Advanced One-Pot Synthesis of Triarylmethanes for Scalable Pharmaceutical Intermediate Manufacturing
Advanced One-Pot Synthesis of Triarylmethanes for Scalable Pharmaceutical Intermediate Manufacturing
The landscape of organic synthesis for high-value scaffolds is constantly evolving, driven by the need for more efficient and sustainable methodologies. A significant breakthrough in this domain is detailed in Chinese Patent CN112159312B, which discloses a novel synthetic method for triarylmethane compounds. These compounds are critical structural motifs found in a vast array of applications, ranging from optical reagents and dyes to biologically active pharmaceutical ingredients with antioxidant and antitumor properties. The patent introduces a groundbreaking strategy that utilizes o-hydroxyphenyl p-QMs (para-quinone methides) as the sole raw material. Through a sophisticated tandem sequence involving an inverse-Friedel-Crafts reaction followed by a 1,6-conjugated addition reaction, this method achieves the construction of the triarylmethane skeleton in a single pot. This represents a paradigm shift from multi-step traditional syntheses, offering a streamlined pathway that is both atom-economical and operationally simple for the production of high-purity pharmaceutical intermediates.
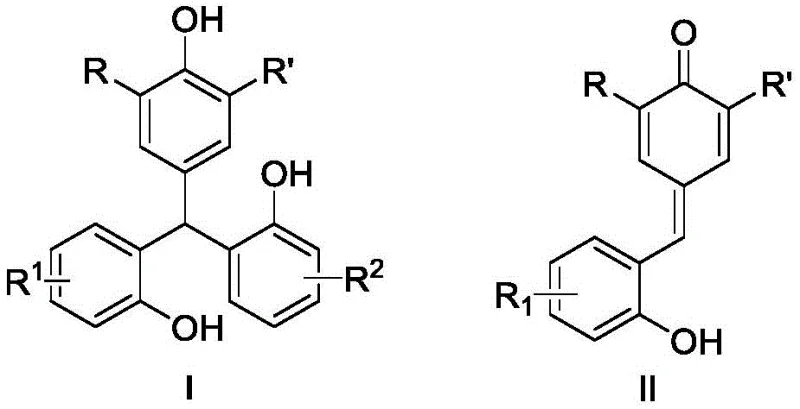
This innovative approach addresses the longstanding challenges associated with the functionalization of electron-rich aromatic systems. By leveraging the unique reactivity of para-quinone methides under Lewis acid catalysis, the process bypasses the need for pre-functionalized organometallic reagents. The general reaction scheme illustrates the transformation where the central carbon of the triarylmethane structure is formed through the sequential addition of aromatic rings. This methodology not only enhances the structural diversity accessible to chemists but also aligns with the principles of green chemistry by reducing waste and energy consumption. For procurement managers and supply chain directors, understanding this underlying chemistry is crucial, as it signals a move towards more robust and cost-effective manufacturing routes for complex fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triarylmethane derivatives has relied heavily on classical Friedel-Crafts alkylation, coupling reactions, or reduction strategies. These conventional pathways are fraught with significant limitations that hinder their efficiency in a modern industrial setting. Traditional Friedel-Crafts reactions often necessitate the prior synthesis of complex precursors such as diphenylmethanol or triarylmethanol, adding extra steps and reducing overall throughput. Furthermore, many established protocols depend on the use of Grignard reagents, which are notoriously unstable, moisture-sensitive, and hazardous to handle on a large scale. The requirement for expensive transition metal catalysts in coupling reactions further exacerbates the cost burden and introduces complications regarding residual metal removal, a critical quality parameter for pharmaceutical intermediates. Additionally, these methods frequently exhibit limited substrate scope, often restricted to electron-rich aromatic hydrocarbons, thereby limiting the chemical diversity of the final products.
The Novel Approach
In stark contrast, the method described in Patent CN112159312B offers a transformative solution by employing a tandem inverse-Friedel-Crafts/1,6-conjugated addition reaction. This novel approach utilizes o-hydroxyphenyl p-QMs as a versatile building block, eliminating the need for unstable organometallic reagents or complex alcohol precursors. The reaction proceeds under mild conditions, typically between 25°C and 60°C, in the presence of a Lewis acid catalyst and water. This one-pot synthesis strategy drastically simplifies the operational workflow, as it combines multiple bond-forming events into a single reaction vessel. The use of water as a co-reagent is particularly advantageous, acting as a proton source to facilitate the reaction without the need for harsh acidic or basic conditions. This results in a process that is not only higher yielding but also significantly safer and more environmentally benign, making it an ideal candidate for the cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into AgPF6-Catalyzed Tandem Reaction
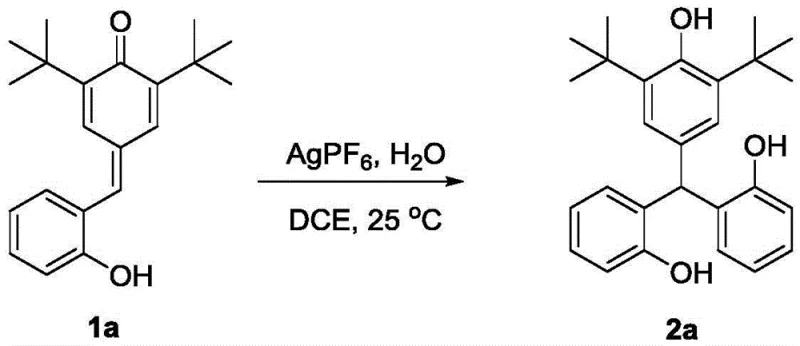
The core of this synthetic innovation lies in the precise activation of the para-quinone methide substrate by a Lewis acid catalyst, such as silver hexafluorophosphate (AgPF6). The mechanism initiates with the coordination of the Lewis acid to the oxygen or the exocyclic double bond of the p-QM, increasing the electrophilicity of the system. This activation triggers the inverse-Friedel-Crafts reaction, where an aromatic ring attacks the electrophilic center. Subsequently, the intermediate undergoes a 1,6-conjugated addition, effectively stitching together the three aryl groups around the central methane carbon. The presence of water plays a pivotal role in this catalytic cycle, likely facilitating proton transfer steps that stabilize the intermediate species and drive the reaction to completion. This mechanistic elegance allows for the formation of the sterically crowded triarylmethane core with high regioselectivity and minimal byproduct formation, ensuring a clean impurity profile that is highly desirable for R&D directors focused on product purity.
Furthermore, the tolerance of this catalytic system to various functional groups is a testament to its robustness. The reaction conditions are sufficiently mild to preserve sensitive substituents such as halogens, nitro groups, and bulky alkyl chains like tert-butyl groups. This broad substrate compatibility means that a single optimized protocol can be applied to generate a diverse library of triarylmethane derivatives, accelerating the drug discovery process. The high experimental yields reported, such as 92% for the conversion of compound 1a to 2a, underscore the efficiency of the catalyst turnover and the thermodynamic favorability of the tandem sequence. For technical teams, this implies a reliable process that minimizes batch-to-batch variability and maximizes resource utilization.
How to Synthesize Triarylmethane Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural parameters to ensure optimal results. The process begins with the preparation of the reaction environment under an inert atmosphere to prevent unwanted oxidation or moisture interference, although water is a deliberate reagent. The precise stoichiometry of the Lewis acid catalyst is critical; typically, a loading of 1-10 mol% relative to the substrate is sufficient to drive the reaction to completion. The choice of solvent, such as 1,2-dichloroethane (DCE), provides the necessary solubility for the organic substrates while maintaining stability under the reaction conditions. Following the reaction period, which generally spans 48 to 96 hours depending on the specific substrate electronics, the workup involves standard purification techniques. The detailed standardized synthesis steps for replicating this high-efficiency transformation are outlined in the guide below.
- Under nitrogen protection, add the Lewis acid catalyst (such as AgPF6) to the reactor at a loading of 1-10 mol% relative to the substrate.
- Introduce the o-hydroxyphenyl p-QMs substrate and the reaction solvent (e.g., 1,2-dichloroethane) into the reactor, followed by the addition of water.
- Stir the reaction mixture at a mild temperature of 25-60°C for 48-96 hours, then purify the resulting triarylmethane product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic methodology offers profound benefits for procurement and supply chain management within the fine chemical sector. The elimination of expensive and hazardous reagents like Grignard reagents directly translates to a reduction in raw material costs and safety compliance overheads. Moreover, the low catalyst loading required for this transformation means that the consumption of precious metal salts is minimized, further driving down the cost of goods sold (COGS). The mild reaction temperatures (25-60°C) reduce energy consumption compared to high-temperature processes, contributing to a lower carbon footprint and reduced utility costs. These factors combined create a compelling economic case for switching to this technology for the commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this reaction eliminates the need for isolating unstable intermediates, which significantly reduces labor time and solvent usage. By avoiding the use of expensive transition metal catalysts often required in cross-coupling alternatives, the process achieves substantial cost savings in catalyst procurement and downstream metal scavenging. The high yields observed across various substrates ensure that raw material waste is minimized, maximizing the output per batch and improving overall process economics without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this method against varying substrate electronic properties ensures a consistent supply of diverse triarylmethane derivatives. Since the starting materials (o-hydroxyphenyl p-QMs) are becoming increasingly accessible and the reaction conditions are not overly sensitive to minor fluctuations, the risk of batch failure is significantly mitigated. This reliability is crucial for maintaining continuous production schedules and meeting the stringent delivery timelines demanded by global pharmaceutical clients, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The simplicity of the post-reaction workup, which typically involves standard column chromatography using petroleum ether and ethyl acetate, facilitates easy scale-up from gram to kilogram scales. The absence of toxic heavy metals and the use of relatively benign solvents align with increasingly strict environmental regulations. This makes the process not only easier to permit but also more sustainable in the long term, ensuring that the supply chain remains resilient against regulatory changes and environmental scrutiny.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled a set of frequently asked questions based on the specific technical details and beneficial effects described in the patent literature. These answers address common concerns regarding reaction scope, scalability, and purification, providing a clear picture of what to expect when implementing this synthesis. Understanding these nuances is essential for assessing the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of this triarylmethane synthesis method over traditional Friedel-Crafts alkylation?
A: Unlike traditional methods that often require complex precursors like diphenylmethanol or unstable Grignard reagents, this novel approach utilizes o-hydroxyphenyl p-QMs as a single starting material. It operates under mild conditions (25-60°C) with a low loading of Lewis acid catalyst, eliminating the need for expensive transition metal catalysts and simplifying the post-reaction purification process significantly.
Q: What is the substrate scope and yield efficiency of this catalytic system?
A: The method demonstrates excellent substrate universality, accommodating various substituents such as alkyl, alkoxy, nitro, halogen, and aryl groups on the benzene rings. Experimental data indicates high efficiency, with isolated yields ranging from 63% to 97% across different substrates, proving its robustness for diverse triarylmethane derivative synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly amenable to scale-up due to its operational simplicity and mild reaction conditions. The use of common solvents like 1,2-dichloroethane and the avoidance of sensitive reagents allow for straightforward handling in large reactors, while the easy separation of products via standard column chromatography ensures consistent quality control for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triarylmethane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in Patent CN112159312B for the production of high-value triarylmethane scaffolds. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of triarylmethane intermediate delivered meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this efficient synthetic route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this technology can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global market for fine chemical intermediates.
