Scalable Green Synthesis of Benzoxanthene Derivatives Using Recyclable Ionic Liquids in Aqueous Media
The pharmaceutical and fine chemical industries are constantly seeking robust, environmentally benign pathways to access complex heterocyclic scaffolds, particularly those exhibiting potent biological activities such as anti-tumor and anti-viral properties. Patent CN102911151A introduces a transformative methodology for the synthesis of benzoxanthene derivatives, a class of compounds critical for drug discovery and laser technology applications. This innovation leverages a functionalized alkaline ionic liquid, specifically ethanolamine acetate, to catalyze the multicomponent condensation of alpha-naphthol, aromatic aldehydes, and 1,3-cyclohexanedione derivatives. Unlike conventional protocols that rely on hazardous organic solvents and corrosive catalysts, this patented process utilizes water as the sole reaction medium, marking a significant advancement in green chemistry principles. The technical breakthrough lies not only in the substitution of volatile organic compounds but also in the exceptional stability and recyclability of the catalytic system, which remains active and does not deactivate in the aqueous environment. For R&D directors and process chemists, this represents a viable route to high-purity intermediates with a drastically simplified work-up procedure, eliminating the need for complex extraction sequences typically associated with organic phase reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzoxanthene skeleton has relied heavily on the use of strong Lewis acids, solid acids, or metal oxides as promoters, often necessitating the use of volatile organic solvents such as dichloromethane or ethanol. These traditional methodologies, while effective in laboratory settings, present severe drawbacks when evaluated for industrial scalability and environmental compliance. The reliance on anhydrous conditions often mandates rigorous drying of reagents and equipment, increasing energy consumption and operational complexity. Furthermore, many conventional catalysts suffer from poor recyclability, leading to substantial accumulation of heavy metal waste or acidic effluents that require costly neutralization and disposal procedures. Equipment corrosion is another critical concern, particularly when using strong mineral acids or hygroscopic Lewis acids, which can compromise reactor integrity over time. Additionally, the separation of the catalyst from the final product often involves tedious chromatographic purification or multiple washing steps, which negatively impacts the overall yield and throughput. The use of beta-naphthol has been more common due to its higher reactivity, leaving the synthesis of alpha-naphthol-based derivatives underexplored and technically challenging due to lower nucleophilicity, further limiting the structural diversity accessible through standard protocols.
The Novel Approach
The methodology disclosed in CN102911151A fundamentally reengineers this synthetic landscape by introducing a water-phase catalytic system driven by ethanolamine acetate ionic liquid. This approach effectively circumvents the limitations of solvent toxicity and catalyst instability. By employing water as the reaction medium, the process inherently enhances safety profiles by eliminating fire hazards associated with organic solvents and reduces the environmental footprint through the avoidance of VOC emissions. The ionic liquid catalyst exhibits remarkable hydrostability, ensuring that it does not hydrolyze or lose activity during the reflux process, which typically occurs between 90°C and 100°C. This stability allows for a streamlined operation where the catalyst remains dissolved in the aqueous phase post-reaction, facilitating simple filtration of the solid product. The ability to synthesize benzoxa[c]anthracene derivatives using alpha-naphthol is particularly noteworthy, as it expands the chemical space available for medicinal chemists without requiring harsh activation methods. The simplicity of the protocol, involving merely mixing the reagents and heating under reflux, suggests a high degree of operational robustness suitable for large-scale manufacturing environments.
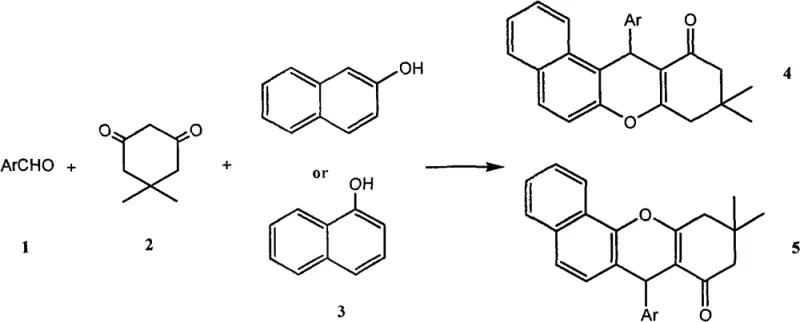
Mechanistic Insights into Ethanolamine Acetate Ionic Liquid Catalysis
The efficacy of this synthesis hinges on the unique dual-functionality of the ethanolamine acetate ionic liquid, which acts as both a Brønsted base and a hydrogen-bond donor/acceptor network within the aqueous medium. The ammonium cation and acetate anion work synergistically to activate the electrophilic carbonyl groups of the aromatic aldehyde and the 1,3-dicarbonyl compound. The acetate anion likely abstracts a proton from the active methylene group of the 1,3-cyclohexanedione derivative, generating a nucleophilic enolate species that attacks the aldehyde carbonyl. Simultaneously, the hydroxyl group of the ethanolamine moiety can participate in hydrogen bonding, stabilizing the transition states and orienting the alpha-naphthol for the subsequent cyclization step. This cooperative catalysis lowers the activation energy barrier significantly, allowing the reaction to proceed rapidly at relatively mild temperatures compared to uncatalyzed thermal conditions. The aqueous environment further enhances this mechanism through the hydrophobic effect, where organic reactants are forced into closer proximity within the water structure, effectively increasing the local concentration and accelerating the reaction kinetics without the need for high-pressure equipment.
Impurity control in this system is inherently managed by the selectivity of the ionic liquid and the solubility characteristics of the byproducts. Since the catalyst is highly specific for the Knoevenagel condensation followed by Michael addition and cyclization, side reactions such as polymerization of the aldehyde or self-condensation of the diketone are minimized. The use of water as a solvent ensures that polar impurities and unreacted starting materials remain in the aqueous filtrate, while the desired benzoxanthene product, being largely non-polar, precipitates out as a solid. This phase separation acts as a built-in purification step, yielding crude products of high purity that often require only a single recrystallization from ethanol to meet stringent pharmaceutical specifications. The absence of nitrogen-containing heterocycles in the ionic liquid structure itself prevents the introduction of genotoxic impurities often associated with amine-based catalysts, a critical consideration for regulatory compliance in API intermediate manufacturing.
How to Synthesize Benzoxanthene Derivatives Efficiently
The operational simplicity of this patented route makes it highly attractive for process development teams aiming to establish a reliable supply chain for complex heterocyclic intermediates. The procedure eliminates the need for inert atmosphere techniques or specialized drying protocols, allowing for execution in standard glass-lined or stainless steel reactors. The stoichiometry is straightforward, utilizing a 1:1:1 molar ratio of the three key components, with the catalyst loading kept low at 5 to 10 mol% relative to the aromatic aldehyde. Water constitutes the bulk of the reaction mass, typically 50% to 80%, ensuring excellent heat transfer and temperature control during the exothermic condensation phases. Following the reaction period of 0.5 to 2.5 hours, the isolation is achieved via simple filtration, avoiding energy-intensive distillation or extraction processes. For detailed standard operating procedures and specific parameter optimization for your target molecule, please refer to the technical guide below.
- Prepare the reaction mixture by combining alpha-naphthol, aromatic aldehyde, and 1,3-cyclohexanedione derivative in a 1: 1:1 molar ratio.
- Add the ethanolamine acetate ionic liquid catalyst (5-10 mol% relative to aldehyde) and water (50-80% of total mass) to the flask.
- Reflux the mixture at 90-100°C for 0.5 to 2.5 hours, then filter the solid crude product and recrystallize from 95% ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this aqueous ionic liquid technology offers profound strategic advantages regarding cost structure and logistical reliability. The shift away from expensive, flammable organic solvents to water dramatically reduces raw material costs and eliminates the regulatory burdens associated with solvent storage and disposal. This transition directly translates to a leaner cost of goods sold (COGS), making the final intermediates more competitive in the global market. Furthermore, the recyclability of the catalyst solution means that the effective consumption of the catalytic promoter is negligible over multiple batches, decoupling production volume from catalyst procurement needs. This creates a more predictable and stable supply chain, less vulnerable to fluctuations in the pricing of specialty chemicals. The robustness of the reaction conditions also implies a lower risk of batch failures due to moisture sensitivity, enhancing overall manufacturing reliability and ensuring consistent delivery schedules to downstream clients.
- Cost Reduction in Manufacturing: The elimination of organic solvents removes a significant line item from the production budget, encompassing not just the purchase price of the solvents but also the costs related to their recovery, recycling, or incineration. Additionally, the low catalyst loading and its ability to be reused without regeneration significantly lower the auxiliary material costs. The simplified work-up procedure reduces labor hours and utility consumption associated with distillation and chromatography, leading to substantial overall process economics improvements that can be passed on to the customer.
- Enhanced Supply Chain Reliability: Utilizing water as a solvent mitigates the risks associated with the supply volatility of petrochemical-derived solvents. The raw materials, including alpha-naphthol and various aromatic aldehydes, are commodity chemicals with well-established global supply chains, ensuring long-term availability. The stability of the catalyst in water means it can be stored and transported with fewer safety restrictions compared to pyrophoric or moisture-sensitive Lewis acids, streamlining logistics and reducing lead times for raw material intake.
- Scalability and Environmental Compliance: The process is inherently scalable because heat management in aqueous systems is superior to that in organic solvents, reducing the risk of thermal runaways during scale-up. From an environmental standpoint, the E-factor of this process is exceptionally low due to the absence of organic waste streams, facilitating easier compliance with increasingly stringent environmental regulations. This green profile enhances the corporate sustainability metrics of any organization adopting this technology, aligning with global ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this water-phase synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of using ionic liquids over traditional Lewis acids for benzoxanthene synthesis?
A: Unlike traditional Lewis acids which often require anhydrous conditions and generate corrosive waste, the ethanolamine acetate ionic liquid described in CN102911151A is stable in water, non-corrosive, and allows for the use of water as a green solvent, significantly simplifying downstream processing and waste treatment.
Q: Can the catalyst system be recycled for industrial scale-up?
A: Yes, the patent explicitly states that the filtered aqueous phase containing the ionic liquid catalyst can be reused directly for subsequent batches without additional treatment, maintaining catalytic activity and reducing raw material consumption.
Q: What is the typical reaction time and temperature for this synthesis?
A: The reaction proceeds efficiently under reflux conditions at temperatures between 90°C and 100°C, with completion times ranging from 0.5 to 2.5 hours depending on the specific electronic nature of the aromatic aldehyde substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxanthene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of transitioning towards sustainable and efficient manufacturing processes for high-value pharmaceutical intermediates. Our technical team has extensively analyzed the potential of the ionic liquid-catalyzed aqueous synthesis described in CN102911151A and is fully prepared to support its industrial implementation. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless and risk-mitigated. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of benzoxanthene derivative meets the exacting standards required for drug substance manufacturing. We are committed to delivering not just a product, but a comprehensive supply solution that balances quality, cost, and environmental responsibility.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific project requirements. By leveraging this green chemistry platform, we can help you optimize your supply chain for both economic and ecological performance. Please contact our technical procurement team to request specific COA data, route feasibility assessments, and sample availability for your next development campaign. Let us partner with you to bring these advanced therapeutic intermediates to market faster and more sustainably.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
