Advanced Catalytic Synthesis of 2-Arylbenzimidazoles for Commercial Pharmaceutical Manufacturing
Advanced Catalytic Synthesis of 2-Arylbenzimidazoles for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective routes for synthesizing privileged heterocyclic scaffolds. A significant breakthrough in this domain is documented in Chinese Patent CN111349048A, which discloses a novel preparation method for 2-arylbenzimidazoles. This class of compounds is ubiquitous in medicinal chemistry, serving as critical intermediates for a wide array of bioactive molecules including antihelmintics, antivirals, and kinase inhibitors. The patented methodology addresses long-standing inefficiencies in traditional synthesis by employing a transition metal-catalyzed condensation between benzonitriles and o-phenylenediamines. By leveraging inexpensive earth-abundant metal salts such as zinc, cobalt, or nickel, this process eliminates the need for harsh acidic conditions and complex ligand systems, thereby offering a compelling value proposition for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-arylbenzimidazoles has relied heavily on condensation reactions that demand severe reaction conditions. Traditional protocols often utilize strong mineral acids or polyphosphoric acid mixtures to drive the cyclization, which presents substantial challenges for industrial scale-up. The use of such corrosive media necessitates specialized reactor materials to prevent equipment degradation, leading to increased capital expenditure and maintenance costs. Furthermore, the post-reaction workup in acid-catalyzed processes is notoriously difficult, often requiring extensive neutralization steps that generate large volumes of saline wastewater, complicating environmental compliance. Alternative methods reported in literature have attempted to use complex organometallic catalysts or iodine-based systems, but these often suffer from high reagent costs, the necessity for expensive ligands, or the requirement for elevated temperatures that increase energy consumption and safety risks.
The Novel Approach
The methodology outlined in patent CN111349048A represents a paradigm shift towards greener and more economical synthesis. By utilizing simple inorganic carbonates as bases and readily available transition metal salts as catalysts, the process achieves high conversion rates under significantly milder thermal conditions. The reaction proceeds efficiently in common organic solvents such as chlorobenzene, THF, or DMF, avoiding the need for exotic or hazardous reagents. This approach not only simplifies the operational procedure but also enhances the safety profile of the manufacturing process. The elimination of strong acids and complex ligand systems streamlines the purification workflow, allowing for easier isolation of the target product with high purity. This robustness makes the method particularly attractive for the commercial production of high-purity pharmaceutical intermediates where impurity profiles are strictly regulated.
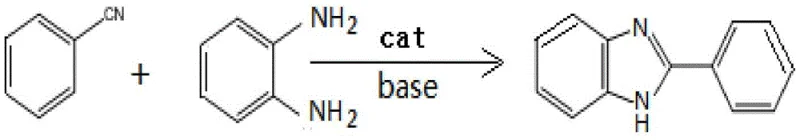
Mechanistic Insights into Transition Metal-Catalyzed Cyclization
The core of this innovation lies in the activation of the nitrile group by the transition metal catalyst, facilitating nucleophilic attack by the amine. In the absence of expensive phosphine ligands, the metal center—whether zinc, cobalt, or nickel—coordinates with the nitrogen of the benzonitrile, increasing its electrophilicity. This coordination lowers the activation energy required for the initial addition of the o-phenylenediamine. Subsequent intramolecular cyclization and dehydration steps lead to the formation of the stable benzimidazole ring system. The choice of metal salt is critical; for instance, zinc chloride has been shown to be highly effective, likely due to its Lewis acidity which complements the basic conditions provided by the carbonate. This synergistic interaction between the metal catalyst and the base ensures a smooth reaction trajectory without the formation of stubborn by-products that often plague acid-catalyzed routes.
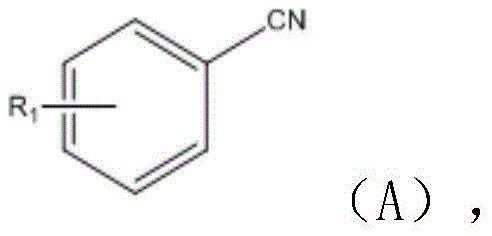
Furthermore, the mechanistic pathway allows for exceptional substrate tolerance, which is crucial for generating diverse chemical libraries. The electronic nature of the substituents on the aromatic rings does not significantly hinder the reaction, as evidenced by the successful synthesis of derivatives bearing electron-withdrawing groups like halogens and cyano groups, as well as electron-donating groups like alkyl and alkoxy moieties. The ability to tolerate functional groups such as hydroxyl (-OH) and thiol (-SH) on the diamine component without protecting group chemistry is a distinct advantage. This chemoselectivity implies that the catalyst system is sufficiently mild to avoid side reactions such as polymerization or over-oxidation, ensuring that the final impurity spectrum is clean and manageable. Such control is vital for R&D teams aiming to rapidly iterate on lead compounds without being bottlenecked by synthetic complexity.
How to Synthesize 2-Arylbenzimidazole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to stoichiometry and reaction parameters to maximize yield and purity. The general protocol involves charging the reactor with the benzonitrile substrate and the o-phenylenediamine derivative in a molar ratio that favors complete consumption of the nitrile, typically using an excess of the diamine. An inorganic base, such as potassium carbonate or cesium carbonate, is added to scavenge protons generated during the cyclization, driving the equilibrium forward. The catalyst loading is kept low, often between 1 to 20 mol%, reflecting the high turnover efficiency of the metal salts. The mixture is then heated under a nitrogen blanket to exclude moisture and oxygen, which could potentially deactivate the catalyst or oxidize sensitive intermediates. Detailed standardized synthesis steps follow below.
- Combine benzonitrile derivatives, o-phenylenediamine substrates, an inorganic carbonate base, and a catalytic amount of zinc, cobalt, or nickel salt in a suitable organic solvent under an inert nitrogen atmosphere.
- Heat the reaction mixture to a moderate temperature range of 80-130°C and maintain stirring for 24 to 36 hours to ensure complete conversion to the target benzimidazole scaffold.
- Upon completion, cool the mixture, remove the solvent via rotary evaporation, perform liquid-liquid extraction with dichloromethane, wash with saturated brine or carbonate solution, dry over anhydrous sodium sulfate, and purify the crude product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits beyond mere technical feasibility. The primary driver for cost optimization is the replacement of expensive, specialized catalysts with commodity chemicals. Zinc, cobalt, and nickel salts are produced in massive quantities globally, ensuring a stable supply chain and shielding the manufacturing process from the volatility associated with precious metals like palladium or rhodium. Additionally, the simplified workup procedure reduces the consumption of auxiliary materials such as quenching agents and extensive washing solvents. This reduction in material usage directly translates to lower operating expenses and a smaller physical footprint for waste treatment facilities, aligning with modern sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of costly ligands and the use of inexpensive metal salts. Traditional methods often require stoichiometric amounts of activating agents or expensive catalytic systems that are difficult to recover. In contrast, this method uses catalytic amounts of cheap salts that do not require complex recovery systems, significantly lowering the bill of materials. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling cycles, contributing to overall operational efficiency. The avoidance of corrosive acids also extends the lifespan of reactor vessels and piping, reducing long-term capital depreciation and maintenance costs.
- Enhanced Supply Chain Reliability: Sourcing raw materials for chemical synthesis can be a bottleneck, especially for specialized reagents. This protocol relies on benzonitriles and phenylenediamines, which are bulk commodities available from multiple global suppliers, ensuring redundancy and security of supply. The solvents used, such as toluene, THF, and DMF, are also standard industrial solvents with robust supply chains. By minimizing dependence on niche reagents or custom-synthesized catalysts, manufacturers can mitigate the risk of production delays caused by supplier shortages. This reliability is critical for maintaining continuous production schedules for active pharmaceutical ingredients (APIs) where downtime is financially detrimental.
- Scalability and Environmental Compliance: Scaling a chemical process from grams to tons often reveals hidden complexities, but this method is inherently scalable due to its homogeneous nature and lack of hazardous gases. The reaction does not generate toxic by-products that require scrubbing, and the absence of strong acids simplifies effluent treatment. The use of nitrogen as the only atmosphere gas is standard and safe. These factors facilitate a smoother technology transfer from R&D to commercial production, reducing the time-to-market for new drug candidates. Moreover, the cleaner profile of the process aids in meeting increasingly stringent environmental regulations regarding waste discharge and solvent emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable reference for technical assessment. Understanding these nuances is essential for evaluating the fit of this technology within your existing manufacturing portfolio.
Q: What are the primary advantages of this catalytic system over traditional acid-catalyzed methods?
A: Unlike traditional methods requiring corrosive polyphosphoric acid or complex metal ligand systems, this patent utilizes inexpensive, earth-abundant metal salts (Zn, Co, Ni) without additional ligands. This significantly simplifies the workup process, reduces equipment corrosion risks, and lowers the overall environmental footprint of the manufacturing process.
Q: What is the substrate scope for the benzonitrile and diamine components?
A: The method demonstrates robust tolerance for various substituents. The benzonitrile component can bear hydrogen, halogens, alkyl, alkoxy, cyano, or ester groups at ortho, meta, or para positions. Similarly, the o-phenylenediamine derivative allows for substitution with hydrogen, halogens, alkyl, or alkoxy groups, and can even include -OH or -SH functionalities, providing high versatility for diverse pharmaceutical scaffolds.
Q: Does this process require specialized high-pressure equipment?
A: No, the reaction operates under standard atmospheric pressure in a nitrogen environment. The temperatures required are moderate (80-130°C), which means the process can be scaled using standard glass-lined or stainless steel reactors commonly found in fine chemical manufacturing facilities, eliminating the need for specialized high-pressure autoclaves.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Arylbenzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the methodology described in CN111349048A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art facilities capable of handling the specific solvent systems and thermal requirements of this benzimidazole synthesis. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, ensuring that every batch of 2-arylbenzimidazole intermediate meets the highest international standards for pharmaceutical applications.
We invite potential partners to leverage our technical expertise to optimize their supply chains. Whether you require custom synthesis of specific substituted derivatives or large-scale production of the core scaffold, our team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and reduce your overall manufacturing costs.
