Advancing Chiral Epoxide Synthesis: A Novel Rare Earth-TADDOL Co-Catalytic System for Pharmaceutical Intermediates
Introduction to Advanced Asymmetric Epoxidation Technology
The synthesis of optically active epoxy compounds represents a cornerstone in modern organic chemistry, serving as critical intermediates for the production of complex natural products and pharmaceutical agents. Patent CN111892559B introduces a groundbreaking advancement in this field by disclosing a highly efficient application of chiral TADDOL ligands combined with rare earth metal amides for the co-catalyzed asymmetric epoxidation of chalcone compounds. This technology addresses long-standing challenges in stereoselective synthesis, offering a robust pathway to generate high-purity chiral epoxides with exceptional enantioselectivity. By leveraging the unique electronic properties of rare earth metals such as Ytterbium and Lanthanum in conjunction with sterically demanding TADDOL ligands, this method achieves yields ranging from 89% to 99% and enantiomeric excess values between 60% and 94%. For R&D directors and process chemists seeking reliable pharmaceutical intermediate supplier solutions, this patent provides a compelling alternative to traditional methodologies that often suffer from limited substrate scope or苛刻 reaction conditions.
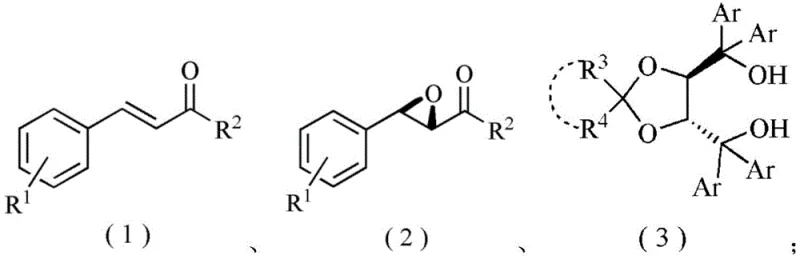
The significance of this innovation lies in its ability to transform alpha,beta-unsaturated ketones into valuable chiral building blocks under relatively mild conditions. The process operates effectively within a temperature range of -40°C to 25°C, utilizing tert-butyl hydroperoxide (TBHP) as the oxidant in the presence of an organic base. This level of control over the stereochemical outcome is paramount for the manufacturing of active pharmaceutical ingredients where optical purity is a strict regulatory requirement. Furthermore, the versatility of the ligand library, encompassing fifteen distinct structural variations (H2L1 to H2L15), allows for fine-tuning the catalytic environment to match specific substrate electronic profiles. As we delve deeper into the technical specifics, it becomes evident that this co-catalytic system represents a substantial leap forward in the commercial scale-up of complex pharmaceutical intermediates, promising both economic and operational advantages for industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this development, the landscape of asymmetric epoxidation for alpha,beta-unsaturated ketones was dominated by methodologies that, while effective in specific niches, presented significant drawbacks for large-scale industrial application. Early reports by Lattanzi et al. utilized S-diphenylprolinol as a chiral small molecule catalyst, but this approach necessitated prohibitively high catalyst loadings of up to 30 mol% and suffered from extended reaction times ranging from 27 to 87 hours. Subsequent modifications by Zhao Gang and colleagues managed to improve enantioselectivity to 96% ee, yet the reaction duration remained impractically long at 144 hours, creating a bottleneck for throughput in a manufacturing setting. Similarly, while List's ammonium salt catalytic system achieved impressive ee values of 99%, it was largely restricted to cyclic unsaturated ketones, limiting its utility for the broad array of acyclic chalcones required in diverse drug synthesis pipelines. Metal-organic complexes also faced hurdles; for instance, Shibasaki's lanthanum-BINOL system required additional additives like Ph3AsO to boost performance, and Feng Xiaoming's scandium-based catalyst, despite low loading, still demanded 48 hours for completion. These historical constraints highlight a critical need for a catalyst system that balances high activity, short reaction times, and broad substrate tolerance without compromising on optical purity.
The Novel Approach
The methodology outlined in Patent CN111892559B fundamentally shifts the paradigm by employing a synergistic co-catalytic system comprising a chiral TADDOL ligand and a rare earth metal amide. This novel approach eliminates the need for the excessive catalyst quantities seen in organocatalytic routes, operating efficiently with metal amide loadings as low as 0.05 to 0.1 molar equivalents. The integration of an organic base, preferably 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU), further enhances the reaction kinetics, allowing the transformation to proceed to completion within 16 hours at temperatures as low as -30°C. This drastic reduction in reaction time compared to the multi-day protocols of the past translates directly into increased reactor turnover and improved energy efficiency. Moreover, the system demonstrates remarkable universality, accommodating a wide variety of substituents on the chalcone backbone, including halogens, alkyl groups, alkoxy groups, and even heteroaromatic rings like furyl and thienyl. The ability to consistently achieve yields near quantitative levels (99%) while maintaining high enantioselectivity positions this technology as a superior choice for cost reduction in chiral epoxide manufacturing, offering a streamlined path from raw materials to high-value intermediates.
Mechanistic Insights into Rare Earth-TADDOL Co-Catalysis
Understanding the mechanistic underpinnings of this catalytic cycle is essential for R&D teams aiming to optimize the process for specific substrates. The reaction initiates with a metathesis reaction between the ligand precursor, such as H2L2, and the rare earth metal amide RE[N(SiMe3)2]3 in solution. This 1:1 exchange generates an active metal-ligand complex that retains a silylamide group, which serves as a crucial reactive site. Upon introduction of tert-butyl hydroperoxide (TBHP), a ligand exchange occurs where the peroxy group coordinates with the rare earth metal center, displacing the silylamine and forming the true active catalyst species. This activation step is pivotal as it primes the metal center for oxygen transfer. Subsequently, the carbonyl oxygen of the alpha,beta-unsaturated ketone substrate coordinates to the Lewis acidic rare earth metal, orienting the double bond for nucleophilic attack by the activated peroxide. This coordination creates a rigid chiral environment dictated by the bulky TADDOL ligand framework, ensuring that the oxygen delivery occurs from a specific face of the alkene.
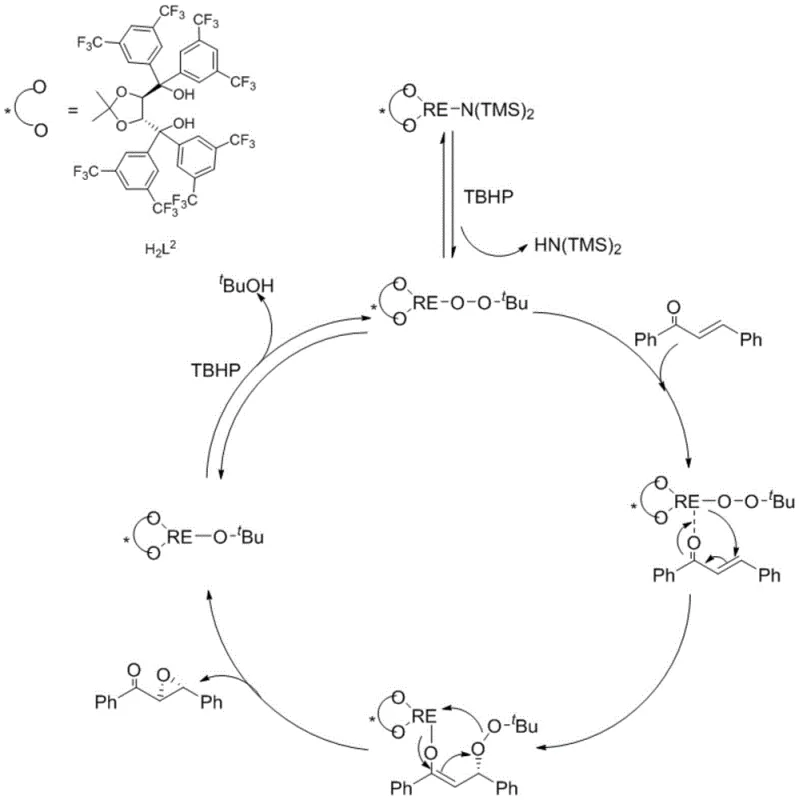
Following the oxygen transfer, a chiral enol-metal intermediate is formed, which then undergoes bond cleavage to release the chiral epoxy product and generate a tert-butoxy rare earth metal complex. The cycle is closed when this tert-butoxy species reacts with another molecule of TBHP, regenerating the active peroxy-rare earth catalyst and releasing tert-butanol as a byproduct. This elegant cycle explains the high turnover and efficiency observed in the experimental data. Crucially, the steric bulk of the TADDOL ligand, particularly variants like H2L2 featuring 3,5-bis(trifluoromethyl)phenyl groups, plays a dominant role in discriminating between the enantiotopic faces of the substrate. The electron-withdrawing nature of these substituents likely enhances the Lewis acidity of the metal center, facilitating stronger substrate binding and faster reaction rates. For process chemists, this mechanistic clarity offers opportunities for further refinement, such as adjusting the electronic properties of the ligand to accommodate more challenging electron-deficient or electron-rich substrates, thereby ensuring consistent quality and reducing lead time for high-purity pharmaceutical intermediates.
How to Synthesize Chiral Epoxy Chalcones Efficiently
Implementing this synthesis protocol requires strict adherence to anhydrous and oxygen-free conditions to preserve the integrity of the sensitive rare earth metal amide catalyst. The standard procedure involves dissolving the rare earth metal amide, typically Yb[N(SiMe3)2]3, in anhydrous acetonitrile under an inert argon atmosphere, followed by the addition of the chiral TADDOL ligand H2L2. An organic base additive, most effectively DBU, is introduced to facilitate the formation of the active catalytic species. Once the catalyst system is pre-formed, the chalcone substrate is added, and the mixture is cooled to the optimal temperature of -30°C before the slow addition of the TBHP oxidant solution in decane. The detailed standardized synthesis steps are provided below to ensure reproducibility and safety during scale-up operations.
- Prepare the catalyst system by mixing Yb[N(SiMe3)2]3 and chiral TADDOL ligand H2L2 in anhydrous acetonitrile under argon protection.
- Add the organic base additive DBU and stir at room temperature, then introduce the chalcone substrate and cool the mixture to -30°C.
- Slowly add tert-butyl hydroperoxide (TBHP) solution in decane, maintain stirring at -30°C for 16 hours, then quench and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this rare earth-TADDOL co-catalytic system offers transformative benefits that extend beyond mere chemical efficiency. The primary advantage lies in the significant optimization of resource utilization; by drastically lowering the required catalyst loading compared to legacy organocatalytic methods, manufacturers can achieve substantial cost savings in raw material procurement. The elimination of the need for massive excesses of chiral modifiers reduces the overall bill of materials, directly impacting the cost of goods sold. Furthermore, the shortened reaction timeline, compressing what used to be multi-day processes into a single 16-hour batch, enhances asset utilization rates, allowing existing reactor infrastructure to produce higher volumes without capital expenditure on new equipment. This efficiency gain is critical for maintaining competitive pricing in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The transition to this catalytic system removes the dependency on expensive transition metal scavengers often required to meet stringent residual metal limits in pharmaceuticals, as rare earth metals can be managed effectively through standard workup procedures like aqueous quenching and chromatography. Additionally, the high atom economy and near-quantitative yields minimize waste generation, reducing the costs associated with waste disposal and solvent recovery. The use of commercially available and relatively stable reagents like TBHP and DBU further stabilizes the supply chain against volatility in specialized reagent markets. By streamlining the purification process due to high crude purity, downstream processing costs are also significantly curtailed, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system across a wide array of substrates means that a single standardized protocol can be applied to produce a diverse portfolio of chiral epoxides, simplifying inventory management and reducing the complexity of production scheduling. The reliance on readily available rare earth metals and established ligand frameworks mitigates the risk of supply disruptions associated with exotic or proprietary catalysts. Moreover, the mild reaction conditions reduce the safety risks associated with high-pressure or high-temperature operations, ensuring uninterrupted production schedules and enhancing worker safety. This reliability is paramount for securing long-term contracts with multinational pharmaceutical companies that demand consistent quality and on-time delivery of critical intermediates.
- Scalability and Environmental Compliance: The protocol's compatibility with standard organic solvents like acetonitrile and decane facilitates straightforward scale-up from laboratory gram-scale to multi-ton commercial production without the need for specialized high-pressure reactors. The reduced reaction time and lower energy requirements for cooling (only -30°C rather than cryogenic temperatures) align with green chemistry principles, lowering the carbon footprint of the manufacturing process. The high selectivity of the reaction minimizes the formation of byproducts, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. This sustainability profile not only meets corporate social responsibility goals but also future-proofs the supply chain against evolving regulatory landscapes regarding chemical manufacturing emissions and waste.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology, derived from the detailed experimental data and background analysis provided in the patent documentation. These insights are intended to clarify the operational parameters and potential applications for partners considering the adoption of this synthetic route. Understanding these nuances is vital for assessing the feasibility of integrating this method into existing production workflows.
Q: What are the optimal reaction conditions for this asymmetric epoxidation?
A: The patent specifies that optimal results are achieved using Yb[N(SiMe3)2]3 as the rare earth source, H2L2 as the chiral ligand, and DBU as the additive in acetonitrile at -30°C.
Q: How does this method compare to traditional organocatalysts in terms of efficiency?
A: Unlike earlier organocatalytic methods that required high catalyst loadings (up to 30 mol%) and long reaction times (up to 144 hours), this co-catalytic system achieves high yields (up to 99%) and enantioselectivity (up to 94% ee) with significantly lower loading and shorter duration.
Q: Is this catalytic system suitable for a wide range of substrates?
A: Yes, the methodology demonstrates broad substrate applicability, successfully epoxidizing various chalcones substituted with electron-donating or electron-withdrawing groups, as well as heteroaryl derivatives like thiophene and furan.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Epoxy Chalcones Supplier
The technological breakthroughs detailed in Patent CN111892559B underscore the immense potential of rare earth-TADDOL co-catalysis in revolutionizing the production of chiral intermediates. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that this sophisticated chemistry can be translated into reliable industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of chiral epoxy chalcones meets the exacting standards required for pharmaceutical synthesis. We understand that consistency is key, and our process engineering team is dedicated to optimizing every variable, from solvent recovery to catalyst recycling, to deliver a product of unmatched quality.
We invite you to collaborate with us to leverage this advanced catalytic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can lower your overall production costs. Please contact us to request specific COA data for our chiral epoxide portfolio and to discuss route feasibility assessments for your target molecules. Together, we can accelerate your drug development timeline with a supply chain partner committed to innovation, quality, and commercial excellence.
