Advanced Metal-Free Synthesis of 2,3,6-Trisubstituted Pyrazine N-Oxides for Commercial Scale-Up
Introduction to Next-Generation Pyrazine Synthesis
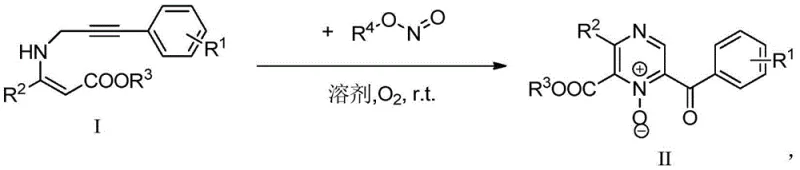
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for nitrogen-containing heterocycles, particularly pyrazine derivatives, which serve as critical scaffolds in bioactive molecules. A significant breakthrough in this domain is documented in Chinese Patent CN110372613B, which discloses a highly efficient preparation method for 2,3,6-trisubstituted pyrazine N-oxide compounds. Unlike traditional methodologies that often rely on harsh acidic or basic environments and transition metal catalysts, this novel approach utilizes a mild, metal-free oxidative cyclization strategy. By employing specific nitrite esters as oxidants under an oxygen atmosphere at room temperature, the process achieves rapid conversion of propargylamine substrates into complex pyrazine cores. This technological advancement represents a paradigm shift towards greener chemistry, offering substantial benefits for manufacturers aiming to reduce environmental impact while maintaining high throughput and product quality in the production of fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted pyrazine derivatives has been fraught with significant operational challenges that hinder large-scale manufacturing efficiency. Conventional routes typically necessitate the use of strong acids or strong bases to drive condensation and cyclization reactions, creating severe corrosion issues for reactor equipment and generating substantial amounts of hazardous waste streams that require costly treatment. Furthermore, many established protocols depend on transition metal catalysts, such as copper or palladium complexes, which not only inflate raw material costs but also introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Removing these trace metals to meet stringent regulatory limits often requires additional purification steps, such as scavenging or recrystallization, which drastically reduce overall yield and extend production lead times. Additionally, traditional oxidative methods frequently employ stoichiometric amounts of aggressive oxidants that pose safety risks and lack atom economy, making them less desirable for modern sustainable manufacturing frameworks.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110372613B offers a streamlined and environmentally benign alternative that directly addresses these industrial pain points. This innovative route leverages tert-butyl nitrite (TBN) as a dual-function reagent acting as both the nitrogen source and the oxidant, facilitating the cyclization under remarkably mild conditions. The reaction proceeds efficiently at room temperature (approximately 25°C) within a mere 30 minutes, eliminating the need for energy-intensive heating or cooling cycles. Crucially, the process is entirely metal-free and operates without the addition of exogenous acids or bases, thereby simplifying the workup procedure to a simple aqueous quench and extraction. This simplicity not only enhances the safety profile of the operation but also significantly improves the purity of the crude product, reducing the burden on downstream purification units. The versatility of this method is further demonstrated by its tolerance to various functional groups on the aromatic rings, allowing for the synthesis of a diverse library of pyrazine N-oxides suitable for drug discovery and development.
Mechanistic Insights into Metal-Free Oxidative Cyclization
The mechanistic pathway of this transformation is a fascinating example of radical-mediated or ionic oxidative cyclization driven by nitrite esters. The reaction initiates with the interaction between the alkyne moiety of the substrate and the nitrite species, likely generating a reactive nitrogen-centered radical or a nitroso intermediate under the oxygen atmosphere. This activated species then undergoes an intramolecular attack on the adjacent enamine double bond, triggering a cascade of cyclization events that construct the six-membered pyrazine ring. The presence of molecular oxygen plays a pivotal role in re-oxidizing intermediate species and driving the reaction equilibrium towards the formation of the stable N-oxide functionality. This mechanism avoids the high-energy barriers associated with thermal cyclization, explaining why the reaction can proceed rapidly at ambient temperatures. The regioselectivity observed in the formation of the 2,3,6-trisubstituted pattern is governed by the electronic properties of the starting acrylate and the steric environment of the propargyl group, ensuring consistent structural outcomes across different substrate variations.
From an impurity control perspective, the absence of metal catalysts inherently eliminates a major class of potential contaminants, specifically heavy metal residues that are notoriously difficult to purge from organic matrices. Furthermore, the mild reaction conditions minimize the formation of thermal degradation byproducts or polymerization side reactions that often plague high-temperature syntheses. The use of tert-butyl nitrite ensures that the nitrogen atoms are incorporated cleanly into the heterocyclic framework without generating complex mixtures of over-oxidized or under-oxidized species. The resulting pyrazine N-oxide products exhibit high chemical stability, and the reaction profile suggests a clean conversion where the starting material is consumed rapidly, as evidenced by TLC monitoring in the patent examples. This clean reaction profile is critical for pharmaceutical manufacturers who must adhere to strict impurity specifications defined by ICH guidelines, as it reduces the complexity of the impurity spectrum and facilitates easier validation of the cleaning and purification processes.
How to Synthesize 2,3,6-Trisubstituted Pyrazine N-Oxides Efficiently
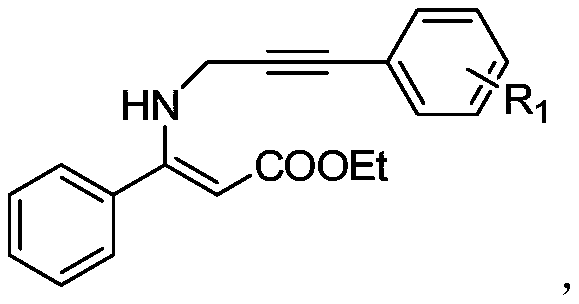
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the stoichiometry of the oxidant and the maintenance of an oxygen-rich environment. The standard protocol involves dissolving the Z-3-substituted-3-(3-arylpropynylamino)acrylate substrate in 1,2-dichloroethane (DCE), which has been identified as the optimal solvent for maximizing yield and reaction rate. Tert-butyl nitrite is then added in a molar ratio ranging from 1.0 to 3.0 equivalents relative to the substrate, with 2.0 equivalents often providing the best balance between conversion and cost. The mixture is stirred under an oxygen balloon or flow at room temperature for approximately 30 minutes until the starting material is fully consumed. Following the reaction, the mixture is quenched with water, and the product is extracted into an organic phase, washed with brine, and concentrated. For detailed operational parameters and safety precautions regarding the handling of nitrite esters, please refer to the standardized synthesis guide below.
- Dissolve the Z-3-substituted-3-(3-arylpropynylamino)acrylate substrate in 1,2-dichloroethane (DCE) under an oxygen atmosphere.
- Add tert-butyl nitrite (TBN) as the oxidant (1.0 to 3.0 equivalents) to the reaction mixture at room temperature (25°C).
- Stir the reaction for 30 minutes, quench with water, extract with ethyl acetate, and purify via column chromatography to obtain the target pyrazine N-oxide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis technology translates into tangible strategic advantages that extend beyond simple chemical transformation. The elimination of transition metal catalysts removes a significant cost center associated with purchasing expensive noble metals and implementing rigorous metal scavenging protocols. This simplification of the bill of materials (BOM) allows for more predictable budgeting and reduces exposure to the volatile pricing of rare earth elements and precious metals. Moreover, the mild reaction conditions imply lower energy consumption for heating and cooling, contributing to a reduced carbon footprint and aligning with corporate sustainability goals. The short reaction time of 30 minutes significantly increases reactor turnover rates, enabling manufacturers to produce larger volumes of high-purity pharmaceutical intermediates within the same timeframe compared to traditional multi-hour or multi-day processes.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of transition metal catalysts from the process. In conventional pyrazine synthesis, catalysts like palladium or copper can account for a substantial portion of raw material costs, and their subsequent removal requires specialized resins or filtration media that add further expense. By utilizing inexpensive tert-butyl nitrite and molecular oxygen, this new method drastically lowers the direct material cost per kilogram of product. Additionally, the simplified workup procedure—requiring only water quenching and extraction rather than complex neutralization and filtration steps—reduces labor hours and solvent consumption. These cumulative efficiencies result in a leaner manufacturing process that offers significant cost savings, making the final API intermediate more competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals that are readily available in bulk quantities globally. Tert-butyl nitrite and common solvents like dichloroethane are produced by numerous chemical suppliers, reducing the risk of single-source dependency that often plagues specialized catalyst supply chains. The robustness of the reaction against varying substrate substituents means that the same process platform can be used to manufacture a wide range of analogues, providing flexibility to respond to changing customer demands for different drug candidates. This versatility ensures that production lines can be quickly adapted without extensive re-validation or retooling, thereby minimizing downtime and ensuring continuous supply continuity for downstream pharmaceutical clients who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory synthesis to multi-ton commercial production is facilitated by the absence of exothermic hazards associated with strong acid-base neutralizations. The reaction operates safely at room temperature, removing the need for complex jacketed reactors capable of extreme temperature control, which simplifies equipment requirements for contract development and manufacturing organizations (CDMOs). From an environmental compliance standpoint, the metal-free nature of the process significantly reduces the load of heavy metals in wastewater streams, easing the burden on effluent treatment plants and ensuring adherence to increasingly strict environmental regulations. The generation of less hazardous waste and the use of greener reagents position this technology as a sustainable choice for long-term manufacturing partnerships, appealing to stakeholders focused on environmental, social, and governance (ESG) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrazine synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: Does this synthesis method require transition metal catalysts?
A: No, the patented method (CN110372613B) is explicitly metal-free, utilizing tert-butyl nitrite and molecular oxygen, which eliminates the need for expensive heavy metal removal processes.
Q: What are the optimal reaction conditions for this pyrazine formation?
A: The optimal conditions involve using 1,2-dichloroethane (DCE) as the solvent at room temperature (25°C) under an oxygen atmosphere, achieving yields up to 71% within 30 minutes.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the mild reaction conditions (room temperature, short reaction time) and absence of hazardous strong acids or bases make this process highly suitable for safe and efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazine N-Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free oxidative cyclization technology described in CN110372613B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory method into a robust, GMP-compliant commercial process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full-scale manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, leveraging our rigorous QC labs to monitor every batch for impurities and ensure consistency. Our state-of-the-art facilities are equipped to handle the specific solvent systems and oxygen atmospheres required for this chemistry safely and effectively.
We invite global pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis route for your next-generation drug candidates. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this metal-free approach can optimize your overall manufacturing budget. We encourage you to contact our technical procurement team today to request specific COA data for our pyrazine derivatives and to discuss route feasibility assessments for your custom synthesis projects. Let us help you accelerate your drug development timeline with reliable, high-quality intermediates produced through cutting-edge green chemistry.
