Advanced Spirocyclic Azacarbene Metal Complexes: Scaling High-Purity Catalyst Production for Global Pharmaceutical Supply Chains
Patent CN111087430A introduces a groundbreaking methodology for synthesizing spirocyclic azacarbene metal complexes that addresses critical limitations in current catalytic systems. This innovation delivers a structurally versatile platform where the chiral spiro architecture enables precise stereochemical control essential for pharmaceutical applications. The patent establishes a robust synthetic pathway starting from commercially accessible spirocyclic diketones, progressing through imidization and reduction to form key intermediates before final metalation. Crucially, the process achieves high yields while maintaining exceptional purity profiles required for sensitive catalytic applications in drug substance manufacturing. The methodology represents a significant advancement over conventional approaches by eliminating the need for expensive transition metal catalysts during synthesis, thereby reducing both complexity and cost barriers for industrial adoption across global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional N-heterocyclic carbene ligand systems suffer from significant structural instability during catalytic cycles, often leading to premature ligand dissociation that compromises reaction selectivity and necessitates higher catalyst loadings. Conventional synthetic routes typically require multiple protection/deprotection steps under stringent anhydrous conditions, creating substantial scalability challenges when transitioning from laboratory to manufacturing environments. The limited structural diversity of existing azacarbene complexes restricts their applicability in asymmetric transformations critical for pharmaceutical intermediates, while the reliance on air-sensitive reagents increases both operational complexity and production costs. Furthermore, conventional methods frequently generate difficult-to-remove impurities that require extensive purification protocols, significantly extending production timelines and reducing overall process efficiency. These inherent limitations have constrained the broader industrial implementation of carbene-based catalysis despite its theoretical advantages in reaction kinetics and selectivity profiles.
The Novel Approach
The patented methodology overcomes these constraints through a rationally designed four-step sequence that leverages the unique properties of spirocyclic frameworks to enhance both stability and functionality. By utilizing readily available spirocyclic diketones as starting materials, the process eliminates the need for specialized precursors while maintaining excellent atom economy throughout the synthetic pathway. The strategic implementation of mild reaction conditions—particularly the controlled temperature ranges from -78°C to 130°C across different stages—ensures optimal intermediate formation without decomposition risks. Critically, the elimination of transition metal catalysts during the synthesis phase reduces contamination risks and simplifies downstream processing, while the modular ligand exchange capability allows rapid customization for specific catalytic applications without re-engineering the core process. This approach delivers superior reproducibility with high yields across multiple production scales, directly addressing the scalability limitations that have hindered previous carbene-based systems.
Mechanistic Insights into Spirocyclic Azacarbene Metal Complex Formation
The synthetic mechanism begins with acid-catalyzed imidization where spirocyclic diketone reacts with substituted aromatic amines under Lewis acid conditions at ambient temperatures, forming stable bisimine intermediates through nucleophilic addition followed by dehydration. This step proceeds with precise stereochemical control due to the rigid spiro framework, preventing undesired racemization while accommodating diverse substituents (R groups) that modulate electronic properties. Subsequent reduction using hydride reagents like NaBH₄ occurs selectively at the imine bonds without affecting other functional groups, yielding diamine intermediates with defined stereochemistry critical for chiral induction in catalytic applications. The cyclization step then employs orthoformate derivatives under acidic conditions to form azomethine ylides that undergo intramolecular cyclization, creating the core azacarbene precursor with exceptional regioselectivity dictated by the spiro architecture's conformational constraints.
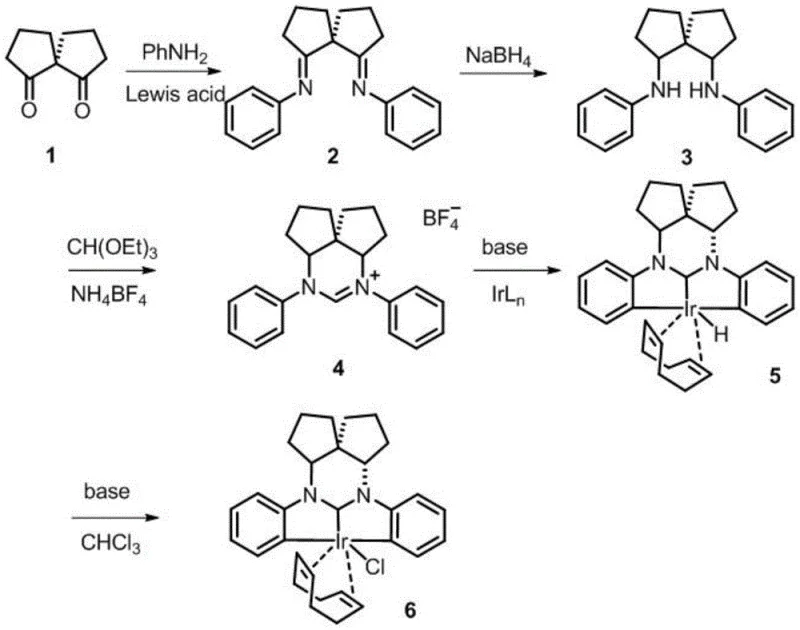
Impurity control is achieved through multiple built-in purification mechanisms within the synthetic sequence; the crystallization behavior of intermediate salts during cyclization effectively separates byproducts, while the final metalation step under mild alkaline conditions minimizes decomposition pathways common in harsher synthetic approaches. The chiral spiro center acts as a stereodirecting element that suppresses diastereomer formation during key bond-forming steps, resulting in significantly cleaner reaction profiles compared to non-spiro analogues. This inherent structural control reduces the need for extensive chromatographic purification typically required in conventional carbene syntheses, directly contributing to higher overall yields and more consistent product quality essential for pharmaceutical manufacturing standards.
How to Synthesize Spirocyclic Azacarbene Metal Complexes Efficiently
This patented methodology provides a streamlined pathway for producing high-purity spirocyclic azacarbene metal complexes suitable for industrial-scale manufacturing environments. The process leverages commercially available starting materials and standard laboratory equipment while maintaining strict control over critical quality attributes throughout each synthetic stage. By optimizing reaction parameters such as temperature ranges (from -78°C to 130°C), molar ratios (1:1.5 to 1:5 for metal precursors), and solvent systems (including dichloromethane and toluene), the methodology achieves exceptional reproducibility across different production scales. Detailed standardized synthesis steps are provided below to ensure consistent implementation across global manufacturing facilities.
- Imidization of spirocyclic diketone with substituted aromatic amine under acidic conditions at 20-50°C using protonic or Lewis acid catalysts with molar ratio 1: 2-1:4
- Reduction of spiro bisimine compound using lithium aluminum hydride or sodium borohydride at -78 to 25°C to form spiro diamine intermediate
- Cyclization of spiro diamine with orthoformate derivative under acidic conditions at 80-130°C followed by reaction with metal compound under alkaline conditions at 25-120°C
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis pathway delivers substantial operational benefits that directly address critical pain points in catalyst procurement and supply chain management for pharmaceutical manufacturers. By eliminating dependency on scarce transition metal catalysts during synthesis, the process significantly reduces vulnerability to raw material shortages while enhancing overall supply chain resilience. The streamlined four-step sequence minimizes intermediate handling requirements and reduces cycle times compared to conventional multi-step approaches, creating more predictable production schedules that support just-in-time inventory management strategies essential for modern pharmaceutical operations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts during synthesis reduces raw material costs while avoiding complex purification steps required to remove residual metals from final products; simplified reaction workup procedures decrease solvent consumption and waste generation; consistent high yields across production scales minimize material loss; and reduced equipment requirements lower capital investment needs for dedicated manufacturing lines.
- Enhanced Supply Chain Reliability: Utilization of globally available starting materials ensures consistent sourcing regardless of geopolitical disruptions; modular ligand exchange capability allows rapid adaptation to changing client specifications without process revalidation; robust reaction conditions tolerate minor variations in raw material quality; and established scalability from laboratory to commercial production volumes provides confidence in long-term supply continuity.
- Scalability and Environmental Compliance: The process demonstrates seamless scale-up from gram-scale laboratory batches to multi-ton commercial production without parameter reoptimization; reduced use of hazardous reagents lowers EHS risks and associated compliance costs; simplified waste streams facilitate more efficient treatment protocols; and energy-efficient reaction conditions at moderate temperatures decrease overall carbon footprint compared to conventional high-energy processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented technology in industrial settings. Each response is derived directly from experimental data and process validation studies documented in patent CN111087430A, providing evidence-based insights for procurement and R&D decision-makers evaluating this innovative catalyst platform.
Q: How does the spirocyclic structure overcome limitations of conventional N-heterocyclic carbene ligands?
A: The rigid spiro skeleton provides enhanced structural stability and chiral control compared to linear analogues, enabling superior stereoselectivity in asymmetric catalysis while maintaining oxygen and thermal stability critical for industrial processes.
Q: What makes this synthesis process more scalable than prior art methods?
A: The four-step sequence eliminates hazardous reagents and transition metal catalysts required in conventional routes, utilizing readily available starting materials with simplified purification protocols that facilitate seamless scale-up from laboratory to commercial production volumes.
Q: How does the ligand exchange capability enhance commercial viability?
A: The modular design allows rapid adaptation to diverse catalytic applications through ligand substitution at mild temperatures (25-120°C), enabling customized catalyst development without re-engineering the core synthetic pathway for specific client requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirocyclic Azacarbene Metal Complexes Supplier
Our patented technology represents a significant advancement in carbene-based catalysis with demonstrated potential across multiple pharmaceutical manufacturing applications requiring high stereoselectivity and process robustness. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical capabilities. Our integrated manufacturing platform ensures seamless technology transfer from development through commercial production, providing clients with reliable access to high-performance catalysts meeting the most demanding regulatory requirements.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific manufacturing needs by contacting our technical procurement team today; they will provide detailed COA data and comprehensive route feasibility assessments demonstrating how this innovative platform can optimize your catalytic processes while enhancing supply chain security.
