Advanced Chiral Quinoline-Imidazoline Ligands for Scalable Pharmaceutical Intermediate Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex chiral scaffolds, particularly gamma-lactams, which serve as ubiquitous core skeletons in numerous biologically active molecules and drugs. Patent CN111925356A introduces a significant breakthrough in this domain by disclosing a novel class of chiral quinoline-imidazoline ligands. These ligands are designed to overcome the historical challenges associated with asymmetric catalysis, offering a green and environmentally friendly route starting from cheap and readily available raw materials. The synthesis is characterized by its simplicity, efficiency, and the production of ligands that are stable to water and oxygen, addressing critical pain points in catalyst handling and storage. This technological advancement provides a new type of ligand product that facilitates the asymmetric difunctionalization of alkenes, specifically enabling the high-yield and high-enantioselective synthesis of chiral cyclic lactams under mild reaction conditions.
The development of efficient chiral ligands has long been a bottleneck in the commercial scale-up of complex pharmaceutical intermediates. In the early stages of chiral ligand development, organophosphine ligands dominated due to their synergistic catalytic activity; however, they often suffer from sensitivity to air and moisture, complicating their industrial application. Furthermore, many asymmetric reactions utilizing chiral phosphine ligands simply cannot achieve the desired transformation, particularly when dealing with monosubstituted alkenes where stereoselectivity is difficult to control via radical pathways. Conventional methods often rely on 1,1-disubstituted alkenes to achieve benzoheterocyclic systems, limiting the structural diversity of accessible compounds. Additionally, synthesizing asymmetric alpha-monosubstituted gamma-lactams from simple lactams is extremely challenging due to potential overalkylation side reactions and epimerization under basic conditions, often resulting in lower enantioselectivities and yields even with stoichiometric chiral bases.
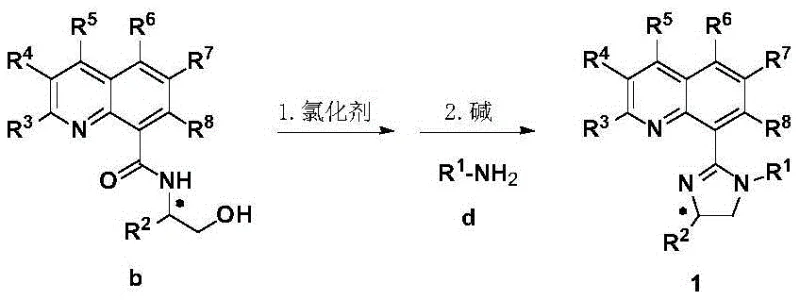
The novel approach presented in this patent fundamentally shifts the paradigm by utilizing a chiral quinoline-imidazoline framework that combines a rigid quinoline skeleton with a tunable imidazoline group. This structural design allows for precise modulation of steric hindrance and electronic cloud density around the metal center, which is key to improving reactivity and enantioselectivity. The synthesis route is remarkably concise, involving the condensation of quinoline-8-carboxylic acid with a chiral beta-amino alcohol, followed by chlorination and cyclization with a primary amine. This method realizes the economical large-scale preparation of target molecules with high efficiency. Unlike traditional methods that struggle with monosubstituted alkene substrates, this ligand system enables the transition metal-catalyzed asymmetric bifunctionalization of alkene-tethered amine acyl chlorides with halides, opening up vast research space and development prospects for synthesizing non-benzo-based chiral heterocyclic compounds that were previously inaccessible.
Mechanistic Insights into Nickel-Catalyzed Asymmetric Difunctionalization
The mechanistic efficacy of these ligands lies in their ability to coordinate with transition metals, specifically nickel, to create a highly defined chiral environment that controls the stereochemical outcome of radical-mediated reactions. In the catalytic cycle, the quinoline-imidazoline ligand forms a complex with a nickel source, such as nickel perchlorate or bis(1,5-cyclooctadiene)nickel, in the presence of a reducing metal like manganese. This active catalytic species facilitates the generation of radical intermediates from the alkene-tethered amine acyl chloride substrate. The bulky tert-butyl group on the imidazoline ring plays a crucial role in shielding one face of the coordinating metal center, thereby directing the migratory insertion of the alkene and the subsequent trapping of the radical species with high fidelity. This steric control is essential for suppressing side reactions such as overalkylation and ensuring that the cyclization proceeds through the desired pathway to form the gamma-lactam ring with excellent optical purity.
Furthermore, the electronic properties of the quinoline backbone contribute to the stability of the catalytic complex, preventing premature decomposition under the reaction conditions. The ligand's insensitivity to water and oxygen suggests that the coordination sphere is robust enough to withstand minor environmental fluctuations, which is a rare and valuable trait in asymmetric catalysis. This robustness translates directly to process reliability, as the catalyst maintains its activity over extended reaction times, typically ranging from 20 to 108 hours depending on the substrate. The ability to tolerate various functional groups on both the amine acyl chloride and the halide coupling partner demonstrates the versatility of this mechanistic system. By effectively managing the radical pathway, which is notoriously difficult to control stereoselectively, this technology solves one of the current difficulties in organic synthesis, allowing for the construction of complex molecules while introducing two distinct functional moieties simultaneously.
How to Synthesize Chiral Quinoline-Imidazoline Ligands Efficiently
The synthesis of these high-performance ligands is designed for operational simplicity and scalability, making it an ideal candidate for technology transfer from laboratory to pilot plant. The process begins with the activation of quinoline-8-carboxylic acid using a carbonyl activator such as isobutyl chloroformate in the presence of a base like N-methylmorpholine, followed by the addition of a chiral beta-amino alcohol to form the amide intermediate. This intermediate is then subjected to chlorination using thionyl chloride or phosphorus pentachloride, generating a reactive species that immediately undergoes cyclization upon treatment with a primary amine and a base like triethylamine. The detailed standardized synthesis steps for producing these ligands with consistent quality and high optical purity are outlined in the guide below.
- Condense quinoline-8-carboxylic acid with a chiral beta-amino alcohol using a carbonyl activator like isobutyl chloroformate and a base such as N-methylmorpholine in dichloromethane to form the amide intermediate.
- Activate the amide intermediate by refluxing with a chlorinating agent such as thionyl chloride or phosphorus pentachloride to generate the imidoyl chloride species in situ.
- React the activated intermediate with a primary amine in the presence of a base like triethylamine at room temperature to cyclize and form the final chiral quinoline-imidazoline ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers substantial strategic benefits beyond mere chemical performance. The primary advantage lies in the inherent stability of the quinoline-imidazoline ligands, which are insensitive to water and oxygen. This characteristic drastically simplifies storage and transportation requirements, eliminating the need for expensive inert atmosphere packaging or cold chain logistics that are often mandatory for sensitive phosphine ligands. Consequently, this leads to significant cost reduction in pharmaceutical intermediate manufacturing by lowering the total cost of ownership associated with catalyst management. The robustness of the ligands ensures a longer shelf life, reducing waste from degraded materials and enhancing overall inventory efficiency for large-scale production facilities.
- Cost Reduction in Manufacturing: The synthetic route for these ligands utilizes cheap and readily available raw materials such as quinoline-8-carboxylic acid and common chiral amino alcohols, avoiding the need for exotic or prohibitively expensive precursors. The reaction conditions are mild, typically operating between 0°C and room temperature for the catalytic step, which significantly reduces energy consumption compared to processes requiring cryogenic conditions or high-temperature reflux. Furthermore, the high catalytic activity means that lower catalyst loadings can potentially be employed to achieve the same turnover, directly decreasing the cost per kilogram of the final chiral lactam product. The elimination of complex purification steps often required to remove metal residues from sensitive catalysts further streamlines the downstream processing costs.
- Enhanced Supply Chain Reliability: The simplicity of the ligand synthesis, involving straightforward condensation and cyclization steps, ensures a reliable and consistent supply of the catalyst itself. Since the starting materials are commodity chemicals, the risk of supply chain disruption due to raw material scarcity is minimized. The high yield and selectivity reported in the patent examples indicate a robust process that is less prone to batch-to-batch variability, which is critical for maintaining continuous manufacturing schedules. This reliability allows supply chain planners to forecast production timelines with greater accuracy, reducing the need for safety stock and minimizing lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is described as green and environmentally friendly, aligning with increasingly stringent global environmental regulations. The use of common solvents like dichloromethane and ethanol, combined with the absence of highly toxic heavy metals in the ligand structure itself, simplifies waste treatment and disposal protocols. The high atom economy of the difunctionalization reaction, which constructs complex rings in a single step, reduces the generation of chemical waste compared to multi-step linear syntheses. This efficiency supports the commercial scale-up of complex polymer additives or drug candidates, ensuring that the manufacturing process remains sustainable and compliant with environmental standards as production volumes increase from grams to tons.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this methodology into existing production lines or new drug discovery pipelines.
Q: What are the stability characteristics of these quinoline-imidazoline ligands?
A: According to patent CN111925356A, these ligands exhibit excellent stability against water and oxygen, making them significantly easier to store and handle compared to sensitive phosphine ligands, which simplifies logistics and reduces degradation risks during transport.
Q: Can these ligands be used for synthesizing non-benzo-based chiral heterocycles?
A: Yes, the patent highlights that these ligands are specifically effective for the asymmetric difunctionalization of monosubstituted alkene-tethered amine acyl chlorides, overcoming previous limitations where only 1,1-disubstituted alkenes were viable for benzoheterocyclic systems.
Q: What metal catalysts are compatible with this ligand system?
A: The technology demonstrates high compatibility with nickel catalysts, specifically using nickel perchlorate or bis(1,5-cyclooctadiene)nickel, along with manganese as a reducing metal, to achieve high enantioselectivity in lactam formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Quinoline-Imidazoline Ligands Supplier
The technological potential of chiral quinoline-imidazoline ligands represents a significant opportunity for advancing the synthesis of high-value pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative academic discoveries are successfully translated into industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch of catalyst and intermediate meets the highest international standards. We understand that consistency is key in asymmetric catalysis, and our manufacturing processes are designed to deliver the precise stereochemical control required for modern drug synthesis.
We invite you to collaborate with us to leverage this advanced catalytic technology for your specific projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your production needs, demonstrating how switching to this robust ligand system can optimize your operational expenditures. Please contact our technical procurement team to request specific COA data for our available ligand variants and to discuss route feasibility assessments for your target molecules. Together, we can accelerate your development timeline and secure a competitive advantage in the global market.
