Advanced Synthesis of 3,4,5-Trifluorophenyl Tert-Butyl Ether for High-Performance Liquid Crystal Manufacturing
Advanced Synthesis of 3,4,5-Trifluorophenyl Tert-Butyl Ether for High-Performance Liquid Crystal Manufacturing
The demand for high-performance liquid crystal materials continues to drive innovation in the synthesis of fluorinated aromatic intermediates. Patent CN101891599B introduces a transformative methodology for producing 3,4,5-trifluorophenyl tert-butyl ether, a critical precursor for fourth-generation liquid crystal intermediates. This technology addresses long-standing inefficiencies in traditional synthetic pathways by leveraging a modified Grignard reaction with organic peroxides. For R&D directors and procurement specialists, this patent represents a pivotal shift towards higher yields, superior purity profiles exceeding 99.6%, and a drastically simplified waste management profile. The ability to synthesize this key intermediate without generating acidic or alkaline industrial wastewater marks a significant advancement in green chemistry for the electronic materials sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4,5-trifluorophenol, the ultimate target molecule, has relied on cumbersome multi-step processes that suffer from poor atom economy and environmental burdens. One conventional route involves the ammoniation of 3,4,5-trifluorobromobenzene followed by diazotization and hydrolysis. As illustrated in the reaction scheme below, this pathway is plagued by low overall yields, typically ranging between 50% and 60%, and generates a complex impurity profile that complicates downstream purification. The diazotization step, in particular, poses safety risks and requires stringent temperature control, while the hydrolysis step often leads to significant product loss.
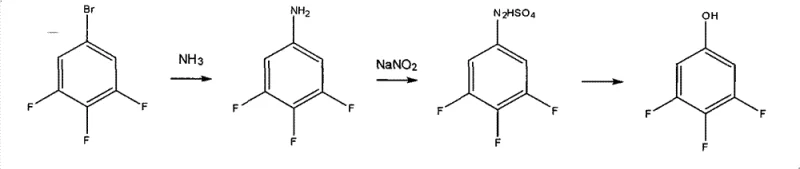
Another established method utilizes a Grignard reagent reacted with a borate ester to form a boronic acid intermediate, which is subsequently oxidized. While chemically sound, this approach is operationally complex and economically inefficient. The oxidation step often requires harsh conditions, and the subsequent recrystallization processes are time-consuming. Furthermore, the overall yield for this boronic acid pathway hovers around 50%, making it less attractive for large-scale commercial manufacturing where cost efficiency is paramount. The generation of boron-containing waste streams also adds to the environmental compliance costs for manufacturers.
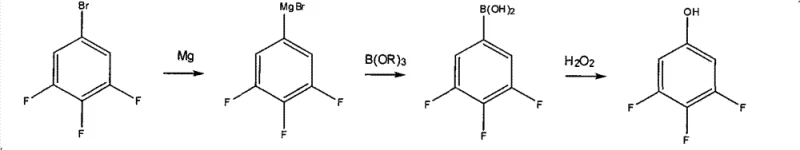
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a direct reaction between the Grignard reagent of 3,4,5-trifluorobromobenzene and tert-butyl peroxybenzoate. This single-step transformation from the Grignard species to the protected phenol ether is highly efficient. By bypassing the boronic acid intermediate and the diazonium salt formation, the process eliminates several unit operations. The reaction proceeds smoothly in a mixed solvent system of toluene and ether, allowing for precise temperature control between -10°C and 25°C. This method not only boosts the yield significantly but also simplifies the work-up procedure, as the by-products precipitate as solids that are easily removed by filtration.
Mechanistic Insights into Grignard-Peroxide Coupling
The core of this technological breakthrough lies in the nucleophilic attack of the 3,4,5-trifluorophenyl magnesium bromide on the peroxide oxygen of tert-butyl peroxybenzoate. Unlike traditional Grignard reactions with ketones or aldehydes, the reaction with organic peroxides involves a unique cleavage of the weak O-O bond. The electron-rich aryl magnesium species attacks the electrophilic oxygen, leading to the expulsion of the benzoate leaving group and the formation of the tert-butyl ether linkage. This mechanism is highly favorable due to the stability of the resulting tert-butyl group and the driving force provided by the formation of the strong magnesium-oxygen bond in the by-product.
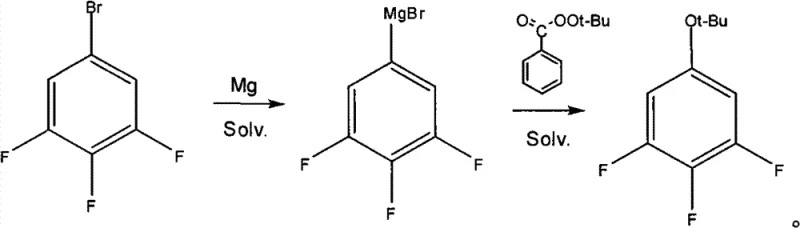
Impurity control is inherently built into this mechanistic pathway. The primary by-product, magnesium bromide benzoate, forms a hydrate upon aqueous quenching. Crucially, this salt precipitates out of the organic phase as large, coarse particles, facilitating rapid and complete filtration. This physical separation prevents the contamination of the organic product layer with inorganic salts or acidic residues, which is a common issue in acid-hydrolysis methods. Consequently, the crude product obtained after solvent removal is of exceptionally high quality, requiring only a single vacuum distillation step to achieve purity levels greater than 99.6%, thereby minimizing thermal degradation risks associated with prolonged processing.
How to Synthesize 3,4,5-Trifluorophenyl Tert-Butyl Ether Efficiently
The synthesis protocol outlined in the patent offers a robust framework for laboratory and pilot-scale production. It begins with the careful formation of the Grignard reagent using magnesium turnings and ethyl bromide as an initiator in a toluene-ether mixture. Once the Grignard species is fully generated, it is added dropwise to a cooled solution of tert-butyl peroxybenzoate. The strict control of exothermicity during this addition is vital for safety and selectivity. Following the reaction, a controlled water quench induces the precipitation of the magnesium salt, allowing for a clean phase separation. For the complete standardized operating procedure and specific molar ratios, please refer to the detailed guide below.
- Prepare the Grignard reagent by reacting 3,4,5-trifluorobromobenzene with magnesium metal in a mixed solvent of toluene and ether at 45-50°C.
- React the formed Grignard reagent with tert-butyl peroxybenzoate at controlled temperatures between -10°C and 25°C to form the ether linkage.
- Quench the reaction with water to precipitate magnesium salts, filter, and purify the crude product via vacuum distillation to achieve >99.6% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling economic and logistical benefits. The elimination of multiple reaction steps and purification stages directly translates to reduced operational expenditures. By avoiding the use of hazardous diazotization reagents and complex boron chemistry, the process lowers the barrier for safe handling and reduces the need for specialized waste treatment infrastructure. The high yield and purity ensure that less raw material is required per kilogram of finished product, optimizing the cost of goods sold (COGS) and enhancing margin potential for downstream liquid crystal manufacturers.
- Cost Reduction in Manufacturing: The process achieves a substantial reduction in manufacturing costs by eliminating the need for expensive transition metal catalysts and complex extraction protocols. The direct conversion of the Grignard reagent to the ether avoids the intermediate isolation steps required in boronic acid routes, significantly cutting down on solvent usage and energy consumption for distillation. Furthermore, the recovery of the magnesium salt by-product as a solid allows for potential valorization or easier disposal, reducing waste treatment fees associated with liquid effluent streams.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as magnesium, toluene, and tert-butyl peroxybenzoate ensures a stable and resilient supply chain. Unlike specialized catalysts that may face geopolitical or logistical bottlenecks, these raw materials are widely available from multiple global suppliers. The robustness of the reaction conditions, which tolerate standard industrial equipment like glass-lined reactors, means that production can be easily scaled or shifted between manufacturing sites without extensive requalification, ensuring continuous supply for critical LCD panel production lines.
- Scalability and Environmental Compliance: The patent explicitly demonstrates successful scale-up in a 1000-liter reactor, producing over 170 kilograms of product in a single batch. This proves the technology is ready for commercial deployment. Environmentally, the process is superior because it avoids the generation of acidic wastewater from Grignard hydrolysis and alkaline wastewater from benzoic acid extraction. The solid by-product formation simplifies effluent management, helping manufacturers meet increasingly stringent environmental regulations regarding industrial discharge and hazardous waste handling.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on yield expectations, safety parameters, and downstream processing capabilities for potential licensees or contract manufacturing partners.
Q: What is the primary advantage of this synthesis route over conventional methods?
A: The primary advantage is the significant increase in yield (up to 88% compared to 50-60%) and the elimination of acidic and alkaline wastewater, simplifying environmental compliance.
Q: How is the by-product managed in this process?
A: The by-product, magnesium bromide benzoate hydrate, precipitates as a solid upon water quenching and can be easily filtered off and recovered, avoiding complex extraction steps.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful scale-up in a 1000-liter glass-lined reactor, producing over 170 kg of product with consistent high purity, proving industrial feasibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trifluorophenyl Tert-Butyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the performance of advanced display technologies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 3,4,5-trifluorophenyl tert-butyl ether meets the exacting standards required for fourth-generation liquid crystal applications. Our commitment to quality assurance ensures consistency that your R&D and production teams can rely on.
We invite you to collaborate with us to optimize your supply chain for liquid crystal intermediates. Our experts can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient synthesis route can lower your total landed cost. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project timelines and quality benchmarks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
