Advanced Benzimidazole-Indole Phosphine Ligands for High-Efficiency Cross-Coupling and Commercial Scale-Up
Introduction to Next-Generation Phosphine Ligand Technology
The landscape of transition metal-catalyzed cross-coupling reactions continues to evolve with the introduction of specialized ligand architectures designed to overcome traditional limitations. Patent CN107459533B introduces a novel class of 2-(3-(disubstituted phosphino)-N-alkyl-1H-indol-2-yl)-N-alkyl-benzimidazole phosphine ligands that represent a significant advancement in catalytic efficiency and operational stability. These compounds feature a unique hybrid skeleton combining indole and benzimidazole moieties, which provides a rigid yet tunable framework for coordinating transition metals such as palladium. Unlike conventional phosphines that often suffer from rapid oxidation or thermal degradation, this new structural motif offers enhanced robustness, making it particularly suitable for demanding industrial applications where reproducibility is paramount.
The strategic design of these ligands addresses critical pain points in modern organic synthesis, specifically the need for high turnover numbers and compatibility with sensitive functional groups. The general structure, as depicted below, highlights the versatility of the substitution patterns available at the nitrogen atoms and the phosphorus center, allowing for precise optimization of the catalytic environment. This level of molecular engineering ensures that the resulting palladium complexes maintain high activity over extended reaction periods, facilitating the synthesis of complex biaryl structures found in numerous active pharmaceutical ingredients.
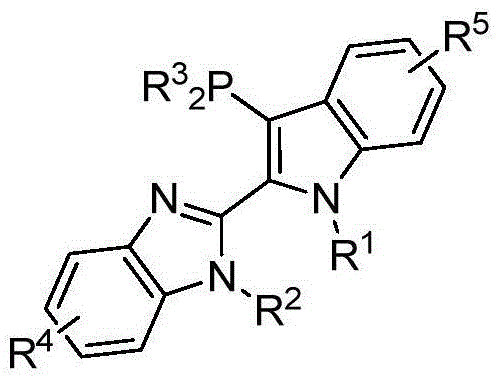
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reliance on standard triarylphosphine ligands in Suzuki-Miyaura couplings has presented several persistent challenges for process chemists and supply chain managers. Traditional ligands often require rigorous exclusion of oxygen and moisture, necessitating expensive glovebox operations or complex Schlenk line techniques that hinder scalability. Furthermore, many established catalytic systems demand relatively high loadings of precious palladium metal to achieve acceptable conversion rates, leading to elevated production costs and significant downstream burdens associated with removing trace heavy metal contaminants from the final product. The instability of these older ligand systems can also result in inconsistent batch-to-batch performance, creating variability that is unacceptable in regulated pharmaceutical manufacturing environments.
The Novel Approach
The methodology outlined in the patent data proposes a transformative solution through the deployment of the benzimidazole-indole scaffold. This novel approach leverages the inherent electronic richness of the indole ring combined with the chelating potential of the benzimidazole unit to create a highly stable coordination sphere around the metal center. The synthesis pathway is streamlined, utilizing readily available starting materials such as indole-2-carboxylic acid and o-phenylenediamine, which mitigates supply chain risks associated with exotic reagents. By installing the phosphine group via a late-stage lithiation strategy, the process ensures high regioselectivity and purity. This robust design not only enhances the longevity of the catalyst but also expands the substrate scope to include challenging electrophiles like aryl chlorides, which are typically less reactive than their bromide or iodide counterparts.
Mechanistic Insights into Ligand Synthesis and Catalytic Activation
Understanding the formation of these sophisticated ligands is crucial for R&D teams aiming to replicate or scale the technology. The synthesis involves a multi-step sequence that builds complexity while maintaining high fidelity. A critical mechanistic step involves the installation of the phosphine functionality via a lithium-halogen exchange protocol. As illustrated in the reaction scheme below, the precursor bromo-intermediate is treated with n-butyllithium at cryogenic temperatures (-75 to -80°C) to generate a reactive organolithium species. This intermediate is subsequently quenched with a disubstituted chlorophosphine, such as chlorodiphenylphosphine or dicyclohexylchlorophosphine, to forge the carbon-phosphorus bond. This low-temperature protocol is essential for preventing side reactions and ensuring the integrity of the sensitive indole backbone.
Once synthesized, the ligand engages with palladium precursors to form the active catalytic species. The bulky and electron-rich nature of the phosphine substituent facilitates the oxidative addition of aryl chlorides, which is often the rate-determining step in cross-coupling cycles. The rigid backbone prevents the formation of inactive palladium black precipitates, thereby sustaining the catalytic cycle for longer durations. This mechanistic stability translates directly to practical benefits, such as the ability to run reactions at lower catalyst loadings without sacrificing yield. The compatibility with various solvents like THF and 1,4-dioxane further underscores the flexibility of this system, allowing process engineers to select conditions that optimize solubility and heat transfer for large-scale reactors.
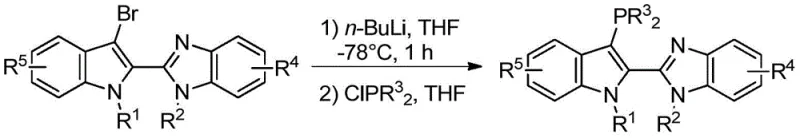
How to Synthesize 2-(3-(Disubstituted Phosphino)-N-alkyl-1H-indol-2-yl)-N-alkyl-benzimidazole Efficiently
The preparation of these high-performance ligands follows a logical four-step progression that balances yield with operational simplicity. The process begins with the condensation of indole-2-carboxylic acid and o-phenylenediamine in ethylene glycol, followed by N-alkylation to introduce the desired steric bulk. Subsequent bromination at the indole 3-position sets the stage for the final phosphination. For detailed standard operating procedures, safety data, and specific stoichiometric ratios required for GMP-compliant production, please refer to the technical guide below.
- Condense indole-2-carboxylic acid with o-phenylenediamine in ethylene glycol with sulfuric acid catalyst under reflux to form the benzimidazole-indole core.
- Perform N-alkylation of the intermediate using sodium hydride or potassium hydroxide with alkyl halides or dimethyl sulfate in THF or DMF.
- Introduce a bromine atom at the 3-position of the indole ring using N-bromosuccinimide (NBS) in tetrahydrofuran at controlled temperatures.
- Execute lithium-halogen exchange with n-butyllithium at -78°C followed by quenching with disubstituted chlorophosphine to install the phosphine group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this ligand technology offers compelling economic and logistical advantages that extend beyond simple reaction yield. The primary value driver is the dramatic reduction in precious metal consumption. By enabling palladium loadings as low as 0.0025 mol%, the technology significantly lowers the direct cost of the catalyst system. More importantly, this reduction minimizes the burden on downstream purification processes. Removing trace palladium from pharmaceutical intermediates often requires specialized scavengers or multiple recrystallization steps; lowering the initial metal input simplifies this workflow, reduces waste generation, and shortens the overall manufacturing cycle time.
- Cost Reduction in Manufacturing: The ability to utilize aryl chlorides instead of more expensive aryl iodides or bromides represents a substantial raw material cost saving. Aryl chlorides are generally more abundant and cheaper to source, yet they traditionally require more active catalysts. This ligand system bridges that gap, allowing the use of cost-effective starting materials without compromising reaction efficiency. Additionally, the air and moisture stability of the ligand reduces the need for expensive inert gas blanketing during storage and handling, further lowering operational expenditures.
- Enhanced Supply Chain Reliability: The synthetic route relies on commodity chemicals such as ethylene glycol, sulfuric acid, and common alkyl halides, which are widely available from multiple global suppliers. This diversification of the supply base mitigates the risk of shortages that can plague specialized reagent markets. The robustness of the ligand also implies a longer shelf life, allowing manufacturers to maintain strategic inventory buffers without fear of degradation, thus ensuring continuous production schedules even during upstream supply disruptions.
- Scalability and Environmental Compliance: The reaction conditions described, such as refluxing in ethylene glycol or stirring in THF at ambient temperatures, are readily scalable from gram to tonne quantities using standard stainless steel reactor infrastructure. The high atom economy and reduced heavy metal usage align with green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing process. This compliance is increasingly critical for maintaining vendor status with major multinational pharmaceutical companies that enforce strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this ligand class in industrial settings. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable foundation for feasibility assessments.
Q: What are the stability characteristics of these benzimidazole-indole phosphine ligands?
A: According to patent CN107459533B, these ligands exhibit exceptional stability against air and moisture, unlike many traditional phosphine ligands. This stability simplifies storage and handling requirements, reducing the need for stringent inert atmosphere conditions during certain processing stages.
Q: How does this ligand system impact palladium catalyst loading in Suzuki reactions?
A: The ligand forms highly active complexes with palladium, allowing for drastically reduced catalyst loading. Experimental data in the patent demonstrates effective catalysis with palladium concentrations as low as 0.0025 mol%, significantly lowering heavy metal residue concerns and raw material costs.
Q: Can the steric and electronic properties of the ligand be tuned?
A: Yes, the molecular architecture allows for extensive modulation. By varying the N-alkyl groups on the indole and benzimidazole rings, as well as the substituents on the phosphorus atom, chemists can fine-tune the steric bulk and electron density to optimize performance for specific substrate classes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole-Indole Phosphine Ligand Supplier
As the demand for efficient and sustainable catalytic solutions grows, NINGBO INNO PHARMCHEM stands ready to support your development and production needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of ligand meets the exacting standards required for sensitive cross-coupling applications. We understand that consistency is key to your success, and our quality management systems are designed to deliver that reliability every time.
We invite you to collaborate with our technical team to evaluate how this advanced ligand technology can optimize your specific synthetic routes. Whether you require a Customized Cost-Saving Analysis for your current palladium-catalyzed processes or need specific COA data to validate performance in your pilot plant, our experts are available to assist. Contact our technical procurement team today to request route feasibility assessments and discuss how we can become your strategic partner in delivering high-purity pharmaceutical intermediates.
