Revolutionizing Acetone Monoarylation with High-Efficiency Carbazolyl Phosphine Ligands for Commercial Scale-Up
Revolutionizing Acetone Monoarylation with High-Efficiency Carbazolyl Phosphine Ligands for Commercial Scale-Up
The landscape of transition metal-catalyzed cross-coupling reactions is undergoing a significant transformation driven by the urgent need for more efficient and cost-effective catalytic systems in the fine chemical industry. Patent CN106588983B introduces a groundbreaking class of carbazolyl phosphorus ligands that address critical bottlenecks in the alpha-monoarylation of carbonyl compounds, particularly acetone. This technology represents a paradigm shift for manufacturers of pharmaceutical intermediates, offering a pathway to construct sp2-sp3 carbon-carbon bonds with unprecedented selectivity and efficiency. By leveraging the unique electronic and steric properties of the carbazole scaffold, these novel ligands facilitate reactions under milder conditions while drastically reducing the required loading of expensive palladium catalysts. For R&D directors and process chemists, this innovation opens new avenues for synthesizing complex molecular architectures that were previously economically unviable due to polyarylation side reactions or excessive catalyst costs.
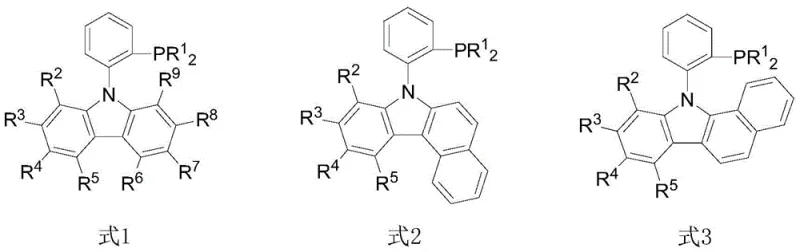
The strategic importance of this patent lies in its ability to solve the longstanding challenge of selective monoarylation using acetone as both reactant and solvent. Traditional methods often struggle with the high reactivity of acetone's six equivalent C-H bonds, leading to uncontrollable polyarylation and difficult purification processes. The carbazolyl phosphorus ligands described herein, represented by Formula 1, Formula 2, and Formula 3, provide a tailored coordination environment that stabilizes the palladium center and modulates its reactivity. This ensures that the reaction stops selectively at the monoarylated stage, delivering high-purity products essential for downstream applications in drug discovery and agrochemical development. As a reliable pharmaceutical intermediates supplier, understanding and adopting such advanced ligand technologies is crucial for maintaining competitiveness in a market that demands both purity and cost efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alpha-monoarylation of acetone has been plagued by significant technical hurdles that hinder its widespread industrial adoption. Conventional catalytic systems typically require high loadings of phosphine ligands, often ranging from 2 mol% to 5 mol%, which not only inflates the raw material costs but also complicates the removal of phosphine oxide byproducts from the final API intermediate. Furthermore, many existing ligands are structurally complex and expensive to synthesize, creating a supply chain vulnerability for large-scale manufacturing operations. The tendency of acetone to undergo multiple arylation events results in a messy impurity profile, necessitating resource-intensive purification steps such as repeated column chromatography or crystallization. These inefficiencies translate directly into longer lead times and higher production costs, making the commercial scale-up of complex pharmaceutical intermediates via this route economically challenging for many organizations.
The Novel Approach
The novel approach detailed in patent CN106588983B overcomes these deficiencies through the rational design of carbazolyl-based phosphine ligands that offer superior catalytic performance at remarkably low concentrations. By utilizing a rigid carbazole backbone substituted with bulky groups like tert-butyl or cyclohexyl, the new ligands create a protective pocket around the palladium active site. This steric environment kinetically favors the formation of the monoarylated product while suppressing further reaction, effectively solving the selectivity issue. Moreover, the synthesis of these ligands is streamlined, involving straightforward copper-catalyzed coupling followed by lithiation, which enhances their availability and reduces the overall cost of goods. This breakthrough enables cost reduction in pharmaceutical intermediates manufacturing by allowing palladium usage to drop to as low as 0.05 mol% without sacrificing yield, a feat that was previously unattainable with standard ligand systems.
Mechanistic Insights into Pd-Catalyzed Alpha-Monoarylation
The exceptional performance of these carbazolyl phosphorus ligands stems from their ability to form highly stable and active palladium complexes that facilitate the key steps of the catalytic cycle: oxidative addition, transmetallation (or enolate formation), and reductive elimination. The electron-rich nature of the phosphine moiety combined with the electron-donating characteristics of the carbazole nitrogen enhances the electron density at the palladium center. This acceleration of the oxidative addition step is critical when dealing with less reactive aryl chlorides, expanding the substrate scope to include a wider variety of commercially available starting materials. Additionally, the bulky substituents on the ligand prevent the aggregation of palladium species into inactive nanoparticles, ensuring that the catalyst remains homogeneous and active throughout the reaction duration. This mechanistic robustness is vital for achieving consistent batch-to-batch reproducibility in a GMP manufacturing environment.
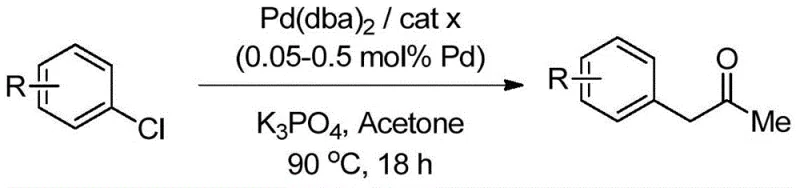
Impurity control is another critical aspect where the mechanistic design of these ligands shines. In traditional systems, the enhanced acidity of the alpha-protons in the monoarylated product often leads to rapid formation of enolates that react again to form diarylated byproducts. The specific steric bulk of the carbazolyl ligands creates a kinetic barrier that slows down this secondary reaction, effectively freezing the process at the desired monoarylation stage. This intrinsic selectivity minimizes the formation of closely related impurities that are difficult to separate, thereby simplifying the downstream purification workflow. For quality control teams, this means a cleaner crude reaction profile and a higher probability of meeting stringent purity specifications without extensive reprocessing. The compatibility of this system with various functional groups, including esters, ketones, and heterocycles, further underscores its versatility for synthesizing diverse high-purity pharmaceutical intermediates.
How to Synthesize Carbazolyl Phosphorus Ligand Efficiently
The preparation of these high-performance ligands follows a robust and scalable two-step protocol that is well-suited for industrial production. The process begins with the N-arylation of substituted carbazoles using dihalobenzenes under copper catalysis, followed by a lithiation-phosphorylation sequence to install the phosphine group. This route avoids the use of sensitive reagents until the final step and utilizes common solvents like xylene and THF, facilitating easy scale-up. The detailed standardized synthesis steps, including specific temperatures, molar ratios, and workup procedures, are outlined in the guide below to ensure successful replication in your laboratory.
- Perform a thermal reaction of octasubstituted-9H-carbazole with 1,2-dibromobenzene using a copper(I) catalyst and base at 150-190°C to form the bromo-intermediate.
- Dissolve the bromo-intermediate in THF, cool to -78°C, and treat with n-butyllithium followed by disubstituted chlorophosphine to yield the final ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ligand technology translates into tangible operational improvements and risk mitigation strategies. The primary value driver is the drastic reduction in catalyst loading, which directly lowers the bill of materials for precious metals. Since palladium is a high-cost commodity with volatile pricing, minimizing its usage from typical levels down to trace amounts significantly insulates the production budget from market fluctuations. Furthermore, the simplified synthesis of the ligand itself implies a more stable and reliable supply base, reducing the risk of shortages that can halt production lines. The ability to run reactions at lower catalyst concentrations also reduces the burden on waste treatment facilities, as there is less heavy metal contamination to manage in the effluent streams.
- Cost Reduction in Manufacturing: The implementation of these ligands eliminates the need for expensive, multi-step ligand syntheses and reduces palladium consumption by orders of magnitude. This dual saving mechanism results in substantial cost savings per kilogram of produced intermediate, improving the overall margin profile of the manufacturing process. By avoiding the use of complex proprietary ligands that carry high licensing or purchase premiums, companies can achieve a more lean and cost-effective production model that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: The synthetic route for these carbazolyl ligands relies on readily available starting materials such as substituted carbazoles and dihalobenzenes, which are commodity chemicals with established supply chains. This reduces dependency on single-source suppliers for exotic reagents and mitigates the risk of supply disruptions. Additionally, the air stability of the final ligand product simplifies logistics, allowing for standard shipping and storage conditions without the need for specialized inert atmosphere packaging, further streamlining the procurement process.
- Scalability and Environmental Compliance: The reaction conditions described in the patent, such as thermal heating in xylene, are easily transferable from bench scale to multi-ton reactors, ensuring seamless commercial scale-up of complex pharmaceutical intermediates. The high selectivity of the reaction reduces the generation of hazardous organic waste associated with purification, aligning with green chemistry principles and regulatory requirements. This environmental compliance not only avoids potential fines but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of carbazolyl phosphorus ligands in industrial processes. These answers are derived directly from the experimental data and technical specifications provided in patent CN106588983B, ensuring accuracy and relevance for decision-makers evaluating this technology for their pipelines.
Q: What is the primary advantage of these carbazolyl ligands over conventional phosphines?
A: These ligands enable palladium catalyst loading as low as 0.05 mol% while maintaining high yields up to 99%, significantly reducing heavy metal residue and catalyst costs compared to traditional systems requiring 2-5 mol% loading.
Q: Are these ligands stable for long-term storage and transport?
A: Yes, the carbazole phosphine ligands described in the patent exhibit excellent stability against air and moisture, simplifying logistics and storage requirements for global supply chains.
Q: Can this catalytic system handle sterically hindered substrates?
A: The steric bulk provided by the carbazole backbone and substituents allows the catalyst to effectively process aryl halides with high steric hindrance, ensuring broad substrate scope for complex intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazolyl Phosphorus Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like those described in CN106588983B for the future of fine chemical synthesis. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications for both the ligands and the resulting intermediates, guaranteeing that every batch meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with our technical team to explore how these novel ligands can optimize your specific synthetic routes. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how integrating these high-efficiency catalysts can drive down your costs and accelerate your time to market.
