Advanced Nickel-Catalyzed Synthesis of Chiral Cyano Cycloalkenones for Commercial Pharmaceutical Manufacturing
Introduction to Breakthrough Nickel-Catalyzed Technology
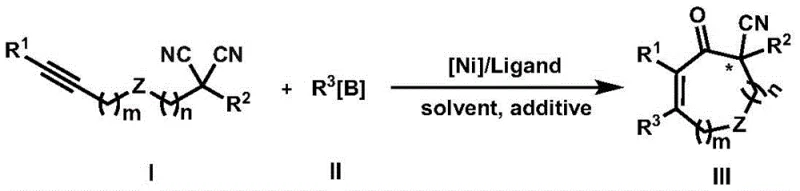
The pharmaceutical industry continuously seeks robust methodologies for constructing sterically congested quaternary carbon centers, particularly those bearing biologically essential cyano groups. Patent CN111499542A introduces a paradigm-shifting preparation method for α-cyano substituted quaternary carbon center cyclic ketene compounds, addressing long-standing synthetic challenges. This technology leverages a sophisticated nickel-catalyzed alkyne insertion and cyclization tandem reaction, utilizing aryl or alkenyl boron reagents alongside alkynyl substituted disubstituted malononitrile compounds. The significance of this innovation lies in its ability to achieve high activity, regioselectivity, and enantioselectivity simultaneously, which is critical for the development of next-generation active pharmaceutical ingredients. By employing a non-precious metal catalyst system, this approach not only enhances chemical efficiency but also aligns with modern green chemistry principles, offering a sustainable pathway for producing high-value fine chemicals and complex drug intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral cyano-containing quaternary carbon centers has been plagued by significant technical hurdles and economic inefficiencies. Prior art methods, such as those reported by Tanaka and Ikariya, relied heavily on expensive precious metal catalysts like rhodium and ruthenium, which imposed substantial cost burdens on large-scale manufacturing processes. Furthermore, these conventional techniques often suffered from poor enantioselectivity, with reported values as low as 30% ee, necessitating costly and yield-reducing recrystallization steps to achieve optical purity. The inherent coordination affinity of the cyano group with transition metals frequently led to catalyst poisoning or unwanted decyanation decomposition, resulting in complex impurity profiles that complicated downstream purification. These limitations restricted the utility of such methods to narrow substrate scopes, rendering them impractical for the diverse library synthesis required in modern drug discovery and development pipelines.
The Novel Approach
In stark contrast, the novel nickel-catalyzed methodology described in the patent offers a transformative solution that overcomes these historical bottlenecks through innovative catalyst design and reaction engineering. By utilizing a tailored nickel complex paired with specific chiral ligands, such as phosphine-oxazoline derivatives, the process achieves exceptional stereocontrol with enantiomeric excess values reaching up to 98% ee in optimized examples. This approach effectively mitigates the risk of decyanation, preserving the integrity of the critical cyano pharmacophore throughout the rigorous cyclization process. The reaction conditions are notably mild, operating effectively in common organic solvents like toluene at moderate temperatures, which enhances operational safety and energy efficiency. Moreover, the inclusion of water as a beneficial additive further stabilizes the catalytic cycle, demonstrating a level of robustness and functional group tolerance that far exceeds previous state-of-the-art technologies, thereby enabling the synthesis of a vastly broader range of structurally diverse intermediates.
Mechanistic Insights into Nickel-Catalyzed Alkyne Insertion and Cyclization
The core of this technological advancement resides in the intricate mechanistic pathway facilitated by the nickel catalyst, which orchestrates a seamless tandem sequence of alkyne insertion and intramolecular cyclization. The catalytic cycle initiates with the oxidative addition or coordination of the nickel species to the alkyne moiety of the disubstituted malononitrile substrate, activated by the chiral ligand environment. This activation lowers the energy barrier for the subsequent nucleophilic attack by the boron reagent, promoting a highly regioselective insertion step that establishes the new carbon-carbon bond with precise stereochemical definition. The chiral ligand plays a pivotal role in discriminating between the prochiral faces of the intermediate, ensuring that the formation of the quaternary center proceeds with high fidelity. Following the insertion, the system undergoes a rapid cyclization event to close the ring, forming the stable cyclic ketene or enamine structure. This mechanism is finely tuned to prevent side reactions, such as polymerization or hydrolysis, which are common pitfalls in similar transition metal-catalyzed transformations.
Impurity control is inherently built into this mechanistic design through the careful selection of reaction parameters and additives that suppress off-cycle pathways. The presence of water, acting as a proton source or stabilizer, helps to manage the reactivity of intermediate organometallic species, preventing the accumulation of reactive byproducts that could lead to tar formation or decomposition. Furthermore, the specific electronic and steric properties of the ligand framework shield the active metal center from deactivation by the strongly coordinating cyano groups, a common failure mode in earlier attempts. This results in a cleaner reaction profile with fewer side products, significantly simplifying the purification workload. The high chemoselectivity ensures that sensitive functional groups on the aryl or alkenyl boron reagents remain intact, allowing for late-stage functionalization strategies. Such mechanistic robustness provides R&D teams with the confidence to scale this chemistry, knowing that the impurity spectrum is well-understood and manageable within standard quality control frameworks.
How to Synthesize α-Cyano Cycloalkenones Efficiently
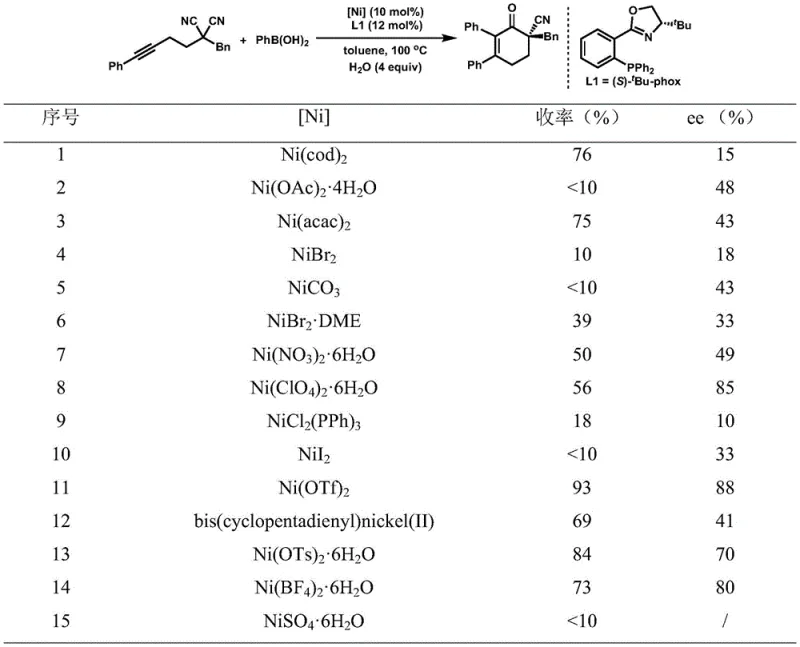
Implementing this synthesis route requires careful attention to the preparation of the catalytic system and the management of reaction conditions to maximize yield and optical purity. The process begins with the in situ generation of the active nickel catalyst by mixing a nickel salt, such as nickel trifluoromethanesulfonate, with a chiral ligand like (S)-tBu-phox in an anhydrous organic solvent under an inert atmosphere. Once the catalyst is formed, the substrates, including the alkynyl malononitrile and the boron reagent, are introduced along with a controlled amount of water to facilitate the transformation. The detailed standardized synthesis steps for this high-efficiency route are outlined in the guide below, providing a clear roadmap for laboratory and pilot-scale execution.
- Prepare the catalytic system by mixing a nickel complex such as Ni(OTf)2 with a chiral ligand like (S)-tBu-phox in an organic solvent under inert atmosphere.
- Add the alkynyl substituted disubstituted malononitrile substrate and the aryl or alkenyl boron reagent to the reaction mixture along with a water additive.
- Heat the reaction mixture to 80-100°C for 16-24 hours, followed by acid workup and purification via column chromatography to isolate the chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this nickel-catalyzed technology presents compelling opportunities for cost optimization and supply chain resilience in the manufacturing of complex pharmaceutical intermediates. The substitution of precious metals like rhodium and ruthenium with abundant nickel drastically reduces the raw material cost base, eliminating the volatility associated with rare earth metal markets. This shift not only lowers the direct cost of goods sold but also simplifies the regulatory burden related to heavy metal residue limits in final drug substances, potentially reducing the need for expensive scavenging steps. The robustness of the reaction conditions allows for the use of standard reactor equipment and common solvents, minimizing capital expenditure requirements for specialized infrastructure. These factors combine to create a manufacturing process that is economically sustainable and less susceptible to supply disruptions caused by geopolitical constraints on critical mineral resources.
- Cost Reduction in Manufacturing: The transition to a nickel-based catalyst system fundamentally alters the cost structure of producing chiral cyano intermediates by removing the dependency on ultra-expensive precious metals. Since nickel is orders of magnitude more abundant and cheaper than rhodium or ruthenium, the direct material cost for the catalyst component is significantly reduced. Furthermore, the high enantioselectivity achieved minimizes the loss of valuable chiral material during purification, improving the overall mass balance and effective yield of the process. The elimination of complex metal removal steps typically required for precious metals further drives down processing costs, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Relying on nickel instead of scarce precious metals mitigates the risk of supply chain bottlenecks that often plague the fine chemical industry. Nickel salts and the requisite ligands are commercially available from multiple global suppliers, ensuring a steady and reliable flow of materials even during periods of market instability. The use of common solvents like toluene and benign additives like water further enhances logistical simplicity, as these materials do not require special handling or storage conditions. This reliability translates to more predictable lead times for customers, allowing for better inventory planning and reducing the need for safety stock holdings in the supply chain.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing mild temperatures and pressures that are easily managed in large-scale industrial reactors. The inclusion of water as a beneficial additive aligns with green chemistry initiatives, reducing the environmental footprint of the manufacturing process. Fewer hazardous byproducts and a cleaner reaction profile mean simpler waste treatment protocols and lower disposal costs. This environmental compatibility ensures compliance with increasingly stringent global regulations regarding chemical manufacturing, facilitating smoother regulatory approvals and maintaining a positive corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production workflows or new drug development projects.
Q: What are the advantages of using nickel over rhodium or ruthenium for this synthesis?
A: Nickel is a non-precious metal, significantly reducing raw material costs compared to rhodium or ruthenium. Additionally, the patented nickel system demonstrates superior enantioselectivity and broader substrate tolerance for constructing cyano-quaternary centers.
Q: How does the process handle the stability of the cyano group during catalysis?
A: The method utilizes specific chiral ligands and mild reaction conditions that prevent the decyanation decomposition often observed with transition metal catalysts, ensuring high integrity of the cyano functional group throughout the transformation.
Q: Is this method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process operates under relatively mild temperatures and uses commercially available solvents like toluene. The use of water as an additive simplifies the reaction profile, making it highly suitable for scale-up in commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable α-Cyano Cycloalkenone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic technologies to deliver high-quality pharmaceutical intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of α-cyano cycloalkenone meets the exacting standards required by top-tier pharmaceutical companies. Our commitment to technical excellence means we can navigate the complexities of chiral synthesis with precision, delivering products that accelerate your drug development timelines.
We invite you to engage with our technical procurement team to discuss how this innovative nickel-catalyzed route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this methodology for your supply chain. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics and our proven capability to deliver reliable, high-purity intermediates at scale.
