Advanced One-Step Synthesis of Substituted 3-Alkyl Four-Membered Cycloenones for Commercial Scale-Up
The chemical landscape for constructing strained ring systems has evolved significantly with the disclosure of patent CN102146028B, which details a robust methodology for synthesizing various substituted 3-alkyl four-membered cycloenone compounds. This technology represents a pivotal shift away from the historically challenging preparation of cyclobutenones, utilizing a direct reaction between 2,3-allenoate esters and dialkylzinc reagents. Unlike conventional pathways that rely on fleeting and hazardous intermediates, this invention leverages a one-pot 1,4-addition followed by cyclization and subsequent 1,2-addition/elimination to generate complex cyclic structures in a single operational step. For R&D directors and process chemists, this approach offers a compelling alternative for accessing high-value scaffolds found in natural products and bioactive molecules. The ability to introduce three different substituents at the 2, 3, and 4 positions of the cycloenone ring with high precision addresses a critical gap in synthetic organic chemistry, providing a reliable foundation for developing new active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetra-atomic ring ketene compounds has been plagued by significant practical and theoretical hurdles that hindered their widespread adoption in industrial manufacturing. Traditional methods predominantly relied on the [2+2] cycloaddition reaction between ketenes and enamine salts, a pathway fraught with instability and handling difficulties. Ketenes are notoriously reactive and often require generation in situ under strictly controlled conditions, while enamine salts can be sensitive to moisture and air, complicating the reaction setup. Furthermore, controlling the regioselectivity in these traditional couplings was exceptionally difficult, often leading to complex mixtures of isomers that required extensive and costly purification efforts. The precursors themselves were not only hard to obtain but also posed safety risks during storage and transport, making the scale-up of such processes economically unviable for large-scale production of fine chemicals.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes readily available 2,3-allenoate esters and dialkylzinc reagents to achieve the same structural complexity with remarkable efficiency. This method operates under relatively mild thermal conditions using common solvents like toluene or xylene, eliminating the need for cryogenic temperatures or exotic catalysts. The reaction proceeds through a concerted mechanism that inherently favors the formation of the desired 3-alkyl four-membered cycloenone skeleton with excellent regioselectivity. By avoiding the use of unstable ketene precursors, the process significantly enhances operational safety and simplifies the supply chain logistics for raw materials. The one-step nature of the transformation means that intermediates do not need to be isolated, reducing solvent consumption and processing time, which translates directly into lower manufacturing costs and a reduced environmental footprint for the production of these valuable intermediates.
Mechanistic Insights into Dialkylzinc-Mediated Cyclization
The core of this technological breakthrough lies in the unique reactivity of dialkylzinc species towards the electron-deficient allenoate system, initiating a cascade of bond-forming events. The mechanism begins with a nucleophilic 1,4-addition of the alkyl group from the dialkylzinc to the central carbon of the allenoate moiety, generating a zinc-enolate intermediate. This intermediate then undergoes an intramolecular cyclization where the enolate oxygen attacks the carbonyl carbon, closing the four-membered ring. Following this cyclization, a 1,2-addition and subsequent elimination step occurs, effectively expelling the methoxy group and establishing the final double bond within the ring system. This sequence ensures that the alkyl group from the dialkylzinc is specifically installed at the 3-position, while the substituents from the original allenoate ester are retained at the 2 and 4 positions, allowing for precise molecular editing.
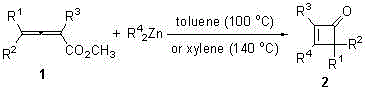
From an impurity control perspective, this mechanistic pathway offers distinct advantages over radical or transition-metal catalyzed alternatives. Because the reaction does not rely on external transition metal catalysts like palladium or rhodium, there is no risk of heavy metal contamination in the final product, a critical parameter for pharmaceutical intermediates. The high regioselectivity inherent in the 1,4-addition step minimizes the formation of positional isomers, which are often the most difficult impurities to separate chromatographically. Additionally, the use of dialkylzinc, while requiring careful handling due to pyrophoricity, results in clean reaction profiles where the primary byproducts are zinc salts that are easily removed during the aqueous workup. This cleanliness of the reaction profile ensures that the final cycloenone products can be purified to high standards using standard techniques like rapid column chromatography or crystallization, ensuring consistent quality for downstream applications.
How to Synthesize 3-Alkyl Four-Membered Cycloenones Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The process begins by charging a reaction vessel with the specific 2,3-allenoate ester substrate and the chosen aromatic solvent, typically toluene or xylene, under an inert nitrogen atmosphere to prevent oxidation of the sensitive organozinc reagent. Once the mixture is agitated, the dialkylzinc solution is added dropwise to control the exotherm, after which the reaction mixture is heated to the requisite temperature, ranging from 100°C for toluene to 140°C for xylene. Detailed standardized operating procedures for scaling this reaction, including specific quenching and purification steps, are outlined in the technical guide below to ensure safe and efficient execution.
- Under nitrogen protection, add 2,3-allenoate ester substrate and toluene solvent to a reaction vessel with stirring.
- Dropwise add dialkylzinc solution (3 equivalents) to the system and heat the oil bath to 100°C or 140°C depending on the solvent.
- After reaction completion, cool to zero degrees, quench with saturated ammonium chloride, extract with diethyl ether, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this zinc-mediated cyclization technology presents a strategic opportunity to optimize the cost structure and reliability of the supply chain for complex cyclic intermediates. The elimination of expensive transition metal catalysts removes a significant cost driver associated with both the purchase of precious metals and the subsequent removal processes required to meet regulatory limits. Furthermore, the reliance on commodity solvents like toluene and xylene, which are widely available and inexpensive compared to specialized chlorinated or ethereal solvents, contributes to substantial cost savings in raw material procurement. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor fluctuations in temperature or moisture compared to traditional ketene chemistry, thereby reducing the risk of batch failures and supply disruptions.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic route and the removal of costly catalytic systems. By utilizing dialkylzinc reagents which are commercially available and relatively inexpensive compared to noble metal catalysts, the direct material costs are significantly lowered. Additionally, the one-pot nature of the reaction eliminates the need for intermediate isolation and purification steps, which reduces labor hours, equipment usage time, and solvent volumes. This process intensification leads to a more streamlined manufacturing workflow that inherently lowers the cost of goods sold, making the final high-purity cycloenone intermediates more competitive in the global market without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the use of stable and readily accessible starting materials such as 2,3-allenoate esters and dialkylzinc solutions. Unlike the unstable ketene precursors used in older methods, these reagents can be sourced from multiple suppliers and stored with standard safety protocols, reducing the risk of raw material shortages. The high yields reported in the patent examples, ranging significantly across different substrates, indicate a robust process that is less prone to variability, ensuring consistent output volumes. This reliability allows for more accurate demand forecasting and inventory management, enabling manufacturers to maintain steady production schedules and meet tight delivery deadlines for their downstream customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: Scaling this process to commercial levels is facilitated by the use of standard reactor materials and the absence of complex catalytic cycles that often require specialized equipment. The workup procedure involves standard aqueous washes with hydrochloric acid and sodium bicarbonate, generating waste streams that are easier to treat and neutralize compared to those containing heavy metals. The high atom economy of the cyclization step, combined with the ability to recover and recycle the aromatic solvents, aligns well with modern green chemistry principles and environmental regulations. This compliance reduces the regulatory burden and potential fines associated with waste disposal, while the scalability ensures that the technology can support production volumes ranging from pilot batches to multi-ton annual requirements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and benefits for stakeholders evaluating this technology for integration into their existing manufacturing portfolios. Understanding these details is crucial for assessing the feasibility of adopting this route for specific target molecules and for planning the necessary resource allocation for process development.
Q: What are the primary advantages of this zinc-mediated cyclization over traditional ketene methods?
A: This method eliminates the need for unstable ketene and enamine salt precursors, offering superior regioselectivity and operational simplicity without requiring transition metal catalysts.
Q: What solvents and temperatures are optimal for this synthesis?
A: The process utilizes common industrial solvents like toluene at 100°C or xylene at 140°C, facilitating easy scale-up and solvent recovery.
Q: How does this route impact impurity profiles for pharmaceutical intermediates?
A: The high regioselectivity of the 1,4-addition/cyclization sequence minimizes side reactions, resulting in cleaner crude products that are easier to purify to stringent specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkyl Four-Membered Cycloenones Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely qualified to adapt the zinc-mediated cyclization protocol described in CN102146028B to meet your specific project needs, ensuring that stringent purity specifications are met through our rigorous QC labs. We understand that the transition from bench-scale discovery to full-scale manufacturing requires not just chemical expertise but also a deep commitment to quality assurance and process safety. By leveraging our state-of-the-art facilities and experienced engineering staff, we can guarantee a consistent supply of high-quality intermediates that adhere to the highest industry standards.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific supply chain requirements. Contact us today to request a Customized Cost-Saving Analysis that details the potential economic benefits of switching to this methodology for your target compounds. Our experts are ready to provide specific COA data from previous runs and comprehensive route feasibility assessments to help you make informed decisions. Let us partner with you to optimize your production costs and secure a reliable supply of these critical building blocks for your next generation of products.
