Advanced Synthesis of Nitrobenzene Compounds: A Green Alternative for Commercial Scale-up
Introduction to Patent CN102180794B
The chemical industry is constantly seeking safer, more sustainable pathways for producing fundamental aromatic building blocks, and Patent CN102180794B presents a significant breakthrough in the synthesis of nitrobenzene compounds. This intellectual property outlines a novel methodology that replaces the hazardous, traditional electrophilic aromatic substitution with a mild, cuprous-catalyzed substitution reaction using arylboronic acids and metal nitrites. By utilizing water as the primary solvent and inexpensive cuprous oxide as the catalyst, this process addresses critical pain points regarding environmental pollution, operational safety, and functional group compatibility that have long plagued the manufacturing of nitro-aromatics. For R&D directors and procurement specialists alike, this technology represents a paradigm shift towards greener chemistry without compromising on the scalability or yield required for industrial applications.
Furthermore, the versatility of this synthetic route allows for the introduction of the nitro group onto aromatic rings bearing a wide array of substituents, including alkyl, halogen, hydroxyl, and even sensitive aldehyde groups, which are typically incompatible with strong oxidizing nitrating agents. The patent explicitly details the reaction conditions, emphasizing operation at near-room temperature (10-30°C) and atmospheric pressure, which drastically reduces the energy footprint and equipment stress compared to conventional high-temperature nitration processes. This document serves as a comprehensive technical analysis of how this specific innovation can be leveraged to optimize supply chains for pharmaceutical intermediates and fine chemicals, ensuring a reliable source of high-purity materials while adhering to increasingly stringent global environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of nitrobenzene and its derivatives has relied heavily on direct nitration using a mixture of concentrated nitric acid and sulfuric acid, a process known for its severe operational hazards and environmental drawbacks. As illustrated in the background art of the patent, this classical electrophilic aromatic substitution often leads to uncontrollable side reactions, such as over-nitration to form dinitrobenzenes, which complicates downstream purification and significantly lowers the overall yield of the desired mono-nitro product. 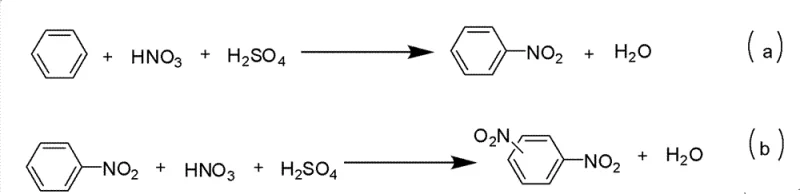
Beyond the issue of selectivity, the conventional mixed-acid method imposes extreme conditions involving high temperatures and strong corrosive environments that necessitate specialized, expensive reactor materials and rigorous safety protocols to prevent runaway exothermic reactions. Moreover, the generation of large volumes of acidic waste streams creates a substantial burden on wastewater treatment facilities, driving up the operational costs associated with environmental compliance and disposal. Perhaps most critically for synthetic chemists, the harsh oxidative nature of the nitrating mixture severely limits the scope of substrates, often destroying or modifying sensitive functional groups like amines, alcohols, or aldehydes present on the aromatic ring, thereby restricting its utility in the synthesis of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in Patent CN102180794B introduces a transformative approach that utilizes arylboronic acids as starting materials, reacting them with metal nitrites in the presence of a cuprous catalyst and ammonia water. This substitution strategy operates under remarkably mild conditions, typically between 10°C and 30°C, effectively eliminating the thermal risks and energy consumption associated with traditional nitration. The use of water as the solvent not only aligns with green chemistry principles by removing volatile organic compounds (VOCs) from the process but also simplifies the workup procedure, as the organic product can be easily extracted while the inorganic salts remain in the aqueous phase.
The structural scope of this new method is exceptionally broad, accommodating various substituents on the aromatic ring as defined by general formulas (I) and (II) in the patent documentation. 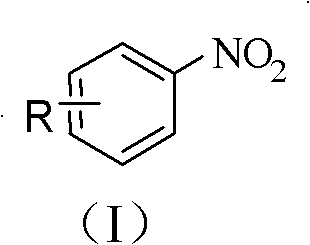
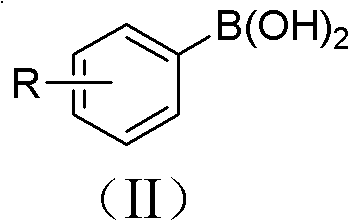
This high functional group tolerance means that manufacturers can synthesize complex nitro-aromatics directly without the need for extensive protecting group strategies, streamlining the synthetic route and reducing the number of unit operations. The ability to tolerate groups such as -OH, -CHO, and -COOH opens up new possibilities for the direct synthesis of advanced intermediates for drugs and agrochemicals that were previously difficult or impossible to access via direct nitration.
Mechanistic Insights into Cuprous-Catalyzed Substitution
The core of this innovation lies in the unique catalytic system comprising a cuprous species, specifically cuprous oxide (Cu2O), and ammoniacal liquor, which facilitates the substitution of the boronic acid group with a nitro group from the metal nitrite source. While the exact mechanistic pathway involves complex coordination chemistry, it is understood that the ammonia acts as a crucial ligand or auxiliary agent that enhances the solubility and reactivity of the copper catalyst in the aqueous medium. This interaction likely promotes the formation of an active copper-nitrite complex that undergoes transmetallation with the arylboronic acid, followed by reductive elimination to forge the C-N bond, thereby installing the nitro functionality with high regioselectivity.
From an impurity control perspective, this mechanism offers a distinct advantage by avoiding the formation of nitronium ions (NO2+), the aggressive electrophiles responsible for the indiscriminate poly-nitration seen in mixed-acid processes. Instead, the copper-mediated pathway proceeds through a controlled substitution mechanism that inherently favors mono-substitution, significantly reducing the formation of dinitro-impurities and simplifying the purification profile. The patent data confirms that this selectivity is robust across a range of substrates, allowing for the isolation of high-purity products with minimal chromatographic effort, which is a critical factor for scaling up processes for GMP-grade pharmaceutical ingredients.
How to Synthesize Nitrobenzene Compounds Efficiently
The practical implementation of this synthesis is straightforward and designed for adaptability in both laboratory and pilot-plant settings. The general protocol involves charging a reaction vessel with the arylboronic acid substrate, the cuprous catalyst, ammoniacal liquor, and the metal nitrite salt in water, followed by stirring at ambient temperature for a period ranging from 3 to 48 hours depending on the electronic nature of the substrate. Detailed standardized operating procedures, including precise molar ratios and specific workup instructions for various derivatives, are essential for reproducibility and quality control.
- Prepare the reaction mixture by combining the arylboronic acid compound, a cuprous catalyst (preferably cuprous oxide), ammoniacal liquor, and a metal nitrite (MNO2 where M is Li, Na, K, Rb, or Cs) in water.
- Maintain the reaction temperature between 10°C and 30°C under atmospheric pressure and allow the substitution reaction to proceed for 3 to 48 hours depending on the substrate.
- Upon completion, extract the product using ethyl acetate, wash the organic phase with alkali or acid as needed, and purify via chromatography or concentration to obtain the high-purity nitrobenzene compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cuprous-catalyzed synthesis offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The shift from hazardous mixed acids to benign aqueous systems fundamentally alters the cost structure of production by eliminating the need for expensive corrosion-resistant equipment and reducing the capital expenditure required for safety infrastructure. Furthermore, the use of water as a solvent removes the costs associated with purchasing, recovering, and disposing of large volumes of organic solvents, leading to a significantly reduced environmental footprint and lower waste treatment fees.
- Cost Reduction in Manufacturing: The utilization of cuprous oxide, an abundant and inexpensive base metal catalyst, replaces the need for precious metal catalysts often required in other cross-coupling methodologies, resulting in substantial raw material cost savings. Additionally, the mild reaction conditions operate at near-room temperature, which drastically cuts down on energy consumption for heating and cooling compared to the high-temperature requirements of traditional nitration. The ability to recycle the catalytic system, as demonstrated in the patent where the aqueous phase was reused multiple times with maintained activity, further amplifies these cost efficiencies by minimizing catalyst turnover and waste generation.
- Enhanced Supply Chain Reliability: The starting materials for this process, specifically arylboronic acids and metal nitrites, are commodity chemicals that are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction against moisture and oxygen (as it can be performed in air) simplifies logistics and storage requirements, ensuring that production schedules are not disrupted by the need for inert atmosphere handling or specialized anhydrous reagents. This reliability translates directly into more predictable lead times and a more resilient supply chain for critical nitrobenzene intermediates.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the absence of exothermic runaway risks, allowing for larger batch sizes without proportional increases in safety engineering controls. The aqueous nature of the waste stream simplifies effluent treatment, making it easier for manufacturing sites to meet strict local and international environmental discharge standards. This compliance advantage future-proofs the supply chain against tightening regulations, ensuring continuous operation without the threat of regulatory shutdowns or fines associated with hazardous chemical processing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrobenzene synthesis technology, derived directly from the experimental data and specifications within Patent CN102180794B. These insights are intended to clarify the operational parameters and potential advantages for stakeholders evaluating this method for their own production portfolios.
Q: What are the primary advantages of this cuprous-catalyzed method over traditional nitration?
A: Unlike traditional mixed-acid nitration which requires harsh conditions and generates significant waste, this method uses water as a solvent, operates at mild temperatures (10-30°C), and exhibits high tolerance for sensitive functional groups like aldehydes and alcohols.
Q: Can the catalyst system be recycled to reduce production costs?
A: Yes, the patent demonstrates that after extracting the product, the remaining aqueous reaction system can be reused for subsequent batches by adding fresh substrates and alkali salts, maintaining consistent yields around 54-55%.
Q: Which cuprous catalyst provides the highest yield for nitrobenzene synthesis?
A: Experimental data indicates that cuprous oxide (Cu2O) is the most effective catalyst, achieving a 60% yield for nitrobenzene, whereas cuprous halides like CuI, CuBr, and CuCl resulted in lower yields of 36%, 30%, and 30% respectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrobenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic routes described in Patent CN102180794B and possess the technical expertise to bring this green chemistry innovation to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of nitrobenzene compounds meets the exacting standards required by the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and product quality.
