Advanced Pd-Catalyzed Synthesis of Branched 1,4-Diene Amides for Pharmaceutical Applications
Advanced Pd-Catalyzed Synthesis of Branched 1,4-Diene Amides for Pharmaceutical Applications
The landscape of organic synthesis for complex pharmaceutical intermediates is constantly evolving, driven by the need for more efficient, selective, and economically viable pathways. A significant breakthrough in this domain is documented in patent CN110627717B, which discloses a novel method for synthesizing branched 1,4-diene amide derivatives. This technology leverages a palladium-catalyzed C-H activation strategy, utilizing 8-aminoquinoline amide as a critical N,N-bidentate directing group. For R&D directors and procurement specialists in the fine chemical sector, this patent represents a pivotal shift away from traditional, often cumbersome coupling reactions towards a more streamlined, atom-economical process. The ability to efficiently realize hydrocarbon olefination at the same carbon position opens new avenues for constructing bioactive scaffolds found in natural products and medicinal molecules, such as lipstatin and biseline hydrochloride analogs.
The core innovation lies in the specific interaction between the transition metal catalyst and the substrate, which allows for the direct functionalization of non-conjugated alkenyl amides. Unlike previous methods that were largely restricted to conjugated systems or electron-deficient olefins, this approach demonstrates remarkable versatility. It accommodates a wide range of substituents, including various alkyl, cycloalkyl, substituted phenyl, naphthyl, and heterocyclic groups. This broad substrate scope is crucial for medicinal chemists who require diverse libraries of compounds for structure-activity relationship (SAR) studies. Furthermore, the reaction operates under mild conditions, typically around 40°C, which significantly reduces energy consumption and thermal stress on sensitive functional groups, thereby enhancing the overall purity profile of the final active pharmaceutical ingredient (API) intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,4-diene skeletons has relied on methods such as catalytic coupling reactions, alkene metathesis, or Morita-Baylis-Hillman reactions. While these techniques have served the industry well, they possess inherent limitations that hinder large-scale commercial adoption. Traditional rhodium or iridium-catalyzed allylation reactions, for instance, often require harsh conditions or expensive catalysts that are difficult to remove to trace levels, posing a risk for pharmaceutical applications. Moreover, many existing protocols are limited to activating carbon-hydrogen bonds ortho to the amide group, predominantly yielding linear coupling products. This lack of regioselectivity restricts the chemical space available to drug designers. Additionally, the reliance on exogenous oxidants in some oxidative coupling strategies generates substantial amounts of chemical waste, complicating downstream processing and increasing the environmental footprint of the manufacturing process. These factors collectively contribute to higher production costs and longer lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology outlined in patent CN110627717B offers a transformative solution to these challenges by employing a palladium-catalyzed hydrocarbon allylation strategy. This novel approach utilizes allyl methyl carbonate derivatives as both the alkenylating reagent and the oxidant, eliminating the need for additional oxidative additives. The reaction proceeds through a highly selective C-H activation mechanism directed by the 8-aminoquinoline moiety, ensuring the formation of branched rather than linear products. This regiocontrol is a distinct advantage, providing access to unique structural motifs that are complementary to conventional Heck reaction products. The operational simplicity is another key benefit; the reaction can be conducted in common organic solvents like dimethyl sulfoxide and methanol under an inert argon atmosphere. As illustrated in the general reaction scheme below, the transformation is clean and efficient, producing methanol and carbon dioxide as the only byproducts, which aligns perfectly with green chemistry principles.
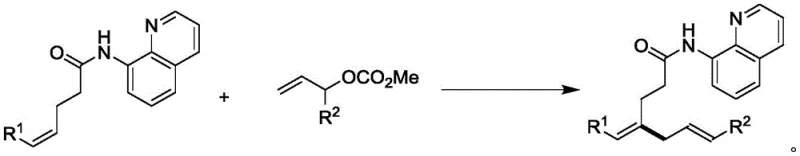
The versatility of this synthetic route is further evidenced by its compatibility with a vast array of functional groups. Whether the substrate contains electron-withdrawing groups like halogens and trifluoromethyl groups, or electron-donating groups like methoxy and alkyl chains, the system maintains high efficiency. This robustness ensures that the process can be adapted for the synthesis of complex drug candidates without extensive re-optimization, making it a reliable choice for a reliable pharmaceutical intermediate supplier aiming to diversify their portfolio.
Mechanistic Insights into Pd-Catalyzed C-H Alkenylation
Understanding the mechanistic underpinnings of this transformation is essential for appreciating its value in process chemistry. The reaction is believed to proceed via a Pd(II)/Pd(IV) catalytic cycle initiated by the coordination of the palladium center to the bidentate 8-aminoquinoline directing group. This coordination brings the metal into close proximity with the target alkenyl C-H bond, facilitating its activation. The subsequent insertion of the allyl species from the methyl carbonate derivative forms a key organopalladium intermediate. A critical feature of this mechanism is the beta-hydride elimination step, which regenerates the double bond and releases the branched 1,4-diene product while restoring the active catalyst species. The use of pivalic acid as an additive plays a crucial role in this cycle, likely acting as a proton shuttle to facilitate the C-H cleavage step and stabilize the transition state. This synergistic effect between the catalyst, additive, and directing group results in the high turnover numbers and selectivity observed in the experimental data.
From an impurity control perspective, this mechanism offers significant advantages. The high regioselectivity imposed by the directing group minimizes the formation of linear isomers, which are often difficult to separate chromatographically. Furthermore, the mild reaction temperature of 40°C suppresses side reactions such as polymerization or decomposition of the diene system, which are common pitfalls in diene synthesis. The specific example of synthesizing (E)-4-benzylidene-N-(quinolin-8-yl)hept-6-enamide, shown below, demonstrates the practical application of these mechanistic principles, yielding a clean product profile with minimal impurities.
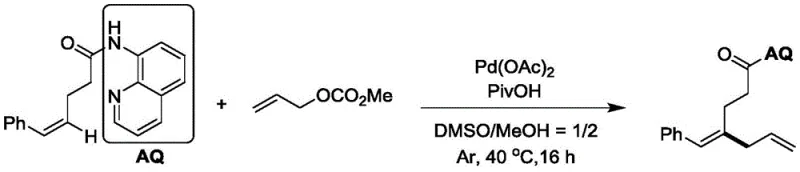
The structural integrity of the products is confirmed through rigorous analytical characterization, including NMR and HRMS, ensuring that the stereochemistry is preserved throughout the transformation. For quality control teams, this predictability translates to simpler validation processes and more consistent batch-to-batch quality, which is paramount when supplying materials for clinical trials or commercial drug production.
How to Synthesize Branched 1,4-Diene Amide Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The process involves the precise combination of the alkenyl quinoline amide substrate with the allyl methyl carbonate derivative in the presence of the palladium catalyst system. The reaction environment must be strictly controlled to exclude oxygen and moisture, typically achieved by sealing the vessel under an argon atmosphere. Following the reaction period, the workup procedure is straightforward, involving standard column chromatography techniques that are familiar to most process chemists. For detailed operational specifics, please refer to the standardized guide below.
- Combine alkenyl quinoline amide, allyl methyl carbonate derivative, palladium acetate catalyst, and pivalic acid additive in a mixed solvent system of DMSO and methanol.
- Seal the reaction vessel under an argon atmosphere and heat the mixture to 40°C, maintaining stirring for approximately 16 hours to ensure complete conversion.
- Upon completion, perform post-treatment by loading the reaction liquid onto a silica gel column, eluting with ethyl acetate and petroleum ether to isolate the pure branched 1,4-diene amide derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible strategic benefits beyond mere chemical novelty. The primary advantage lies in the potential for significant cost reduction in API manufacturing. By eliminating the need for expensive external oxidants and utilizing commercially available, economical starting materials like allyl methyl carbonate, the raw material costs are inherently lower. The high atom economy of the reaction means that a greater proportion of the input mass is converted into the desired product, reducing waste disposal costs and improving overall process efficiency. Furthermore, the mild reaction conditions translate to lower energy requirements for heating and cooling, contributing to a reduced carbon footprint and lower utility expenses over the lifecycle of the product.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis eliminates multiple steps often required in traditional diene construction, such as pre-functionalization of substrates or protection/deprotection sequences. This step economy directly correlates to reduced labor costs, shorter equipment occupancy times, and lower solvent consumption. The ability to achieve high yields without the need for precious metal scavengers or complex purification trains further enhances the economic viability, making it an attractive option for cost-sensitive generic drug production.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including palladium salts, pivalic acid, and various substituted alkenyl amides, are readily available from global chemical suppliers. This abundance mitigates the risk of supply disruptions that can plague specialized reagent markets. Additionally, the robustness of the reaction across a wide range of substrates means that a single manufacturing platform can be used to produce a diverse array of intermediates, allowing for greater flexibility in responding to changing market demands and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The simplicity of the post-treatment process, which relies on standard silica gel chromatography, facilitates easy scale-up from gram to kilogram and eventually to ton scales. The generation of benign byproducts like methanol and carbon dioxide simplifies waste management and ensures compliance with increasingly stringent environmental regulations. This environmental compatibility is a key selling point for companies aiming to meet sustainability goals and maintain a positive corporate social responsibility profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. These insights are derived directly from the patent specifications and are intended to clarify the capabilities and limitations of the technology for potential partners and clients.
Q: What is the key advantage of using 8-aminoquinoline amide in this synthesis?
A: The 8-aminoquinoline amide serves as a robust N,N-bidentate directing group that enables highly selective C-H activation at the alkenyl position, facilitating the formation of branched 1,4-diene structures which are difficult to access via conventional linear coupling methods.
Q: Does this process require expensive external oxidants?
A: No, one of the significant economic advantages of this method is that it does not require an exogenous oxidant. The allyl methyl carbonate derivative functions effectively as both the alkenylating reagent and the internal oxidant source, simplifying the reaction setup and reducing waste.
Q: What represents the maximum yield achievable with this protocol?
A: According to the patent data, the reaction conditions are optimized to achieve yields as high as 93%, demonstrating excellent efficiency and atom economy suitable for industrial scaling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Branched 1,4-Diene Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in modern drug discovery and development. Our team of expert chemists has extensively evaluated the methodology described in patent CN110627717B and is fully equipped to leverage this chemistry for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to manufacturing is seamless and efficient. Our state-of-the-art facilities are designed to handle sensitive organometallic reactions with the utmost precision, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality standards for every batch delivered.
We invite you to collaborate with us to unlock the full potential of branched 1,4-diene amide derivatives in your pipeline. Whether you require custom synthesis for early-stage research or large-scale supply for commercial manufacturing, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are eager to provide you with specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your project timelines and optimize your budget.
