Advanced Visible-Light Catalyzed Synthesis of 2-Cyanoalkylsulfonyl 3,4-Dihydronaphthalene Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex sulfur-containing scaffolds, which are pivotal motifs in numerous bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN111039737B, which discloses a novel synthetic strategy for preparing 2-cyanoalkylsulfonyl-substituted 3,4-dihydronaphthalene compounds. This technology leverages visible light photocatalysis to drive a sophisticated cascade reaction involving methylenecyclopropanes (MCPs), cyclobutanone oxime esters, and potassium metabisulfite (K2S2O5). Unlike traditional thermal methods that often require harsh conditions or expensive sulfur sources, this approach operates under mild parameters, achieving high yields through a unique radical mechanism that cleaves two carbon-carbon sigma bonds while simultaneously forming new carbon-carbon and carbon-sulfur bonds in a single pot.
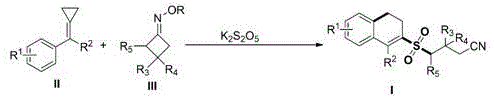
For R&D directors and process chemists, the mechanistic elegance of this transformation offers a compelling alternative to legacy sulfone synthesis routes. The reaction initiates with the visible-light-mediated homolytic cleavage of the N-O bond in the cyclobutanone oxime ester, generating an imino radical that undergoes beta-scission to release a cyanoalkyl radical. This radical species then efficiently traps sulfur dioxide released from the decomposition of K2S2O5, forming a sulfonyl radical. This highly reactive intermediate subsequently attacks the strained double bond of the methylenecyclopropane, triggering a ring-opening and cyclization sequence that constructs the dihydronaphthalene core with high regioselectivity. The ability to utilize simple, inexpensive inorganic salts as the sulfur source, rather than specialized gas surrogates, represents a substantial advancement in process safety and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of sulfone functionalities has relied heavily on the insertion of sulfur dioxide using reagents such as DABSO (DABCO·(SO2)2), sodium sulfite, or thiourea dioxide. While effective in certain contexts, these traditional methods often suffer from significant drawbacks when applied to complex scaffold synthesis. For instance, the use of DABSO can be cost-prohibitive for large-scale manufacturing due to the expense of the amine-sulfur dioxide adduct. Furthermore, many classical approaches require stoichiometric amounts of strong bases or transition metal catalysts that necessitate rigorous removal steps to meet pharmaceutical purity standards. In the specific context of synthesizing fused ring systems like dihydronaphthalenes, conventional radical cyclizations often struggle with competing side reactions or require high temperatures that degrade sensitive functional groups, leading to lower overall yields and difficult purification profiles.
The Novel Approach
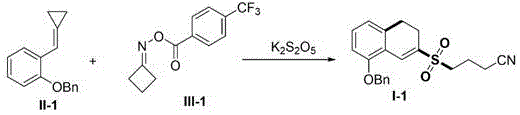
The methodology described in patent CN111039737B overcomes these hurdles by integrating visible light photocatalysis with a dual C-C bond cleavage strategy. By employing K2S2O5 as a solid, easy-to-handle sulfur dioxide source, the process eliminates the need for handling gaseous SO2 or expensive surrogates. Experimental data from the patent highlights the superiority of this system; when K2S2O5 was replaced with DABSO in model reactions, the yield plummeted from 81% to merely 25%. Similarly, substituting with thiourea dioxide only achieved a 58% yield. This novel approach not only enhances reaction efficiency but also broadens the substrate scope, tolerating a wide array of electronic environments on the aromatic rings, including halogens, nitro groups, and trifluoromethyl substituents, making it a versatile tool for medicinal chemistry campaigns.
Mechanistic Insights into Visible-Light Catalyzed Radical Cascade
The core of this synthetic innovation lies in the precise orchestration of radical species under visible light irradiation. The photocatalyst, typically Ru(bpy)3Cl2 or Ir(ppy)3, absorbs photons to reach an excited state, facilitating the single-electron transfer (SET) or energy transfer required to activate the cyclobutanone oxime ester. This activation triggers the fragmentation of the four-membered ring, a process driven by the relief of ring strain, which releases the critical cyanoalkyl radical. This radical acts as the linchpin of the reaction, first capturing the transient SO2 to form a sulfonyl radical, and then engaging the methylenecyclopropane. The subsequent ring-opening of the cyclopropane moiety is thermodynamically favorable and directs the formation of the six-membered dihydronaphthalene ring. This cascade effectively stitches together three distinct components into a complex architecture with high atom economy, minimizing waste generation.
From an impurity control perspective, the mildness of the reaction conditions (60-100°C) is paramount. High-temperature thermal radical reactions often lead to polymerization or non-selective radical coupling, creating complex impurity profiles that are difficult to separate. In contrast, the photochemical pathway described here proceeds with high selectivity, as evidenced by the clean NMR spectra and high isolated yields reported across dozens of examples in the patent. The use of 2,6-lutidine as a mild organic base further ensures that acid-sensitive groups remain intact, preserving the integrity of diverse functional handles needed for downstream derivatization in drug discovery pipelines.
How to Synthesize 2-Cyanoalkylsulfonyl 3,4-Dihydronaphthalene Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and reproducibility. The protocol involves a one-pot procedure where all reagents are combined in a polar aprotic solvent, preferably acetonitrile, under an inert atmosphere. The reaction is driven by blue LED irradiation, which provides the necessary energy to sustain the catalytic cycle without overheating the mixture. Detailed standard operating procedures regarding stoichiometry, light intensity, and workup protocols are critical for success.
- Charge a Schlenk sealed tube reactor with methylenecyclopropane compound (Formula II), cyclobutanone oxime ester (Formula III), K2S2O5, Ru(bpy)3Cl2 photocatalyst, 2,6-lutidine base, and acetonitrile solvent.
- Maintain an inert argon atmosphere and irradiate the mixture with a 5W blue LED lamp while heating to 80°C with stirring for approximately 18 hours.
- Upon completion monitored by TLC, wash the mixture with brine, extract with ethyl acetate, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic methodology offers tangible strategic benefits beyond mere chemical novelty. The shift towards using commodity chemicals like potassium metabisulfite instead of specialized sulfur surrogates directly impacts the bill of materials, potentially lowering the raw material costs significantly. Furthermore, the operational simplicity of the process—requiring only standard glassware, LED lights, and moderate heating—reduces the capital expenditure associated with specialized high-pressure or cryogenic reactors often needed for traditional sulfone synthesis.
- Cost Reduction in Manufacturing: The elimination of expensive sulfur dioxide surrogates like DABSO is a primary driver for cost optimization. By utilizing K2S2O5, a widely available and inexpensive industrial chemical, manufacturers can drastically reduce the cost of goods sold (COGS). Additionally, the high efficiency of the reaction minimizes solvent usage and waste disposal costs associated with low-yielding processes. The avoidance of transition metal catalysts in high loadings, or the ability to use earth-abundant alternatives like Eosin Y in some variations, further contributes to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials ensures a stable supply chain. Methylenecyclopropanes and cyclobutanone oxime esters can be sourced from multiple vendors or synthesized via established routes, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by the need for specialized reagents that may have long lead times or regulatory restrictions, ensuring consistent delivery of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The mild reaction temperatures and the use of common organic solvents facilitate straightforward scale-up from gram to kilogram scales. The process generates minimal hazardous waste compared to methods using toxic heavy metals or corrosive gases. This aligns with modern green chemistry principles, simplifying environmental compliance and reducing the burden on wastewater treatment facilities, which is a critical consideration for sustainable manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light catalyzed synthesis. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear picture of the technology's capabilities and limitations for potential partners.
Q: What are the key advantages of using K2S2O5 over traditional sulfur dioxide sources like DABSO?
A: According to patent CN111039737B, using K2S2O5 as the sulfur dioxide source significantly improves reaction efficiency compared to DABSO or thiourea dioxide. In optimization trials, K2S2O5 yielded 81% of the target product, whereas DABSO only achieved 25% and thiourea dioxide reached 58%, demonstrating superior cost-effectiveness and atom economy for large-scale production.
Q: Is this photocatalytic method scalable for industrial manufacturing?
A: Yes, the process utilizes mild reaction conditions (60-100°C) and commercially available reagents like acetonitrile and inexpensive inorganic salts. The absence of harsh reagents and the use of standard LED light sources make the transition from laboratory scale to commercial tonnage feasible without requiring specialized high-pressure equipment.
Q: What is the substrate scope for this synthesis method?
A: The method exhibits broad substrate tolerance. It accommodates various substituents on the aromatic rings of both the methylenecyclopropane and the oxime ester, including electron-withdrawing groups like halogens, nitro, and cyano groups, as well as electron-donating groups like alkyl and alkoxy chains, ensuring versatility for diverse pharmaceutical intermediate libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyanoalkylsulfonyl 3,4-Dihydronaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic methods in modern drug synthesis. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the one described in CN111039737B can be seamlessly translated into industrial reality. Our facility is equipped with state-of-the-art photochemical reactors and stringent purity specifications managed by rigorous QC labs, guaranteeing that every batch of 2-cyanoalkylsulfonyl 3,4-dihydronaphthalene intermediate meets the highest global quality standards.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this efficient synthetic route for their pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring your supply chain is optimized for both performance and economic efficiency.
