Advanced Manufacturing of Spirotryprostatin A via Copper-Catalyzed Cyclization
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spirocyclic oxindole natural products like Spirotryprostatin A has been plagued by significant inefficiencies that hinder commercial viability. Traditional methodologies often rely heavily on noble metal catalysts, particularly palladium, which not only inflate the raw material costs but also introduce complex regulatory hurdles regarding heavy metal residue limits in final pharmaceutical products. Furthermore, conventional routes frequently suffer from excessive step counts, leading to cumulative yield losses that make large-scale production economically unfeasible. The reliance on indole or oxindole starting materials in older strategies severely restricts structural diversity, limiting the ability of medicinal chemists to explore analog libraries effectively. These legacy processes often require harsh reaction conditions that compromise the integrity of sensitive functional groups, resulting in difficult-to-remove impurities that complicate downstream purification. Consequently, the supply chain for these critical intermediates has remained fragile, with long lead times and inconsistent quality posing risks to drug development timelines.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN115677711A introduces a transformative strategy that leverages inexpensive cuprous iodide (CuI) as the primary catalyst, fundamentally altering the cost structure of Spirotryprostatin A manufacturing. This innovative route utilizes readily available o-iodoaniline derivatives as starting materials, bypassing the limitations of pre-functionalized indole scaffolds and enabling greater flexibility in molecular design. The process is characterized by its exceptional atom economy and high product yields, achieved through a streamlined sequence that minimizes waste generation and energy consumption. By employing mild reaction conditions, such as heating at 70°C in tetrahydrofuran, the method ensures operational safety and ease of scale-up without compromising stereochemical integrity. The integration of a tandem reaction sequence allows for the rapid construction of the complex chiral quaternary carbon center, a critical structural motif, in a single operational step. This holistic improvement in synthetic efficiency positions the new method as a superior choice for reliable pharmaceutical intermediate supplier networks seeking robust and sustainable manufacturing solutions.
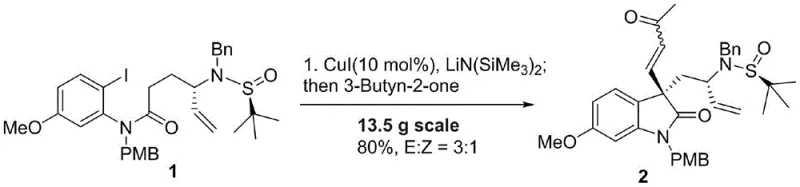
Mechanistic Insights into CuI-Catalyzed Cyclization
The cornerstone of this advanced synthesis lies in the initial copper-catalyzed cascade reaction, which elegantly constructs the core oxindole skeleton with high precision. The mechanism initiates with the activation of the o-iodoaniline substrate by the CuI catalyst in the presence of lithium bistrimethylsilylamide, generating a reactive organometallic species in situ. Upon the addition of 3-butyn-2-one, a sophisticated tandem cyclization occurs, driven by the nucleophilic attack of the nitrogen atom onto the activated alkyne moiety. This process is meticulously controlled to favor the formation of the desired E-isomer over the Z-isomer, although both can be managed through careful chromatographic separation. The use of LiN(SiMe3)2 as a base is critical, as it facilitates the deprotonation steps necessary for ring closure while maintaining compatibility with the sensitive sulfinyl auxiliary. This catalytic cycle exemplifies high atom economy, as the majority of atoms from the starting materials are incorporated into the final product structure, minimizing the formation of stoichiometric byproducts. The robustness of this copper-mediated transformation ensures consistent reproducibility, a vital factor for maintaining batch-to-batch consistency in commercial API intermediate production.
Following the initial cyclization, the formation of the spiro-center is achieved through a carefully orchestrated acid-mediated cascade that demonstrates exquisite chemoselectivity. The treatment with hydrochloric acid triggers the removal of the sulfinyl protecting group, simultaneously activating the molecule for an intramolecular aza-Michael addition. This dual-functionality of the acid reagent simplifies the process by combining deprotection and cyclization into a single unit operation, thereby reducing processing time and solvent usage. The subsequent steps involve strategic functional group manipulations, such as the removal of the p-methoxybenzyl (PMB) group using trifluoromethanesulfonic acid, which proceeds cleanly without affecting the newly formed spiro-junction. Oxidative cleavage of the terminal alkene using osmium tetroxide and sodium periodate is another critical juncture, where the reaction conditions are tuned to prevent over-oxidation of the indole nitrogen. Each transformation is designed to preserve the stereochemical information established in the early stages, ensuring that the final natural product retains its biological activity. This level of mechanistic control is essential for producing high-purity pharmaceutical intermediates that meet the rigorous standards of global regulatory bodies.
How to Synthesize Spirotryprostatin A Efficiently
The execution of this synthesis requires strict adherence to the optimized parameters outlined in the patent to maximize yield and purity at every stage. Operators must ensure that the initial copper-catalyzed step is performed under an inert atmosphere, typically high-purity nitrogen, to prevent oxidation of the sensitive organometallic intermediates. Temperature control is paramount, particularly during the addition of the acetylene ketone, where cooling to 0°C helps regulate the exothermic nature of the reaction and improves stereoselectivity. Workup procedures involve standard aqueous quenches followed by extraction with ethyl acetate, allowing for the efficient recovery of the product from the reaction mixture. Purification is primarily achieved through silica gel column chromatography, utilizing gradient elution systems of petroleum ether and ethyl acetate to separate closely related isomers and byproducts. For the later stages involving hydrogenation and Grignard reactions, specialized equipment such as hydrogen balloons and cryogenic cooling baths are necessary to maintain safety and reaction efficacy. Detailed standardized synthesis steps are provided below to guide process engineers in implementing this route effectively.
- Mix o-iodoaniline compounds with CuI and LiN(SiMe3)2 in THF, heat to 70°C, then add 3-butyn-2-one to form the chiral oxindole.
- Treat the oxindole with 1mol/L HCl solution to remove the sulfinyl group and induce aza-Michael addition, forming the spiro-cycle.
- Remove the PMB protecting group using trifluoromethanesulfonic acid, followed by oxidative cleavage of the alkene using OsO4/NMO and NaIO4.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this novel synthesis route offers compelling advantages that directly address the pain points of cost volatility and supply chain fragility in the fine chemical sector. The displacement of precious metal catalysts with base metal alternatives creates a substantial buffer against market fluctuations in palladium prices, stabilizing the cost of goods sold over the long term. Furthermore, the simplified operational profile reduces the burden on quality control laboratories, as fewer heavy metal tests are required for release, accelerating the time from production batch to shipment. The use of commodity chemicals and solvents ensures that raw material sourcing remains resilient, even during periods of global supply chain disruption. This robustness translates into enhanced reliability for downstream pharmaceutical manufacturers who depend on uninterrupted flows of critical intermediates for their own production schedules. By adopting this technology, companies can secure a competitive edge through lower manufacturing costs and improved supply security.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts represents a direct and significant reduction in raw material expenditure, which is a primary driver of overall manufacturing costs. Additionally, the high atom economy of the reaction sequence means that less starting material is wasted, further driving down the effective cost per kilogram of the final product. The streamlined nature of the synthesis reduces the number of isolation and purification steps, which in turn lowers labor costs and utility consumption associated with extended processing times. Solvent recovery becomes more efficient due to the reduced complexity of the waste stream, contributing to additional operational savings. These cumulative financial benefits allow for a more aggressive pricing strategy in the marketplace while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: Sourcing o-iodoaniline derivatives and cuprous iodide is significantly less risky than procuring specialized palladium complexes, as these materials are produced by a wide network of global chemical suppliers. This diversification of the supply base mitigates the risk of single-source failures and ensures continuity of supply even if one vendor faces production issues. The mild reaction conditions reduce the likelihood of unplanned shutdowns due to equipment failure or safety incidents, fostering a more stable production environment. Shorter synthesis cycles mean that inventory turnover is faster, allowing the supply chain to respond more敏捷 ly to changes in customer demand. This agility is crucial for maintaining service levels in a dynamic market where lead times are often a key differentiator between suppliers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily transferable from laboratory glassware to industrial reactors without requiring exotic engineering solutions. The avoidance of highly toxic reagents and the use of greener catalytic systems align with increasingly stringent environmental regulations, reducing the compliance burden on the manufacturing facility. Waste generation is minimized through high-yield transformations, lowering the costs associated with hazardous waste disposal and treatment. Energy consumption is optimized by operating at moderate temperatures, contributing to a lower carbon footprint for the manufacturing process. These sustainability credentials are becoming increasingly important for pharmaceutical customers who are under pressure to reduce the environmental impact of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the experimental data and technical specifications detailed in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is critical for assessing the feasibility of integrating this route into existing manufacturing portfolios. The responses cover aspects ranging from catalyst handling to final product specification, offering a comprehensive overview of the process capabilities.
Q: How does the CuI catalytic system improve cost efficiency compared to traditional Palladium methods?
A: The patented method replaces expensive noble metal palladium catalysts with inexpensive cuprous iodide (CuI). This substitution drastically reduces raw material costs and eliminates the need for complex heavy metal removal processes typically required for Pd residues, leading to significant downstream purification savings.
Q: What are the scalability advantages of this synthesis route for commercial production?
A: The process operates under mild reaction conditions, primarily at 70°C for the key cyclization step, avoiding extreme cryogenic temperatures for most stages. The use of common solvents like THF and DCM, combined with standard workup procedures like silica gel chromatography, ensures the route is highly adaptable for large-scale manufacturing from kilograms to tons.
Q: How is high purity maintained throughout the multi-step synthesis?
A: High purity is achieved through a combination of high atom economy reactions and rigorous purification protocols at each stage. The specific use of selective reagents like trifluoromethanesulfonic acid for deprotection and controlled oxidative cleavage minimizes side-product formation, ensuring the final API intermediate meets stringent quality specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirotryprostatin A Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of novel therapeutics, and we are committed to delivering excellence in every batch. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical trials to full-scale market launch. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every shipment meets your exact requirements. Our expertise in copper-catalyzed processes allows us to optimize this specific route further, potentially unlocking even greater efficiencies and cost savings for your specific application. We view ourselves not just as a vendor, but as a strategic partner dedicated to supporting your long-term growth and success in the competitive pharmaceutical landscape.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits of switching to this superior manufacturing route. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will empower you to make informed decisions for your supply chain. Let us collaborate to bring your next breakthrough therapy to market faster and more efficiently, leveraging our technical prowess and commitment to quality.
