Advanced Microwave-Assisted Synthesis of N-Methyl-2-Cyano-3,4-Disubstituted Pyrrole Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, particularly pyrrole derivatives, which serve as critical scaffolds in anticancer, antibacterial, and antitumor agents. A significant technological breakthrough in this domain is detailed in patent CN110713451B, which discloses a novel microwave radiation-assisted synthesis method for N-methyl-2-cyano-3,4-disubstituted pyrrole compounds. This innovation represents a paradigm shift from traditional multi-step, transition-metal-dependent protocols to a streamlined, one-pot process driven by sodium iodide catalysis. By leveraging the unique dielectric heating properties of microwave radiation, this method achieves exceptional reaction efficiency and atom economy, directly addressing the pressing needs of modern pharmaceutical intermediate manufacturing for speed and purity. The ability to synthesize these complex heterocycles without the burden of toxic oxidants or expensive transition metals positions this technology as a highly attractive route for scalable production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of N-methyl-2-cyano-3,4-disubstituted pyrroles was fraught with significant operational and economic challenges that hindered large-scale adoption. Conventional techniques, such as those reported in literature (e.g., Org. Lett. 2016), typically relied on copper triflate salts as catalysts and required stoichiometric amounts of hazardous oxidants like dichloro dicyano benzoquinone (DDQ). These processes were not only costly due to the price of reagents but also generated substantial chemical waste, complicating environmental compliance and waste disposal logistics. Furthermore, the reaction conditions were often harsh, requiring prolonged heating times of up to 24 hours under inert atmospheres, which severely impacted throughput. Perhaps most critically for process chemists, the resulting crude mixtures frequently necessitated tedious chromatographic purification to remove metal residues and byproducts, leading to poor overall yields often hovering around 44% and inconsistent product quality.
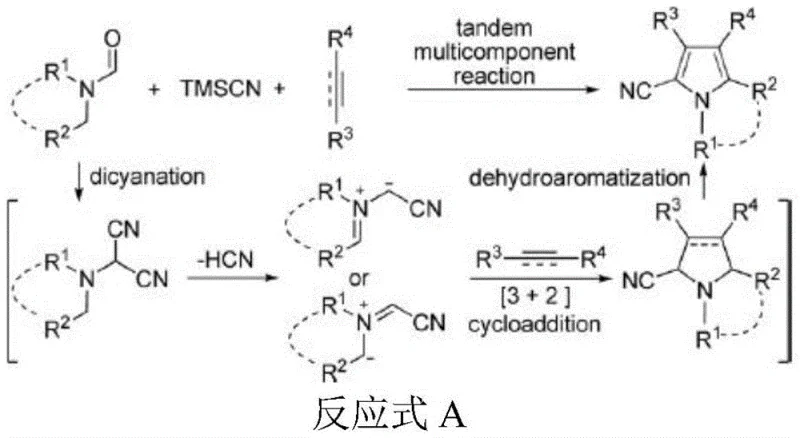
The Novel Approach
In stark contrast, the methodology outlined in patent CN110713451B introduces a green chemistry approach that eliminates the need for transition metals and stoichiometric oxidants entirely. By utilizing sodium iodide as a cheap and abundant catalyst in conjunction with microwave irradiation, the reaction proceeds under solvent-free conditions, drastically reducing the volume of organic solvents required and simplifying the workup procedure. This novel route transforms the synthesis into a highly efficient one-pot transformation where alkynes, trimethylsilyl cyanide (TMSCN), and N,N-dimethylformamide (DMF) converge rapidly. The elimination of chromatographic purification steps is a major commercial advantage, as the product can often be isolated through simple extraction and concentration. This shift not only enhances the safety profile of the manufacturing process by removing toxic reagents but also significantly improves the overall process mass intensity (PMI), making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Sodium Iodide-Catalyzed Cycloaddition
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the sodium iodide catalyst under microwave conditions. Unlike the complex oxidative mechanisms of copper-catalyzed routes, this process operates through a direct intermolecular [3+2] cycloaddition. The sodium iodide activates the trimethylsilyl cyanide and DMF to generate a reactive nitrilium intermediate (IM1) in situ. This intermediate then undergoes a concerted cycloaddition with the alkyne substrate to form the pyrrole ring directly. The microwave radiation plays a dual role here: it provides rapid, uniform heating that accelerates the formation of the intermediate and lowers the activation energy for the cycloaddition step. This synergy allows the reaction to reach completion in merely 10 minutes, a fraction of the time required by thermal methods, while maintaining high selectivity for the 2-cyano-3,4-disubstituted isomer.
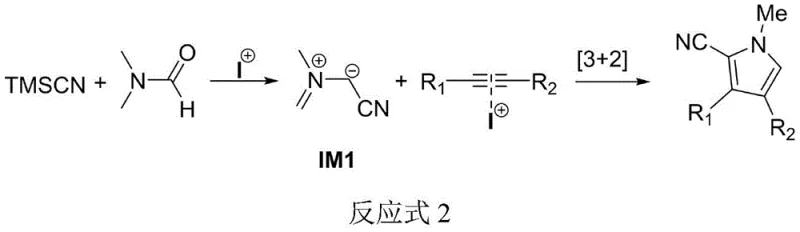
From an impurity control perspective, this mechanism offers superior cleanliness compared to oxidative methods. The absence of strong oxidants like DDQ prevents the formation of over-oxidized byproducts or polymerization species that often plague pyrrole syntheses. Additionally, the specific activation of the nitrile source by iodide ions ensures that the cyano group is incorporated efficiently at the 2-position of the pyrrole ring, minimizing regio-isomeric impurities. For R&D directors focused on purity profiles, this means a simpler impurity spectrum that is easier to characterize and control during scale-up. The high atom efficiency of the reaction ensures that the majority of the starting material mass is incorporated into the final product, reducing the load on downstream purification units and ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications with minimal effort.
How to Synthesize N-Methyl-2-Cyano-3,4-Disubstituted Pyrrole Efficiently
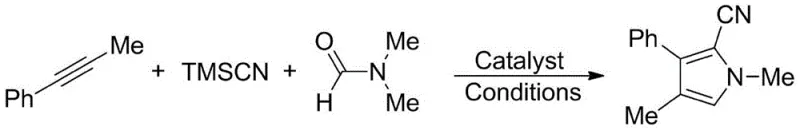
Implementing this synthesis protocol requires precise control over reaction parameters to maximize the benefits of the microwave-assisted system. The general procedure involves charging a reaction vessel with equimolar amounts of the alkyne substrate, trimethylsilyl cyanide, and DMF, followed by the addition of 10 mol% sodium iodide. The mixture is then subjected to microwave irradiation at a specific power setting (optimized at 300W) and maintained at 100°C for a short duration of 10 minutes. This rapid protocol contrasts sharply with the day-long reactions of the past. Following the reaction, the workup is remarkably straightforward: the crude mixture is dissolved in ethyl acetate, washed with saturated brine to remove inorganic salts, and concentrated under vacuum. This simplicity underscores the practical utility of the method for both laboratory screening and pilot plant operations.
- Charge a reaction vessel with equimolar amounts of alkyne, trimethylsilyl cyanide (TMSCN), and N,N-dimethylformamide (DMF), along with 10 mol% sodium iodide catalyst.
- Subject the mixture to microwave radiation at 300W power and maintain a temperature of 100°C for approximately 10 minutes to drive the [3+2] cycloaddition.
- Upon completion, dissolve the crude product in ethyl acetate, wash with saturated saline solution, separate the organic layer, and concentrate under vacuum to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this microwave-assisted synthesis offers tangible strategic advantages that extend beyond mere chemical yield. The primary benefit is the drastic simplification of the supply chain for raw materials. By replacing expensive and potentially supply-constrained transition metal catalysts like copper triflate with commodity-grade sodium iodide, manufacturers can secure a more stable and cost-effective supply base. Furthermore, the solvent-free nature of the reaction reduces the procurement volume of organic solvents, lowering both material costs and the logistical burden of solvent storage and recovery. This aligns perfectly with global initiatives to reduce the carbon footprint of chemical manufacturing, offering a competitive edge in markets where sustainability credentials are increasingly valued by end-users.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the elimination of costly reagents and the reduction of processing time. By removing the need for stoichiometric oxidants like DDQ and expensive copper catalysts, the direct material cost per kilogram of product is significantly lowered. Additionally, the reduction of reaction time from 24 hours to just 10 minutes dramatically increases equipment turnover rates, allowing existing reactor capacity to produce substantially more product over the same period. The avoidance of chromatographic purification further reduces operational expenditures by saving on silica gel, solvents, and labor hours associated with column chromatography, leading to substantial overall cost savings.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for uninterrupted drug production. This method enhances reliability by utilizing robust, non-sensitive reagents that are widely available in the global chemical market. Sodium iodide and DMF are commodity chemicals with stable pricing and abundant supply, unlike specialized organometallic catalysts which can be subject to geopolitical supply shocks. Moreover, the simplicity of the workup procedure reduces the risk of batch failures due to complex purification errors, ensuring a consistent and reliable output of material that meets quality standards, thereby securing the continuity of the downstream supply chain.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this microwave-assisted route is inherently scalable, particularly with the advent of continuous flow microwave reactors. The solvent-free design minimizes the generation of hazardous waste streams, simplifying compliance with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions and wastewater treatment. The high atom efficiency means less waste is generated per unit of product, reducing disposal costs and environmental liability. This green profile facilitates smoother regulatory approvals and supports the long-term sustainability goals of modern chemical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity on catalyst selection, reaction optimization, and substrate scope. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows.
Q: Why is Sodium Iodide preferred over Copper catalysts for this pyrrole synthesis?
A: Sodium iodide offers a significantly more cost-effective and environmentally benign alternative to copper triflate salts. Unlike copper systems which require stoichiometric oxidants like DDQ and generate heavy metal waste, the NaI catalytic system operates under solvent-free conditions with high atom efficiency, eliminating the need for complex chromatographic purification and reducing downstream processing costs.
Q: What are the optimal microwave conditions for maximizing yield in this reaction?
A: Experimental data indicates that a microwave power of 300W at a temperature of 100°C for 10 minutes provides the optimal balance between reaction rate and product selectivity. Lower power settings result in incomplete conversion, while higher temperatures or excessive power can lead to side reactions and reduced yields, making precise control essential for industrial reproducibility.
Q: Can this method accommodate disubstituted alkynes with low reactivity?
A: Yes, the innovative combination of sodium iodide catalysis and microwave assistance specifically addresses the low reactivity often observed with disubstituted alkynes. The enhanced energy transfer from microwave radiation activates the substrates effectively, allowing for high conversion rates even with sterically hindered or electron-deficient alkyne derivatives that typically struggle in conventional thermal processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methyl-2-Cyano-3,4-Disubstituted Pyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the microwave-assisted synthesis described in patent CN110713451B for the production of high-value pyrrole intermediates. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into robust commercial manufacturing processes. Our facilities are equipped with advanced microwave synthesis capabilities and rigorous QC labs, ensuring that we can deliver extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to maintaining stringent purity specifications for all our outputs, leveraging our deep understanding of the NaI-catalyzed mechanism to minimize impurities and maximize yield consistency for our global clientele.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain for critical pyrrole intermediates is optimized for efficiency, cost, and reliability in the competitive global market.
