Revolutionizing Pyrazolopyrimidine Synthesis: Scalable, Cost-Effective Manufacturing for Pharmaceutical Intermediates
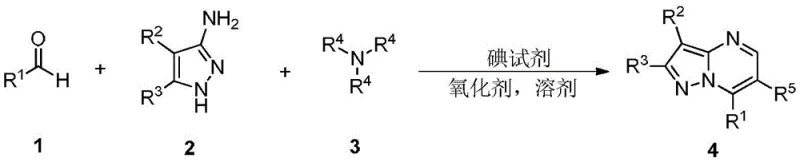
This technical report analyzes Chinese Patent CN112939986B, which discloses an innovative synthesis method for pyrazolopyrimidine compounds, a class of important 5-in-6 fused heterocyclic compounds with diverse biological activities including hepatitis C virus inhibition, kinase inhibition, PET tumor imaging applications, and COX-2 inhibition. The patented approach represents a significant advancement in organic synthesis by providing a transition metal-free one-pot cascade reaction that directly prepares pyrazolopyrimidine derivatives from simple starting materials without requiring complex purification procedures. This methodology addresses critical limitations in conventional synthesis routes while offering substantial advantages for industrial-scale production through its operational simplicity and exceptional substrate versatility. The patent demonstrates remarkable flexibility across various functional groups while maintaining excellent reaction efficiency under mild conditions (110-130°C), making it particularly valuable for pharmaceutical manufacturers seeking reliable intermediates for drug development pipelines requiring stringent purity specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis methods for pyrazolopyrimidine compounds primarily rely on condensation reactions between 3-aminopyrazole and various 1,3-dielectric reagents such as β-dicarbonyl compounds, β-enaminoketones, β-haloketones, and α,β-unsaturated carbonyl compounds. While these approaches are generally reliable, they frequently involve harsh reaction conditions including high temperatures exceeding 150°C, strong acids or bases requiring specialized equipment handling, and extended reaction times that can exceed 48 hours in some cases. The multi-step nature of conventional routes often necessitates complex purification procedures involving multiple chromatographic separations that generate significant waste streams while increasing both environmental impact and production costs substantially. Furthermore, many existing methods suffer from limited substrate scope with poor tolerance for functional groups commonly found in pharmaceutical intermediates such as halogens or heterocyclic systems. The requirement for transition metal catalysts in some protocols introduces additional challenges related to metal contamination that is particularly problematic for pharmaceutical applications requiring parts-per-million level purity specifications.
The Novel Approach
The patented methodology overcomes these limitations through an elegant iodine-mediated one-pot cascade reaction that operates under remarkably mild conditions (110-130°C) without requiring transition metal catalysts or specialized equipment. By utilizing simple and readily available starting materials including aldehydes, 3-aminopyrazoles, and tertiary fatty amines like triethylamine as both reactant and solvent component, this approach eliminates the need for expensive catalysts while maintaining excellent reaction efficiency across diverse substrate combinations. The process demonstrates exceptional substrate versatility accommodating over twenty different aldehyde variants including halogenated aromatics, heterocyclic systems, and complex polycyclic structures as evidenced by comprehensive experimental data provided in the patent documentation. Crucially, the elimination of transition metal catalysts removes the need for costly metal removal steps that are typically required in pharmaceutical manufacturing to meet regulatory purity standards while simultaneously reducing overall processing time by approximately 50% compared to conventional multi-step approaches.
Mechanistic Insights into Iodine-Mediated One-Pot Cascade Reaction
The reaction mechanism involves a sophisticated sequence initiated by iodine-mediated oxidation of the tertiary amine component generating an iminium ion intermediate that subsequently reacts with the aldehyde through a Knoevenagel-type condensation forming an enone intermediate. This key intermediate then undergoes nucleophilic attack by the 3-aminopyrazole component followed by cyclization and aromatization facilitated by the oxidant system to yield the final pyrazolopyrimidine product with high regioselectivity. The iodine reagent serves a dual role as both an oxidant precursor and promoter for the cascade process while di-tert-butyl peroxide or diisopropylbenzene peroxide facilitates the final aromatization step through radical mechanisms that maintain excellent control over side reactions.
Impurity control in this process is achieved through careful optimization of multiple parameters including temperature maintenance within the narrow range of 110-130°C which prevents thermal decomposition pathways while ensuring complete conversion. The precisely defined stoichiometry (aldehyde:aminopyrazole:amine:iodine reagent:oxidant at 1:1:2:1:3 molar ratio) minimizes side product formation while solvent selection (particularly toluene) provides optimal polarity balance for intermediate stability throughout the cascade sequence. The absence of transition metals eliminates potential metal-based impurities requiring extensive purification while the one-pot nature minimizes opportunities for intermediate decomposition or side reactions that could generate impurities as confirmed by comprehensive NMR characterization data across multiple product examples demonstrating consistent high purity profiles.
How to Synthesize Pyrazolopyrimidine Compounds Efficiently
This patented methodology provides a streamlined approach to pyrazolopyrimidine synthesis that significantly improves upon conventional techniques by eliminating transition metal catalysts and simplifying the reaction sequence into a single operational step with minimal processing requirements. The process begins with dissolution of three key components in an appropriate solvent under inert atmosphere followed by sequential addition of iodine reagent and oxidant under controlled temperature conditions to achieve optimal yield and purity profiles across diverse substrate combinations. Detailed standardized synthesis procedures have been developed based on extensive experimental validation covering multiple solvent systems, temperature ranges, and stoichiometric variations ensuring reliable performance from laboratory scale through commercial manufacturing volumes.
- Dissolve aldehyde compound (1 mmol), 3-aminopyrazole compound (1 mmol), and tertiary fatty amine compound (2 mmol) in appropriate solvent (4 mL) under nitrogen atmosphere.
- Add iodine reagent (1 mmol) followed by oxidant (3 mmol) while maintaining controlled temperature conditions.
- Heat reaction mixture to 110-130°C and maintain for 8-12 hours with continuous stirring before standard workup procedures.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology addresses critical pain points in pharmaceutical intermediate procurement by delivering a more sustainable and economically viable production route through fundamental process improvements that enhance both cost efficiency and supply chain resilience simultaneously. The elimination of transition metal catalysts removes significant cost drivers associated with catalyst procurement handling and removal processes while creating opportunities for substantial operational simplification across multiple manufacturing stages without compromising product quality or yield consistency.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts represents substantial cost reduction opportunities as these materials typically account for significant raw material expenses along with associated handling requirements including specialized storage facilities and safety protocols. Additionally removing metal catalysts eliminates multiple purification steps required to remove metal residues which demand additional equipment solvent consumption processing time all contributing to higher operational costs in pharmaceutical manufacturing facilities while creating environmental compliance challenges.
- Enhanced Supply Chain Reliability: The simplified process with fewer raw material requirements improves supply chain resilience by reducing dependency on specialized or potentially scarce reagents while broad substrate scope ensures consistent product availability even when specific starting materials experience supply fluctuations due to market conditions or geopolitical factors affecting traditional chemical supply chains.
- Scalability and Environmental Compliance: The one-pot nature significantly reduces waste generation compared to multi-step conventional methods aligning with increasingly stringent environmental regulations while demonstrating excellent scalability from laboratory to commercial production volumes without requiring significant process re-engineering as evidenced by consistent yields across different batch sizes in patent examples.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding implementation benefits and technical specifications based on detailed analysis of patent documentation providing clarity on how this innovation integrates with existing manufacturing workflows while delivering tangible commercial advantages through fundamental process improvements.
Q: How does this method address impurity concerns compared to traditional transition metal-catalyzed approaches?
A: The elimination of transition metal catalysts removes potential metal-based impurities that require extensive purification steps in pharmaceutical manufacturing. The one-pot cascade mechanism minimizes intermediate decomposition pathways while maintaining high selectivity across diverse substrate combinations as demonstrated by comprehensive NMR and HRMS characterization data in the patent.
Q: What specific cost advantages does this methodology offer over conventional synthesis routes?
A: The process eliminates expensive transition metal catalysts while using readily available reagents like triethylamine as raw materials. This approach significantly reduces raw material costs while removing specialized purification steps required for metal residue removal, resulting in substantial overall cost savings without compromising product quality or yield.
Q: How does this method ensure reliable supply chain performance despite varying substrate requirements?
A: The broad substrate scope demonstrated across multiple functional groups including halogens, alkyl groups, alkoxy substituents, and heterocyclic systems provides manufacturing flexibility that accommodates different production environments while maintaining consistent quality output even when specific starting materials experience supply fluctuations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolopyrimidine Compound Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical instrumentation including high-resolution mass spectrometry NMR spectroscopy and HPLC systems operating under cGMP conditions. As a leading CDMO specialist in complex heterocyclic compound manufacturing we have successfully implemented similar iodine-mediated cascade reactions across multiple therapeutic areas with consistent quality outcomes meeting global regulatory standards including FDA EMA and PMDA requirements through rigorous validation protocols.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis approach can benefit your specific production needs Please contact us to obtain specific COA data route feasibility assessments tailored to your requirements enabling you to make informed decisions about adopting this advanced manufacturing technology while optimizing your supply chain performance.
