Advanced Palladium-Catalyzed Synthesis of Sulfonyl Ketoamides for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex molecular scaffolds efficiently. Patent CN111187184A introduces a groundbreaking approach for the synthesis of sulfonyl ketoamide derivatives, a structural motif prevalent in numerous bioactive compounds. This innovation addresses long-standing synthetic challenges by utilizing a palladium-catalyzed carbonylation strategy that couples 1,3-dicarbonyl compounds with organic sulfonyl azides under a carbon monoxide atmosphere. The significance of this technology is underscored by the presence of ketoamide skeletons in critical therapeutic agents such as Teriflunomide, Piroxicam, and Lymecycline, which are essential for treating conditions ranging from multiple sclerosis to rheumatoid arthritis. 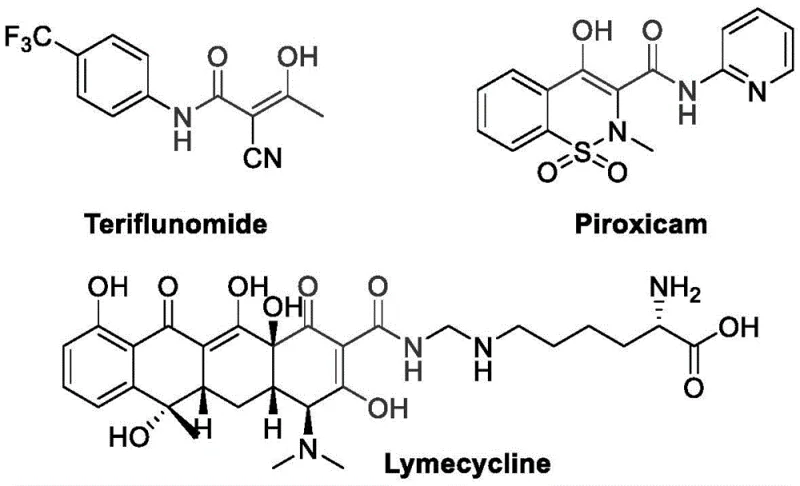
For procurement managers and supply chain directors, this patent represents a pivotal shift towards more reliable pharmaceutical intermediate supplier capabilities. By replacing hazardous and unstable reagents with accessible starting materials, the process not only enhances safety profiles but also streamlines the supply chain for high-purity sulfonyl ketoamides. The method eliminates the need for specialized handling of isocyanates, thereby reducing logistical complexities and potential downtime in manufacturing facilities. This technological leap ensures a more consistent supply of critical intermediates, directly supporting the continuity of downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonyl ketoamides has been hindered by the reliance on sulfonyl isocyanates as key electrophilic partners. These reagents are notoriously unstable, prone to decomposition upon exposure to moisture, and often require stringent storage conditions that inflate inventory costs. Furthermore, the preparation of sulfonyl isocyanates typically involves harsh reaction conditions and toxic phosgene equivalents, posing significant environmental and safety risks in an industrial setting. The narrow substrate scope associated with traditional nucleophilic substitution reactions limits the structural diversity achievable, forcing chemists to develop bespoke, low-yielding routes for substituted analogs. Consequently, the conventional pathway often results in inconsistent batch quality and elevated production costs due to extensive purification requirements needed to remove isocyanate-derived impurities.
The Novel Approach
In stark contrast, the methodology disclosed in CN111187184A leverages the stability and commercial availability of sulfonyl azides combined with carbon monoxide insertion. This strategy bypasses the isolation of reactive isocyanate intermediates entirely, generating them in situ under mild catalytic conditions. The reaction demonstrates exceptional tolerance for various functional groups, allowing for the efficient synthesis of diverse derivatives including those with electron-donating and electron-withdrawing substituents. 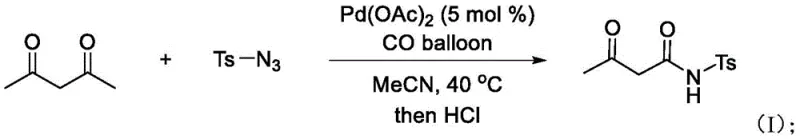
By employing a simple palladium catalyst system in common solvents like acetonitrile, the process achieves high conversion rates with minimal byproduct formation. This novel approach not only simplifies the operational workflow but also aligns with green chemistry principles by avoiding toxic reagents and reducing waste generation. For manufacturers, this translates to a drastic simplification of the production process, enabling cost reduction in API manufacturing through improved atom economy and reduced waste disposal expenses.
Mechanistic Insights into Pd-Catalyzed Carbonylative Amidation
The core of this transformative synthesis lies in the unique catalytic cycle mediated by palladium species. The reaction initiates with the activation of the sulfonyl azide by the palladium catalyst, likely forming a reactive palladium-nitrene intermediate. This species subsequently undergoes insertion of carbon monoxide to generate a transient palladium-isocyanate complex in situ. Unlike free isocyanates which are unstable, this metal-bound intermediate is sufficiently stabilized to undergo selective nucleophilic attack by the enol form of the 1,3-dicarbonyl compound. The final step involves protonolysis or reductive elimination to release the desired sulfonyl ketoamide product and regenerate the active palladium catalyst for the next turnover.
Understanding this mechanism is crucial for R&D directors focused on impurity control and process optimization. The specificity of the palladium center ensures that side reactions, such as the Curtius rearrangement or azide decomposition, are minimized. Experimental data confirms that the only significant byproduct is benzenesulfonamide, which arises from the hydrolysis of excess sulfonyl azide and is easily removed during aqueous workup. 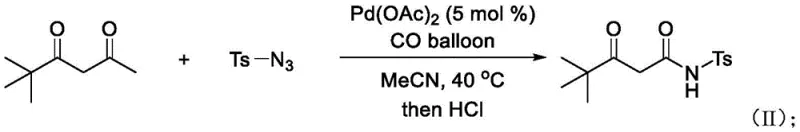
This high level of chemoselectivity allows for the synthesis of complex intermediates without the need for protecting group strategies. The ability to tune the electronic properties of the catalyst and ligands further offers opportunities to optimize reaction rates and yields for specific substrates, ensuring that the process remains robust across different batches and scales.
How to Synthesize 3-oxo-N-tosylbutyramide Efficiently
The practical implementation of this chemistry is straightforward, requiring standard laboratory equipment and readily available reagents. The protocol typically involves charging a reaction vessel with the 1,3-dicarbonyl substrate, the sulfonyl azide, and a catalytic amount of palladium acetate in acetonitrile. The system is then pressurized with carbon monoxide, often using a simple balloon setup for small-scale trials, and heated to moderate temperatures ranging from 40°C to 80°C. Detailed standardized synthesis steps for scaling this reaction are provided below.
- Charge a reaction vessel with 1,3-dicarbonyl compound (e.g., acetylacetone), organic sulfonyl azide (e.g., p-toluenesulfonylazide), and palladium acetate catalyst (5 mol%) in acetonitrile solvent.
- Introduce carbon monoxide (CO) gas into the system using a balloon and heat the mixture to 40-80°C for 8-18 hours while monitoring reaction progress via TLC.
- Upon completion, add dilute hydrochloric acid, extract the organic layer with ethyl acetate, remove solvent under reduced pressure, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits that extend beyond mere chemical novelty. For procurement managers, the shift away from unstable isocyanates to stable azides mitigates supply chain risks associated with reagent degradation and specialized transport requirements. The use of commodity chemicals like acetylacetone and tosyl azide ensures a steady flow of raw materials, preventing bottlenecks that often plague specialty chemical sourcing. This reliability is paramount for maintaining continuous manufacturing schedules and meeting tight delivery windows for downstream clients.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous isocyanate precursors directly lowers the bill of materials. Furthermore, the mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure alternatives. The simplified workup procedure, which avoids complex chromatographic separations in favor of standard extraction and crystallization, significantly reduces labor and solvent costs. These factors collectively contribute to a more economically viable production model, allowing for competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By utilizing a catalytic system that tolerates a wide range of substrates, manufacturers can rapidly adapt to changing demand for different ketoamide derivatives without retooling entire production lines. The robustness of the palladium catalyst ensures consistent performance over extended periods, minimizing batch failures and rework. This operational stability enhances the overall reliability of the supply chain, providing customers with confidence in the continuity of their raw material supply for critical drug formulations.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with standard solvents and atmospheric CO pressure. The absence of toxic heavy metal waste streams, aside from trace palladium which can be scavenged, simplifies environmental compliance and waste treatment. This alignment with green chemistry standards not only reduces regulatory burdens but also enhances the corporate sustainability profile of manufacturers adopting this technology, appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfonyl ketoamide synthesis platform. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity for technical teams evaluating this route for adoption.
Q: Why is the sulfonyl azide route preferred over traditional sulfonyl isocyanate methods?
A: Traditional methods rely on sulfonyl isocyanates, which are unstable, difficult to store, and require harsh preparation conditions. The novel azide-based route utilizes stable, commercially available sulfonyl azides and mild CO insertion, significantly improving safety and substrate scope.
Q: What represents the optimal catalyst system for this transformation?
A: Experimental data indicates that Palladium(II) acetate (Pd(OAc)2) is the superior catalyst, achieving yields up to 87% in acetonitrile. Other transition metals like Cobalt, Copper, or Rhodium showed significantly lower activity or failed to produce the target sulfonyl ketoamide.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process operates under mild temperatures (40-80°C) and atmospheric CO pressure using standard solvents like acetonitrile. The simple workup involving acid quenching and extraction makes it highly amenable to commercial scale-up without complex purification hurdles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonyl Ketoamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of sulfonyl ketoamide delivered meets the highest industry standards for quality and consistency.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific supply chain needs. By leveraging our expertise in process development, we can provide a Customized Cost-Saving Analysis tailored to your production volumes. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global marketplace.
