Advanced Photocatalytic Synthesis of 3-Alkylquinoxalinone Derivatives for Commercial Scale-Up
Introduction to Next-Generation Quinoxalinone Synthesis
The field of medicinal chemistry continuously seeks efficient pathways to access bioactive heterocyclic scaffolds, among which the quinoxalinone core stands out as a privileged structure. As detailed in the groundbreaking patent CN111635370A, a novel photocatalytic preparation method for 3-alkylquinoxalinone derivatives has been developed, addressing critical limitations in existing synthetic methodologies. These derivatives are not merely academic curiosities; they serve as potent pharmacophores in the design of antitumor agents, antibacterial compounds, and HIV-I reverse transcriptase inhibitors, as illustrated by their structural relevance to Alose reductase inhibitors and MDR antagonists.
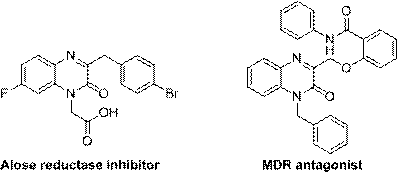
The significance of this technological advancement lies in its ability to functionalize the C3 position of the quinoxalinone ring through a direct, green, and atom-economical process. By leveraging visible light irradiation, this method circumvents the need for hazardous oxidants and extreme thermal conditions that have historically plagued the synthesis of these valuable intermediates. For R&D directors and process chemists, this represents a paradigm shift towards sustainable manufacturing, offering a robust platform for generating diverse libraries of high-purity pharmaceutical intermediates with exceptional selectivity and yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-alkylquinoxalinone derivatives has relied heavily on strategies that impose significant burdens on both operational safety and environmental compliance. Traditional approaches often necessitate the use of stoichiometric amounts of strong oxidants, such as peroxides, or require expensive and potentially toxic transition metal catalysts to drive the C-H functionalization. These conventional routes frequently suffer from poor atom economy, generating substantial quantities of chemical waste that complicate downstream purification and increase the overall cost of goods. Furthermore, the harsh reaction conditions associated with these methods, including elevated temperatures and aggressive reagents, can lead to substrate decomposition and the formation of complex impurity profiles, thereby compromising the purity required for pharmaceutical applications.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN111635370A introduces a sophisticated three-component tandem reaction driven by visible light photocatalysis. This innovative approach utilizes substituted quinoxalinones, substituted alkenes, and N-alkanoyloxyphthalimides as readily available starting materials, reacting them under mild nitrogen-protected conditions. The use of clean visible light as the energy source eliminates the thermal stress on sensitive functional groups, while the photocatalytic cycle ensures high efficiency with minimal catalyst loading. As depicted in the general reaction scheme below, this strategy enables the direct installation of alkyl groups at the C3 position with remarkable precision.
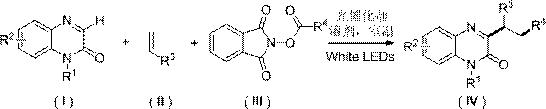
This novel pathway not only simplifies the operational workflow by removing the need for external oxidants but also significantly enhances the sustainability profile of the manufacturing process. The compatibility of this method with a wide range of substrates, including those bearing halogen, alkoxy, and amino substituents, underscores its versatility for producing complex molecular architectures. For procurement and supply chain teams, this translates to a more reliable sourcing strategy, as the reliance on hazardous reagents is minimized, and the use of common organic solvents like DMSO or THF streamlines the logistics of raw material acquisition.
Mechanistic Insights into Photocatalytic C3 Alkylation
The mechanistic underpinning of this transformation involves a sophisticated interplay of photoredox catalysis and radical chemistry, which is crucial for R&D professionals to understand for process optimization. Upon irradiation with visible light, the photocatalyst, such as Ir(ppy)3 or Eosin Y, enters an excited state capable of engaging in single-electron transfer (SET) processes. The N-alkanoyloxyphthalimide acts as a radical precursor, undergoing reductive cleavage to generate an alkyl radical species alongside a phthalimide anion. This decarboxylative radical generation is the key driving force that allows for the mild activation of the alkylating agent without the need for harsh initiators.
Subsequently, the generated alkyl radical adds across the double bond of the substituted alkene, forming a new carbon-centered radical intermediate. This intermediate then attacks the electron-deficient C3 position of the quinoxalinone ring, facilitated by the presence of an acidic additive like trifluoroacetic acid which likely protonates the substrate to enhance its electrophilicity. The final step involves rearomatization and proton loss to yield the stable 3-alkylquinoxalinone product. This intricate cascade ensures high regioselectivity, minimizing the formation of side products and simplifying the purification process. The ability to control this radical pathway through the choice of light wavelength and catalyst loading provides a powerful handle for tuning the reaction outcome.
How to Synthesize 3-Alkylquinoxalinone Derivatives Efficiently
Implementing this photocatalytic protocol requires careful attention to reaction parameters to maximize yield and purity, particularly when scaling from milligram to kilogram quantities. The standard procedure involves charging a reaction vessel with the quinoxalinone substrate, the alkene partner, and the N-alkanoyloxyphthalimide precursor in a specific molar ratio, typically favoring a slight excess of the alkene and radical precursor to drive the reaction to completion. The addition of a suitable photocatalyst, such as Ir(ppy)3 at a loading of 0.03 equivalents, and an acid additive is critical for initiating the catalytic cycle. Detailed standardized synthesis steps are provided in the guide below.
- Combine substituted quinoxalinone, substituted alkene, and N-alkanoyloxyphthalimide in a reaction vessel with photocatalyst and additive.
- Add organic solvent such as DMSO or THF and purge the system with nitrogen to create an inert atmosphere.
- Irradiate the mixture with visible light LEDs at room temperature for 6-10 hours, followed by extraction and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this photocatalytic technology offers profound benefits that extend beyond mere chemical elegance, directly impacting the bottom line and supply chain resilience. The elimination of stoichiometric oxidants and the reduction in catalyst loading significantly lower the raw material costs associated with production. Moreover, the mild reaction conditions reduce energy consumption, as there is no need for extensive heating or cooling systems, leading to substantial operational expenditure savings. The simplified workup procedure, which avoids complex quenching steps for hazardous reagents, further accelerates the manufacturing timeline.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and hazardous oxidants drastically reduces the cost of goods sold. By utilizing visible light as a traceless reagent, the process minimizes waste disposal costs and avoids the expensive purification steps often required to remove heavy metal residues from pharmaceutical intermediates. This lean manufacturing approach ensures that the final product is not only cost-effective but also meets stringent regulatory standards for residual impurities without additional processing.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including substituted quinoxalinones and simple alkenes, are commodity chemicals that are widely available from multiple global suppliers. This diversity in sourcing mitigates the risk of supply disruptions that often plague specialized reagent-dependent processes. Furthermore, the stability of the photocatalysts and the robustness of the reaction conditions mean that production can be maintained consistently, ensuring reliable delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The use of LED light sources makes this process inherently scalable, as photochemical reactors can be easily modularized to increase throughput without compromising reaction efficiency. The green nature of the process, characterized by low waste generation and the absence of toxic byproducts, aligns perfectly with modern environmental regulations. This compliance reduces the regulatory burden on manufacturing sites and facilitates smoother audits, making it an ideal candidate for long-term sustainable production strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this photocatalytic method, derived directly from the patent specifications and experimental data. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific conditions and outcomes reported in the intellectual property documentation.
Q: What are the primary advantages of this photocatalytic method over traditional oxidation methods?
A: This method utilizes clean visible light energy instead of harsh chemical oxidants or expensive transition metal catalysts, resulting in milder reaction conditions, reduced waste generation, and improved atom economy.
Q: Which photocatalysts are most effective for this transformation according to the patent data?
A: The patent highlights Ir(ppy)3 as the preferred photocatalyst, though organic dyes like Eosin Y and Rose Bengal, as well as Ru(bpy)3Cl2, are also effective depending on the specific substrate requirements.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process operates at room temperature with simple LED light sources and readily available raw materials, making it highly scalable and operationally convenient for industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkylquinoxalinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photocatalytic technologies in modern pharmaceutical synthesis. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN111635370A can be seamlessly translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-alkylquinoxalinone derivatives meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for high-purity pharmaceutical intermediates.
