Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Advanced Drug Discovery
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Advanced Drug Discovery
The rapid evolution of medicinal chemistry demands efficient access to complex heterocyclic scaffolds that can modulate biological targets with high precision. A recent breakthrough detailed in patent CN116253692A introduces a novel preparation method for trifluoromethyl substituted 1,2,4-triazine compounds, addressing critical bottlenecks in the synthesis of these valuable pharmacophores. These nitrogen-containing heterocycles are renowned for their broad spectrum of biological activities, ranging from anticancer and antifungal properties to antihypertensive and antimalarial effects, making them indispensable building blocks in modern drug discovery pipelines. The incorporation of the trifluoromethyl group further enhances these molecules by improving metabolic stability, lipophilicity, and bioavailability, which are paramount parameters for lead optimization. This new methodology stands out by utilizing cheap and readily available starting materials, specifically chlorohydrazones and trifluoroacetyl sulfur ylides, catalyzed by inexpensive potassium carbonate under ambient conditions.
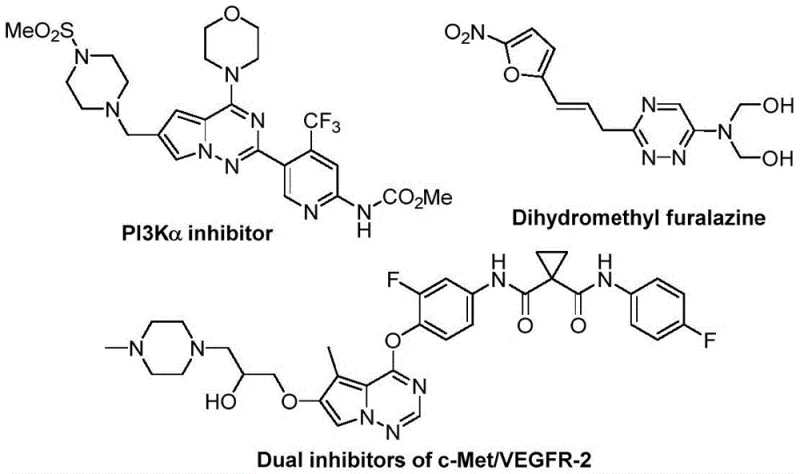
For R&D directors and process chemists, the significance of this invention lies in its ability to bypass the harsh conditions typically associated with triazine formation. Traditional routes often require expensive transition metal catalysts or extreme temperatures that limit functional group tolerance and increase the complexity of purification. By contrast, this air-tolerant, room-temperature protocol offers a streamlined pathway to high-purity intermediates. As a reliable pharmaceutical intermediates supplier, understanding such technological shifts is crucial for maintaining a competitive edge in providing cost-effective solutions for complex API synthesis. The method not only simplifies the operational workflow but also aligns with green chemistry principles by eliminating heavy metal residues, thereby reducing the burden on downstream purification and waste management systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine core has relied on condensation reactions between amidrazones and 1,2-diketones or alkynes, as well as multicomponent reactions involving hydrazides and dicarbonyl compounds. While these classical approaches have served the industry for decades, they are increasingly viewed as suboptimal for modern high-throughput synthesis due to several inherent drawbacks. Conventional methods frequently necessitate the pre-synthesis of specialized substrates, which adds extra steps to the overall synthetic sequence and lowers the cumulative yield. Furthermore, many of these cyclization protocols require elevated temperatures, strong acids or bases, and inert atmospheres, which escalate energy consumption and operational risks. The structural diversity achievable through these older routes is often restricted, limiting the ability of medicinal chemists to rapidly explore structure-activity relationships (SAR) around the triazine core. Additionally, the use of stoichiometric amounts of coupling reagents or toxic solvents in traditional methods generates significant chemical waste, posing challenges for environmental compliance and cost reduction in pharmaceutical manufacturing.
The Novel Approach
The innovative strategy disclosed in the patent data revolutionizes this landscape by employing a [3+3] cycloaddition reaction between chlorohydrazones and trifluoroacetyl sulfur ylides. This approach fundamentally shifts the paradigm from step-wise condensations to a more convergent assembly of the heterocyclic ring. The reaction proceeds efficiently in common organic solvents like tetrahydrofuran (THF) at temperatures ranging from 20 to 40°C, effectively eliminating the need for energy-intensive heating or cryogenic cooling. Crucially, the system utilizes potassium carbonate as a mild, non-toxic promoter instead of expensive or hazardous transition metal catalysts. This metal-free nature is a game-changer for supply chain reliability, as it removes the dependency on fluctuating markets for precious metals like palladium or copper. The robustness of this method allows for the direct formation of the trifluoromethyl-substituted triazine skeleton with high atom economy, minimizing byproduct formation and simplifying the isolation of the final product.
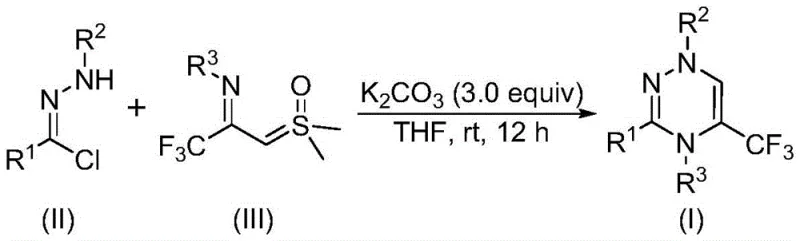
Mechanistic Insights into Potassium Carbonate Promoted Cycloaddition
From a mechanistic perspective, the transformation involves a sophisticated interplay of nucleophilic activation and cycloaddition dynamics that ensures high selectivity and yield. The process initiates with the deprotonation of the chlorohydrazone by potassium carbonate, which facilitates the elimination of hydrogen chloride to generate a reactive nitrile imine intermediate in situ. This highly electrophilic species then engages in a synergistic [3+3] cycloaddition with the trifluoroacetyl sulfur ylide, a unique dipolarophile that delivers the essential trifluoromethyl group directly into the ring structure. During this cyclization event, a molecule of dimethyl sulfoxide is extruded, driving the equilibrium forward and stabilizing the newly formed aromatic triazine system. Alternatively, the reaction may proceed through a stepwise mechanism involving intermolecular nucleophilic addition followed by intramolecular nucleophilic substitution, both pathways converging to the same thermodynamic product. The mild basicity of potassium carbonate is sufficient to drive this cascade without promoting unwanted side reactions such as hydrolysis or polymerization, which are common pitfalls in stronger basic environments.
Impurity control is inherently superior in this system due to the chemoselectivity of the nitrile imine intermediate and the mild reaction conditions. In traditional metal-catalyzed cross-couplings, trace metal residues can coordinate with product amines or heteroatoms, leading to difficult-to-remove impurities that fail stringent regulatory specifications for API intermediates. By avoiding heavy metals entirely, this protocol ensures a cleaner crude reaction profile, significantly reducing the load on purification columns and crystallization steps. The tolerance for various functional groups on the aromatic rings of the starting materials—such as methoxy, halogen, methyl, and trifluoromethyl substituents—demonstrates the versatility of the mechanism. This broad substrate scope implies that the electronic and steric properties of the reactants do not severely hinder the cyclization efficiency, allowing for the synthesis of a diverse library of analogs with consistent quality. For quality assurance teams, this translates to more predictable impurity profiles and easier validation of the manufacturing process.
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent ratios and mixing protocols to maximize conversion. The standard procedure involves charging a reaction vessel with the chlorohydrazone derivative, the trifluoroacetyl sulfur ylide, and three equivalents of potassium carbonate in a suitable aprotic solvent. Tetrahydrofuran is identified as the preferred medium due to its ability to dissolve both organic precursors and the inorganic base effectively, ensuring homogeneous reaction kinetics. The mixture is stirred at room temperature for a duration of 10 to 14 hours, a timeframe that balances complete conversion with operational throughput. Upon completion, the workup is remarkably straightforward: the solid salts are removed by filtration, and the filtrate is concentrated and purified via standard silica gel chromatography. This simplicity makes the method highly attractive for rapid scale-up campaigns where time-to-market is a critical factor.
- Mix chlorohydrazone, trifluoroacetyl sulfur ylide, and potassium carbonate in an organic solvent like THF.
- Stir the reaction mixture at room temperature (20-40°C) under an air atmosphere for 10 to 14 hours.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits that extend beyond mere chemical curiosity. The shift towards metal-free catalysis represents a significant opportunity for cost reduction in pharmaceutical intermediates manufacturing, primarily by eliminating the procurement of expensive noble metal catalysts and the associated ligand systems. Furthermore, the removal of heavy metals simplifies the regulatory compliance landscape, as there is no need for costly and time-consuming metal scavenging processes or rigorous testing for residual metal limits in the final product. The use of potassium carbonate, a commodity chemical with stable pricing and global availability, insulates the production process from the volatility often seen in the specialty catalyst market. This stability allows for more accurate long-term budgeting and reduces the risk of supply disruptions caused by geopolitical factors affecting rare earth or precious metal exports.
- Cost Reduction in Manufacturing: The economic model of this synthesis is bolstered by the use of low-cost inorganic bases and commercially available starting materials that do not require complex multi-step preparation. By operating at room temperature, the process drastically reduces energy consumption compared to refluxing conditions, leading to lower utility costs per kilogram of product. The high atom efficiency of the [3+3] cycloaddition minimizes raw material waste, ensuring that a greater proportion of the input mass is converted into valuable product rather than discarded byproducts. Additionally, the simplified workup procedure reduces the consumption of silica gel and solvents during purification, further driving down the variable costs associated with production. These cumulative savings can be passed down the supply chain, offering competitive pricing for high-purity intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain resilience. Since the reaction tolerates air and moisture to a reasonable extent and does not require strictly anhydrous or oxygen-free environments, the operational complexity is reduced, lowering the barrier for contract manufacturing organizations (CMOs) to adopt the process. The starting materials, such as acyl chlorides, hydrazines, and aromatic amines, are bulk chemicals with established global supply networks, ensuring consistent availability even during market fluctuations. The scalability of the method from gram to multi-kilogram levels without significant re-optimization means that supply can be ramped up quickly to meet sudden increases in demand from clinical trials or commercial launches. This flexibility is crucial for maintaining continuity of supply in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with modern green chemistry mandates. The absence of toxic heavy metals eliminates a major source of hazardous waste, simplifying disposal procedures and reducing the environmental footprint of the manufacturing site. The use of THF, while requiring proper handling, is a well-understood solvent with established recycling protocols, further enhancing the sustainability profile of the process. The mild reaction conditions reduce the risk of thermal runaways or pressure build-ups, creating a safer working environment for plant operators. As regulatory bodies worldwide tighten restrictions on chemical emissions and waste discharge, adopting such inherently cleaner technologies future-proofs the supply chain against evolving compliance requirements, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl triazine synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the practical implications for industrial application. Understanding these nuances helps stakeholders make informed decisions about integrating this route into their existing portfolios or sourcing strategies. The answers reflect a balance between theoretical potential and proven experimental results, ensuring realistic expectations for performance and scalability.
Q: What are the key advantages of this new triazine synthesis method?
A: The method described in patent CN116253692A operates under mild conditions (room temperature, air atmosphere) without heavy metal catalysts, significantly reducing production costs and environmental impact compared to traditional high-temperature cyclizations.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of inexpensive inorganic bases like potassium carbonate and the absence of sensitive catalysts make the process highly robust and easily scalable from gram to multi-kilogram levels for commercial supply.
Q: What is the structural diversity achievable with this route?
A: The reaction tolerates a wide range of substituents on the hydrazine and ylide components, allowing for the synthesis of diverse derivatives including phenyl, naphthyl, and various halogenated or alkoxy-substituted triazines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in accelerating drug development timelines. Our technical team has extensively analyzed the protocol described in CN116253692A and is fully prepared to leverage this chemistry for your custom synthesis projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trifluoromethyl 1,2,4-triazine intermediate delivered meets the highest international standards for pharmaceutical applications. We understand that consistency and quality are non-negotiable in the life sciences industry, and our integrated quality management systems are designed to provide total transparency and traceability.
We invite you to collaborate with us to optimize your supply chain for these critical heterocyclic building blocks. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how this innovative synthesis can enhance your project's economics and timeline. Let us be your strategic partner in navigating the complexities of fine chemical manufacturing, delivering value through scientific excellence and operational reliability.
