Advanced Pd-Catalyzed Route for High-Purity Pyrrolone Intermediates Enabling Commercial Scale-Up and Cost-Efficient Pharmaceutical Manufacturing
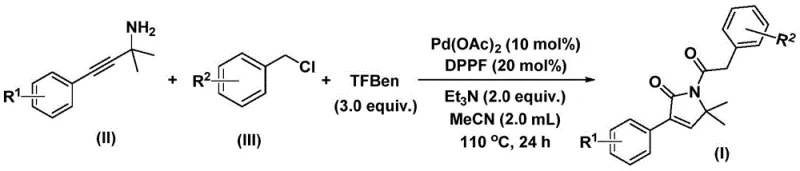
Patent CN112694430B introduces a groundbreaking one-step synthesis methodology for producing 1,5-dihydro-2H-pyrrole-2-ketone compounds that serve as essential structural frameworks in critical bioactive molecules including the antibacterial agent althiomycin and antidiabetic drug glimepiride. This innovative approach employs palladium-catalyzed bis-carbonylation chemistry to directly transform commercially available propargyl amines and benzyl chlorides into high-value pyrrolone intermediates under moderate reaction conditions of 100–120°C over 24–48 hours without requiring pressurized carbon monoxide handling. The method demonstrates exceptional operational simplicity through its use of inexpensive catalysts and ligands while maintaining broad substrate compatibility across various functional groups such as methoxy, fluoro, chloro, and trifluoromethyl moieties as evidenced by experimental yields ranging from 70% to 92%. Crucially, this breakthrough eliminates multi-step sequences inherent in conventional syntheses thereby significantly enhancing process efficiency while ensuring consistent production of pharmaceutical-grade intermediates suitable for direct incorporation into active ingredient manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for constructing the pyrrolone scaffold typically involve multi-step sequences requiring harsh reaction conditions that severely limit functional group tolerance and result in low overall yields due to cumulative losses at each transformation stage. These methods frequently depend on stoichiometric quantities of expensive reagents or generate substantial waste streams that complicate environmental compliance during large-scale manufacturing operations while increasing disposal costs significantly. Conventional carbonylation techniques often necessitate specialized equipment for handling pressurized carbon monoxide gas which introduces serious safety hazards not present in standard pharmaceutical production facilities thereby restricting accessibility for many manufacturers. The limited substrate scope observed in existing literature prevents broad application across diverse molecular architectures required for modern drug discovery programs targeting antibacterial or antidiabetic therapies while prolonged reaction times exceeding several days substantially increase production costs through extended equipment utilization periods.
The Novel Approach
The patented methodology overcomes these constraints through an elegant palladium-catalyzed bis-carbonylation process operating under ambient pressure using a safe carbon monoxide substitute derived from phenol tricarboxylate esters that eliminates hazardous gas handling requirements entirely. By employing commercially available palladium acetate with DPPF ligand in acetonitrile solvent at moderate temperatures between 100–120°C this one-step transformation efficiently converts propargyl amines and benzyl chlorides into target pyrrolones within practical timeframes of 24–48 hours while demonstrating remarkable versatility across various substituents including methoxy fluoro chloro and trifluoromethyl groups as validated through fifteen experimental examples yielding products between 70% and 92%. Post-reaction processing is streamlined through simple filtration followed by silica gel-assisted column chromatography purification eliminating complex metal removal steps typically required in transition metal-catalyzed syntheses thereby reducing both operational complexity and associated costs significantly.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
The catalytic cycle initiates with oxidative addition of benzyl chloride to palladium(0) species generated in situ from palladium acetate and DPPF ligand forming a key benzylpalladium intermediate that undergoes migratory insertion with carbon monoxide liberated from phenol tricarboxylate ester yielding an acylpalladium complex which then engages propargyl amine through nucleophilic attack at the alkyne terminus triggering cyclization to form a five-membered ring palladacycle prior to second carbon monoxide insertion constructing the six-membered transition state before reductive elimination produces the final pyrrolone product. This dual carbonylation mechanism operates with exceptional regioselectivity due to steric and electronic properties of DPPF ligand preventing undesired β-hydride elimination pathways commonly observed in similar transformations while maintaining optimal catalyst activity throughout the reaction duration through precise temperature control at 100–120°C.
The high purity profile achieved stems from inherent selectivity of this palladium-catalyzed cascade reaction which avoids common impurities such as dimerization byproducts or over-reduced species associated with traditional methods through controlled reaction parameters that suppress side reactions like homocoupling or hydrolysis pathways. Triethylamine base effectively neutralizes hydrochloric acid generated during benzyl chloride activation without promoting epimerization or decomposition of sensitive functional groups present across diverse substrates while mild conditions prevent thermal degradation pathways that typically generate colored impurities in heterocyclic syntheses thereby eliminating costly additional purification steps required to meet stringent pharmaceutical quality standards.
How to Synthesize Pyrrolone Intermediate Efficiently
This patented process enables reliable production of high-purity pyrrolone intermediates through a straightforward procedure optimized for both laboratory-scale development and commercial manufacturing environments leveraging readily accessible starting materials and standard equipment while maintaining exceptional reproducibility across multiple production batches as demonstrated in extensive experimental validation studies.
- Combine palladium acetate catalyst (10 mol%), DPPF ligand (20 mol%), triethylamine base (2.0 equiv.), phenol tricarboxylate CO substitute (3.0 equiv.), propargyl amine substrate (II), and benzyl chloride substrate (III) in acetonitrile solvent.
- Heat reaction mixture to 100–120°C under inert atmosphere with stirring for 24–48 hours while monitoring conversion via standard analytical methods.
- After completion, perform filtration through silica gel followed by column chromatography purification using standard elution protocols to isolate target pyrrolone product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis route directly addresses critical pain points in pharmaceutical intermediate supply chains by eliminating hazardous reagents and complex purification requirements that traditionally increase production costs while extending lead times for essential building blocks used across multiple therapeutic areas including antibacterial and antidiabetic drug development programs.
- Cost Reduction in Manufacturing: The elimination of pressurized carbon monoxide handling removes significant capital expenditure requirements for specialized reactor systems while reducing operational safety costs associated with high-pressure gas management protocols; utilization of inexpensive commercial catalysts alongside readily available starting materials creates substantial cost savings compared to conventional multi-step syntheses requiring expensive protecting groups or rare metal catalysts.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through globally available raw materials with stable supply chains avoiding single-source dependencies common in traditional synthetic routes; simplified process flow reduces vulnerability to intermediate shortages by consolidating multiple synthetic steps into a single operation with minimal equipment requirements ensuring consistent delivery schedules.
- Scalability and Environmental Compliance: Ambient pressure operation enables seamless scale-up from laboratory to commercial production without major equipment modifications while generating minimal waste streams through high atom economy inherent in carbonylation cascade mechanism meeting increasingly stringent environmental regulations without additional processing steps.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN112694430B's experimental data and process specifications providing insights relevant to pharmaceutical manufacturing decision-makers.
Q: What limitations does conventional pyrrolone synthesis face compared to this patented method?
A: Traditional routes require multi-step sequences with pressurized CO handling and exhibit narrow substrate scope due to poor functional group tolerance. This patented method eliminates hazardous gas requirements while accommodating diverse substituents including halogens and trifluoromethyl groups through its ambient-pressure bis-carbonylation mechanism.
Q: How does this process ensure high purity without expensive purification steps?
A: The regioselective palladium-catalyzed cascade reaction minimizes side products through controlled temperature conditions and ligand-mediated suppression of β-hydride elimination pathways. Triethylamine base neutralizes HCl byproducts without causing decomposition, enabling direct chromatographic purification without additional metal removal steps.
Q: What scalability advantages does this one-step synthesis offer for commercial production?
A: The process operates under standard pressure conditions using common equipment with minimal waste generation through high atom economy carbonylation chemistry. This allows seamless scale-up from laboratory batches to multi-ton annual production volumes while maintaining consistent yield profiles across diverse substrate combinations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolone Intermediate Supplier
Our patented technology demonstrates significant potential for producing high-purity pyrrolone intermediates essential for next-generation pharmaceutical development programs NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical instrumentation ensuring consistent quality across all batch sizes.
Contact our technical procurement team today to request specific COA data and route feasibility assessments for your application needs We offer a Customized Cost-Saving Analysis to demonstrate how this innovative synthesis can optimize your supply chain economics while ensuring reliable delivery schedules.
