handling of corrosive gases and the generation of toxic bromide waste streams that require costly abatement systems. The final ammonolysis step often necessitates a large excess of ammonia to drive the reaction, leading to complex recovery loops and inflated utility costs, making cost reduction in polyamide manufacturing difficult to achieve with these legacy technologies.
The Novel Approach
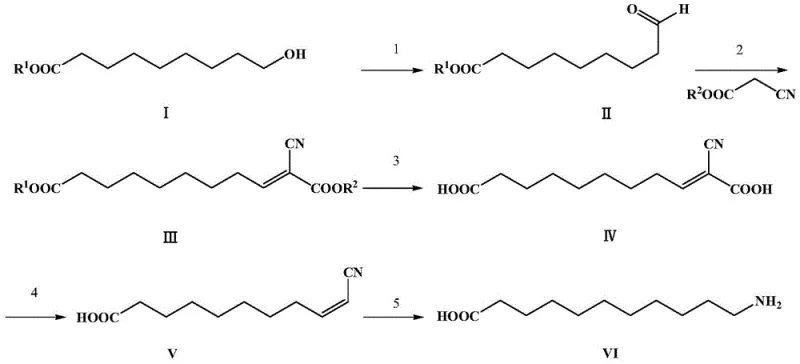
The methodology outlined in the patent data circumvents these structural inefficiencies by employing a linear synthetic sequence rooted in selective oxidation and carbon-carbon bond formation. By starting with 9-hydroxynonanoate, the process utilizes mild oxidants like manganese dioxide or copper oxide to generate the corresponding aldehyde under gentle conditions, typically between 25°C and 50°C. This aldehyde then undergoes a Knoevenagel condensation with cyanoacetate, effectively extending the carbon chain while introducing the necessary nitrogen functionality via the nitrile group. This strategic design avoids the use of elemental halogens entirely. The subsequent steps involve a streamlined hydrolysis and a selective decarboxylation to position the functional groups correctly, followed by a single-pot hydrogenation. This route not only simplifies the engineering requirements by removing high-temperature cracking furnaces but also enhances the overall atom economy, positioning it as a superior choice for the commercial scale-up of complex amino acids.
Mechanistic Insights into Oxidative Condensation and Decarboxylation
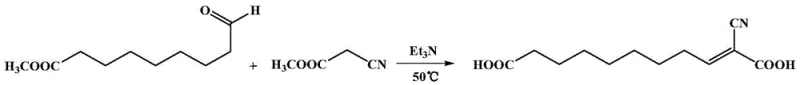
The core chemical innovation lies in the precise control of oxidation states and the regioselectivity of the condensation reaction. In the initial step, the use of heterogeneous oxidants such as MnO2 allows for the selective transformation of the primary alcohol to an aldehyde without over-oxidizing to the carboxylic acid, preserving the ester moiety at the other end of the chain. This selectivity is crucial for maintaining the integrity of the 9-carbon backbone. Following this, the Knoevenagel condensation acts as the chain-extending engine. Under basic catalysis (using amines like triethylamine or inorganic bases like KOH), the active methylene group of methyl cyanoacetate attacks the carbonyl carbon of the 9-oxononanoate. This forms an alpha,beta-unsaturated nitrile intermediate. The presence of the electron-withdrawing cyano and ester groups stabilizes the transition state, allowing the reaction to proceed efficiently at moderate temperatures, thereby minimizing side reactions such as polymerization or self-condensation of the aldehyde.
Following the condensation, the mechanism proceeds through a hydrolysis and decarboxylation sequence that is critical for establishing the final 11-carbon chain length with the terminal amine. The hydrolysis step converts the ester groups into free carboxylic acids, setting the stage for thermal decarboxylation. The patent highlights that this decarboxylation can be achieved thermally or with acid catalysis in solvents like dioxane at approximately 100°C. This step selectively removes one carboxyl group adjacent to the double bond, shifting the unsaturation and preparing the molecule for the final reduction. The resulting 10-cyano-10-alkene decanoic acid is then subjected to catalytic hydrogenation. Here, the mechanism involves the simultaneous reduction of the carbon-carbon double bond and the nitrile group to a primary amine. The use of Raney Nickel or Pd/C facilitates the adsorption of hydrogen onto the metal surface, where it is transferred to the substrate. This dual-reduction capability is vital for achieving high-purity 11-aminoundecanoic acid in a single finishing step, avoiding the need for separate protection and deprotection strategies often seen in older synthetic routes.
How to Synthesize 11-Aminoundecanoic Acid Efficiently
Implementing this synthesis requires careful attention to solvent selection and catalyst loading to maximize yield and minimize impurities. The process begins with the dissolution of the hydroxy-ester feedstock in a polar aprotic or ester solvent, followed by the controlled addition of the oxidant. Once the aldehyde is confirmed, the cyanoacetate and base catalyst are introduced for the condensation phase. The reaction mixture is then hydrolyzed using aqueous base, acidified, and subjected to thermal decarboxylation. The final hydrogenation is conducted in a pressure vessel, requiring strict control of hydrogen partial pressure and temperature to ensure complete conversion of the nitrile without hydrogenolysis of the carboxylic acid group. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- Selective oxidation of 9-hydroxynonanoate using manganese dioxide or copper oxide to form 9-oxononanoate.
- Knoevenagel condensation of the oxo-intermediate with cyanoacetate followed by hydrolysis to yield a dicarboxylic acid derivative.
- Thermal decarboxylation of the dicarboxylic acid intermediate to generate the unsaturated nitrile compound.
- Catalytic hydrogenation using Raney Nickel or Pd/C under pressure to reduce the nitrile and double bond simultaneously.
- Final purification via crystallization or washing to obtain white powder 11-aminoundecanoic acid with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this oxidative condensation route offers tangible strategic benefits beyond mere technical feasibility. The most significant advantage is the decoupling from the castor oil market, which is subject to agricultural fluctuations and geographic concentration. By utilizing synthetic fatty acid derivatives as feedstocks, manufacturers can secure a more stable and predictable supply chain, reducing lead time for high-purity polymer intermediates and mitigating the risk of raw material shortages. Furthermore, the elimination of hydrobromic acid removes the need for specialized Hastelloy-lined reactors and complex scrubbing systems required to handle corrosive bromine vapors. This simplification of the plant infrastructure translates directly into lower capital expenditure (CAPEX) and reduced maintenance costs over the lifecycle of the production facility.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by operating key reaction steps at mild temperatures (25-50°C), which drastically lowers energy consumption compared to the high-heat pyrolysis required for castor oil cracking. Additionally, the avoidance of excessive ammonia usage in the final step reduces the load on recovery distillation columns, leading to substantial utility savings. The use of common base catalysts like triethylamine or potassium hydroxide, which are inexpensive and readily available, further drives down the variable cost of goods sold, making the final monomer more price-competitive in the global engineering plastics market.
- Enhanced Supply Chain Reliability: By removing the dependency on natural oil extraction and the complex fractionation of cracked products, this synthetic route offers a more robust supply continuity. The reagents involved, such as cyanoacetate and manganese dioxide, are commodity chemicals with mature global supply networks, ensuring that production schedules are not disrupted by agricultural harvest cycles or weather events. This reliability is critical for long-term contracts with automotive and aerospace clients who require guaranteed delivery of Nylon 11 components for safety-critical applications.
- Scalability and Environmental Compliance: The process is inherently greener, generating significantly less hazardous waste than the traditional bromination route. The absence of bromide salts in the effluent simplifies wastewater treatment and reduces the environmental compliance burden. Moreover, the reaction steps are amenable to continuous flow processing or large-batch reactor operations, facilitating easy scale-up from pilot plants to multi-ton commercial production without encountering the heat transfer limitations often associated with exothermic bromination reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aimed at clarifying the operational parameters and quality outcomes for potential licensees and manufacturing partners.
Q: Why is this oxidative condensation route superior to traditional castor oil cracking?
A: Traditional methods rely on high-energy pyrolysis of castor oil which suffers from incomplete cracking, coking, and difficult separation of isomers. This new route operates at mild temperatures (25-50°C) for key steps, avoiding high energy consumption and significantly simplifying downstream purification.
Q: How does this process address environmental concerns regarding bromination?
A: Conventional synthesis often utilizes hydrobromic acid for bromination, which generates corrosive waste and requires specialized equipment. This patent eliminates the bromination step entirely, replacing it with a Knoevenagel condensation and decarboxylation sequence that uses standard organic solvents and base catalysts, drastically reducing hazardous waste generation.
Q: What catalysts are suitable for the final hydrogenation step?
A: The patent demonstrates flexibility in the final reduction step, successfully utilizing both Raney Nickel and Palladium on Carbon (Pd/C) catalysts. This allows manufacturers to optimize based on availability and cost, with reaction pressures ranging from 2 to 6 MPa and temperatures around 100°C to ensure complete reduction of both the nitrile and alkene functionalities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 11-Aminoundecanoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the performance of downstream polyamides. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the absence of halogenated impurities and isomeric byproducts, guaranteeing that every batch of 11-aminoundecanoic acid meets the exacting standards required for high-end Nylon 11 applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this greener route can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable future for your polymer manufacturing operations.
