Scalable One-Pot Synthesis of Pyranoindolone Fused Medium-Ring Derivatives for Advanced Drug Discovery
Introduction to Novel Pyranoindolone Synthesis Technology
The pharmaceutical industry continuously seeks efficient pathways to access complex heterocyclic scaffolds that serve as critical cores for bioactive molecules. Patent CN108997362B, published in July 2021, introduces a groundbreaking methodology for the construction of pyranoindolone fused medium-ring derivatives, a class of compounds known for their potent biological activities including antitumor properties. This technology addresses the longstanding challenge of synthesizing seven and eight-membered rings fused to the pyranoindolone core, which are notoriously difficult to access via traditional cyclization strategies. The disclosed method employs a tandem sequence involving base-promoted C-C bond insertion followed by a catalytic oxidative coupling, offering a streamlined alternative to multi-step syntheses. By utilizing readily available indole alkynones and cyclic ketones, this approach significantly lowers the barrier to entry for producing these high-value intermediates. The structural versatility allowed by this protocol enables the rapid generation of diverse libraries for drug discovery programs, positioning it as a key asset for R&D teams focused on oncology and other therapeutic areas. 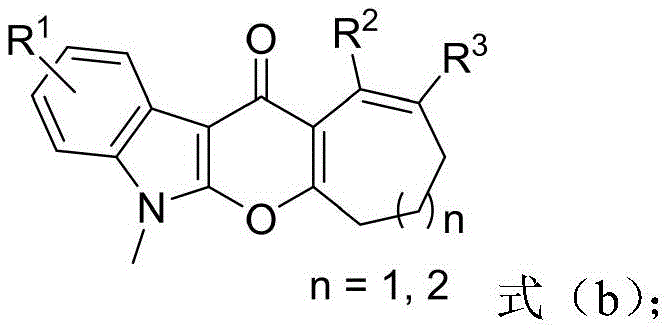
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of medium-sized rings fused to indole scaffolds has relied heavily on transition metal catalysis involving precious metals such as Rhodium, Rhenium, or Gold. Literature precedents, including works by Stoltz and Tambar, often necessitate the use of highly strained three or four-membered ring precursors or complex phenylalkyne derivatives to drive the ring expansion thermodynamics. These traditional routes suffer from significant drawbacks, including the high cost of noble metal catalysts, which complicates the economic viability of large-scale production. Furthermore, the requirement for specialized, often unstable precursors increases the complexity of the supply chain and introduces safety hazards associated with handling strained cyclic systems. The purification processes associated with removing trace heavy metals from the final active pharmaceutical ingredient (API) intermediates add further operational burdens and cost, making these conventional methods less attractive for commercial manufacturing where cost of goods sold (COGS) is a critical metric.
The Novel Approach
In stark contrast, the methodology described in CN108997362B utilizes a base-promoted ring expansion strategy that bypasses the need for strained ring precursors entirely. By employing simple cyclic ketones, such as cyclohexanone or cyclopentanone derivatives, the reaction achieves ring expansion through a C-C bond insertion mechanism driven by a mild inorganic base like Cesium Carbonate. This is followed by a Zinc-catalyzed oxidative C-H/O-H coupling step that closes the pyran ring efficiently. This dual-step one-pot protocol not only simplifies the operational workflow by eliminating intermediate isolation but also replaces expensive noble metals with earth-abundant Zinc salts. The use of air as the terminal oxidant in the second step further enhances the green chemistry profile of the process. This shift from precious metal catalysis to base-metal catalysis represents a paradigm shift in the synthesis of these complex architectures, offering a robust and economically superior route for industrial application. 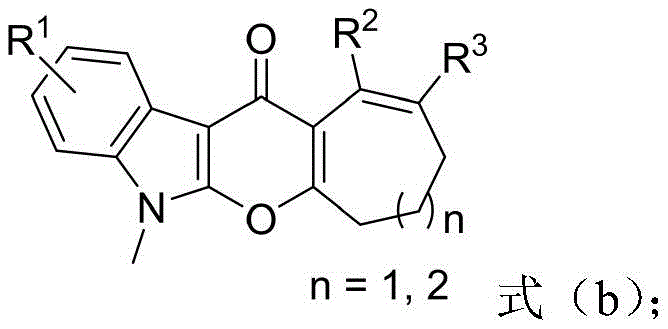
Mechanistic Insights into Base-Promoted Ring Expansion and Zinc Catalysis
The mechanistic pathway of this transformation is a sophisticated interplay of nucleophilic addition and radical oxidative coupling. In the first stage, the base facilitates the deprotonation of the cyclic ketone, generating a nucleophilic enolate species that attacks the electron-deficient alkyne moiety of the indole alkynone substrate. This initial C-C bond formation triggers a cascade that results in the expansion of the carbocyclic ring, forming a seven or eight-membered intermediate with a pendant hydroxyl group. This step is critical as it establishes the carbon framework of the medium ring without the need for pre-installed strain. The reaction conditions, specifically the use of polar aprotic solvents like DMSO and temperatures around 60°C, are optimized to favor this insertion while minimizing side reactions such as polymerization or hydrolysis of the sensitive alkyne functionality.
The subsequent cyclization involves a Zinc-catalyzed activation of the sp2 C-H bond adjacent to the newly formed ring system. The addition of an oxidant, preferably Potassium Persulfate, generates radical species that facilitate the coupling between the activated C-H bond and the hydroxyl group generated in the first step. This oxidative C-H/O-H coupling effectively closes the pyran ring, aromatizing the system to yield the final pyranoindolone structure. The choice of Zinc Iodide as the catalyst is particularly advantageous due to its Lewis acidity, which coordinates with the oxygen atoms to lower the activation energy for the coupling event. This mechanism ensures high regioselectivity and chemoselectivity, tolerating a wide range of functional groups including halogens, nitriles, and esters, which is essential for the late-stage functionalization required in medicinal chemistry campaigns. 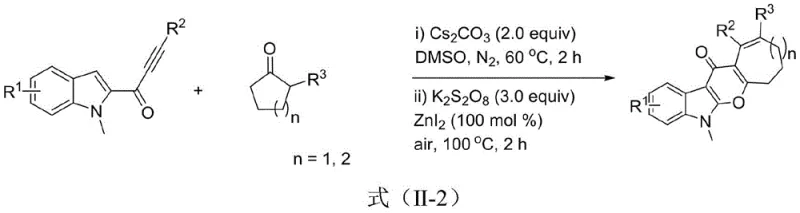
How to Synthesize Pyranoindolone Derivatives Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it highly accessible for process chemistry teams aiming to scale up production. The protocol consolidates two distinct chemical transformations into a single reactor vessel, thereby reducing solvent consumption and processing time. The reaction begins under an inert nitrogen atmosphere to protect the sensitive initial intermediates, utilizing Cesium Carbonate as the promoter in Dimethyl Sulfoxide at moderate temperatures. Once the ring expansion is complete, the reaction environment is switched to air, and the oxidant and catalyst are introduced to drive the final cyclization. This seamless transition between anaerobic and aerobic conditions within the same pot is a key feature that enhances throughput. For detailed standard operating procedures and specific stoichiometric ratios validated by experimental data, please refer to the technical guide below.
- Mix indole alkynone and cyclic ketone with Cs2CO3 in DMSO under nitrogen at 60°C for 2 hours to effect C-C bond insertion.
- Add potassium persulfate and zinc iodide catalyst to the reaction mixture without isolation.
- Heat the mixture to 100°C under air atmosphere for 2 hours to complete the C-H/O-H coupling cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers substantial strategic benefits that directly impact the bottom line and operational resilience. The elimination of precious metal catalysts such as Rhodium and Gold removes a major source of cost volatility and supply risk, as the prices of these metals are subject to significant geopolitical and market fluctuations. By substituting them with Zinc salts and Cesium Carbonate, manufacturers can secure a more stable and predictable cost structure for raw materials. Additionally, the use of commodity chemicals like cyclic ketones and indole derivatives ensures that sourcing is not bottlenecked by specialized suppliers, thereby enhancing supply chain reliability and reducing lead times for critical starting materials.
- Cost Reduction in Manufacturing: The economic impact of switching from noble metal catalysis to base-metal catalysis cannot be overstated. The removal of expensive catalysts drastically reduces the direct material costs associated with each batch. Furthermore, the one-pot nature of the synthesis eliminates the need for intermediate isolation, filtration, and drying steps, which translates to significant savings in labor, energy, and solvent usage. The simplified downstream processing also reduces the load on waste treatment facilities, contributing to overall operational efficiency and lower environmental compliance costs.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity-grade reagents ensures that production schedules are not disrupted by the scarcity of specialized precursors. Traditional methods often require custom-synthesized strained rings or complex alkynes that have long lead times and limited supplier bases. In contrast, the substrates for this method are standard catalog items or easily synthesized in-house, providing procurement managers with greater flexibility and bargaining power. This robustness is crucial for maintaining continuous supply of API intermediates, especially in the face of global logistics challenges.
- Scalability and Environmental Compliance: The process is inherently scalable due to its straightforward thermal profile and the use of common solvents like DMSO, which are well-understood in large-scale manufacturing environments. The absence of toxic heavy metals simplifies the regulatory approval process for new drug applications, as residual metal limits are easier to meet. Moreover, the use of air as an oxidant in the second step minimizes the generation of hazardous chemical waste, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the primary advantages of this synthesis over traditional methods?
A: Unlike conventional methods requiring expensive precious metals like Rhodium or Gold and strained ring precursors, this patent utilizes earth-abundant Zinc catalysts and simple cyclic ketones, significantly reducing raw material costs and simplifying purification.
Q: What is the scope of substrates for this reaction?
A: The method demonstrates excellent universality, accommodating various substituents on the indole ring (H, alkyl, halogen) and diverse electron-withdrawing groups on the cyclic ketone, yielding seven and eight-membered fused rings with yields ranging from 50% to 92%.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the one-pot nature eliminates intermediate isolation steps, and the use of air as an oxidant in the second step alongside common solvents like DMSO makes the protocol highly amenable to commercial scale-up with reduced waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyranoindolone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this zinc-catalyzed synthesis route for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. Our state-of-the-art facilities are equipped to handle the specific thermal and atmospheric requirements of this one-pot process, while our rigorous QC labs guarantee stringent purity specifications that meet the demanding standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cost-effective technology for your drug development pipeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific target molecules, demonstrating how this route can optimize your COGS. Please contact our technical procurement team today to request specific COA data for related analogues and comprehensive route feasibility assessments for your projects.
