Advanced Grignard Carboxylation for Commercial Scale Production of Montelukast Intermediates
Introduction to Novel Synthetic Pathways for Montelukast Precursors
The pharmaceutical industry continuously seeks robust and scalable methods for producing key intermediates, particularly for high-volume drugs like Montelukast Sodium. Patent CN112624921A introduces a transformative approach to synthesizing 1-hydroxymethyl cyclopropyl acetic acid, a critical building block in the asthma medication supply chain. This innovation shifts away from hazardous traditional chemistries toward a more efficient Grignard-based carboxylation strategy. By leveraging direct carbon dioxide fixation, the process not only streamlines the molecular construction but also addresses significant safety and environmental concerns associated with legacy manufacturing protocols. This technical advancement represents a pivotal shift for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
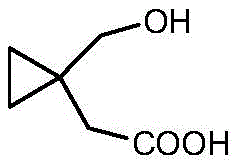
The structural integrity of 1-hydroxymethyl cyclopropyl acetic acid is paramount for the efficacy of the final API. As illustrated in the chemical structure, the molecule features a strained cyclopropane ring substituted with both a hydroxymethyl group and an acetic acid moiety. Maintaining the stability of this ring during synthesis is a common challenge, yet the disclosed method achieves this with remarkable precision. The ability to construct this specific architecture efficiently allows manufacturers to secure a steady supply of high-purity Montelukast intermediates, ensuring that downstream drug production remains uninterrupted and compliant with stringent regulatory standards regarding impurity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
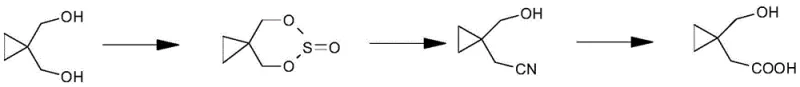
Historically, the synthesis of this vital intermediate has relied on a convoluted multi-step pathway starting from 1,1-cyclopropane dimethanol. This traditional route necessitates sulfurous acid esterification followed by ring-opening and a subsequent substitution reaction using virulent sodium cyanide to introduce the nitrile group. The reliance on cyanide introduces severe public safety risks and imposes strict regulatory controls that complicate logistics and waste management. Furthermore, the initial steps involving diol protection often suffer from poor selectivity, leading to significant material loss and the generation of complex impurity mixtures that are difficult to separate. These inefficiencies result in elevated production costs and extended timelines, creating bottlenecks for cost reduction in API manufacturing.
The Novel Approach
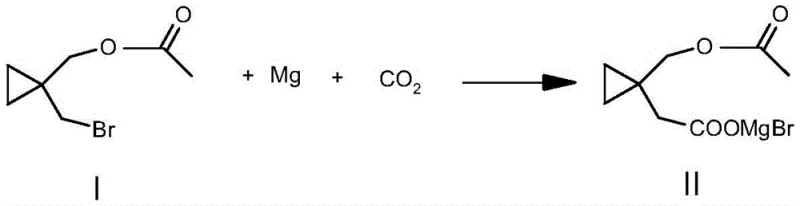
In stark contrast, the methodology described in patent CN112624921A bypasses these hazardous and inefficient steps entirely. The new strategy utilizes bromomethyl cyclopropyl acetate as a starting material, engaging it directly in a Grignard reaction followed by immediate carboxylation with carbon dioxide. This telescoped approach effectively merges reagent formation and carbon-carbon bond formation into a single operational sequence. By eliminating the need for toxic cyanide reagents and avoiding the selectivity issues of diol protection, the process drastically simplifies the workflow. This simplification translates directly into enhanced operational safety and a substantial reduction in the environmental footprint, making it an ideal candidate for modern, green chemistry-compliant facilities seeking commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Grignard Carboxylation and Hydrolysis
The core of this technological breakthrough lies in the precise execution of the Grignard carboxylation mechanism. The process begins with the activation of magnesium metal—using initiators such as elemental iodine or red aluminum—to facilitate the insertion of magnesium into the carbon-bromine bond of the starting acetate. This generates a highly reactive organomagnesium species in situ. Crucially, the reaction system is maintained under anhydrous conditions using solvents like tetrahydrofuran or anhydrous diethyl ether to prevent premature quenching of the Grignard reagent. Once formed, this nucleophile attacks dried carbon dioxide, which is introduced under controlled pressure (0.01-0.03 MPa) and low temperature (5-10°C). This specific thermal and pressure control is vital to maximize the yield of the carboxylated intermediate while suppressing side reactions such as enolization or Wurtz-type coupling.
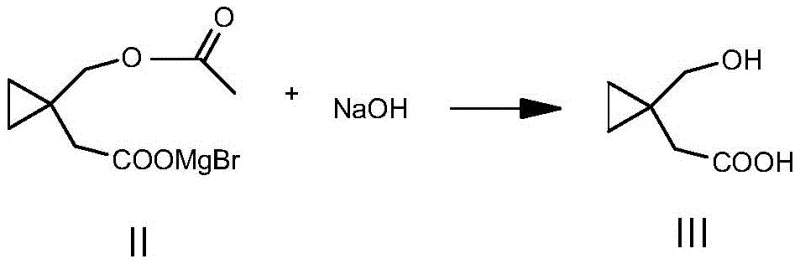
Following the successful fixation of carbon dioxide, the resulting magnesium salt undergoes a straightforward alkaline hydrolysis to reveal the free carboxylic acid and the hydroxymethyl group. The use of bases like sodium hydroxide or potassium hydroxide at moderate temperatures (25-30°C) ensures complete cleavage of the ester functionality without compromising the sensitive cyclopropane ring. This step is critical for impurity control, as harsh acidic or basic conditions could potentially open the strained ring. The patent data indicates that this mild hydrolysis, followed by careful pH adjustment and extraction, yields a product with exceptional purity, often exceeding 98% as determined by HPLC. This high level of chemical fidelity is essential for reducing lead time for high-purity intermediates in the final drug substance synthesis.
How to Synthesize 1-Hydroxymethyl Cyclopropyl Acetic Acid Efficiently
Implementing this synthesis requires careful attention to the initiation of the Grignard reaction and the management of gas-liquid mass transfer during the carbonation phase. The protocol dictates the use of freshly planed magnesium or high-surface-area powder to ensure rapid initiation at 25-30°C. Once the Grignard reagent is established, the system is cooled, and dried CO2 is bubbled through the solution to drive the equilibrium toward the carboxylated product. The detailed standardized synthetic steps, including specific stoichiometric ratios and workup procedures, are outlined below to guide process engineers in replicating this high-yielding route.
- Preparation of Grignard Reagent: React bromomethyl cyclopropyl acetate with magnesium strips or powder in anhydrous THF or ether, initiated by iodine or red aluminum at 25-30°C.
- Carbon Dioxide Fixation: Introduce dried carbon dioxide into the reaction system at 5-10°C and 0.01-0.03 MPa to form magnesium bromide cyclopropyl acetate.
- Alkaline Hydrolysis: Treat the intermediate with sodium hydroxide or potassium hydroxide at 25-30°C, followed by acidification to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic advantages beyond mere technical feasibility. The elimination of sodium cyanide removes a major regulatory hurdle and safety liability, significantly lowering the cost of compliance and insurance. Furthermore, the reduction in synthetic steps from a multi-stage sequence to essentially two main operations drastically improves the overall throughput of the manufacturing plant. This efficiency gain means that facilities can produce larger batches in less time, enhancing the reliability of supply for downstream API manufacturers who depend on consistent delivery schedules to meet global market demand.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic tree. By removing the need for expensive protecting group chemistry and toxic cyanide reagents, the raw material costs are significantly lowered. Additionally, the higher overall yield reported in the patent examples implies less waste of starting materials and solvents. The avoidance of complex purification steps required to remove cyanide-related impurities further reduces operational expenditures, resulting in a more cost-effective production model that enhances margin potential for suppliers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable starting materials like bromomethyl cyclopropyl acetate and magnesium. Unlike specialized cyanide sources which may face shipping restrictions or supply volatility, the reagents for this Grignard process are commodity chemicals with robust global availability. This stability ensures that production schedules are less likely to be disrupted by raw material shortages. Moreover, the simplified process flow reduces the risk of batch failures due to operational complexity, guaranteeing a more consistent output of critical intermediates for the pharmaceutical sector.
- Scalability and Environmental Compliance: From an environmental perspective, this route aligns perfectly with modern green chemistry principles. The absence of heavy metal catalysts and toxic cyanide waste streams simplifies effluent treatment and reduces the burden on waste disposal infrastructure. The process is inherently scalable, as the Grignard carboxylation is a well-understood unit operation in fine chemical manufacturing. This scalability allows producers to easily ramp up from pilot batches to multi-ton commercial production without encountering the nonlinear challenges often associated with more exotic catalytic systems, ensuring long-term sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on how this technology can be integrated into existing manufacturing frameworks to improve efficiency and safety profiles.
Q: Why is the Grignard carboxylation route preferred over the traditional cyanide substitution method?
A: The traditional route relies on toxic sodium cyanide and involves complex protection/deprotection steps with poor selectivity. The Grignard route eliminates cyanide usage entirely, significantly improving operational safety and reducing environmental hazards while shortening the synthetic sequence.
Q: What are the critical control parameters for the carbon dioxide fixation step?
A: Strict moisture control is essential; dried carbon dioxide must be used to prevent Grignard reagent decomposition. The reaction should be maintained at a low temperature of 5-10°C and a pressure of 0.01-0.03 MPa to ensure optimal nucleophilic addition and minimize side reactions.
Q: How does this synthesis method impact the purity profile of the final Montelukast intermediate?
A: By avoiding the harsh conditions and multiple steps of the conventional route, this method reduces the formation of difficult-to-remove impurities. The patent reports HPLC purity levels exceeding 98%, facilitating easier downstream processing for API manufacturers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Hydroxymethyl Cyclopropyl Acetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust intermediate supply chains for the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of large-scale API manufacturing. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1-hydroxymethyl cyclopropyl acetic acid supports the highest standards of drug safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this greener, more efficient method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements.
