Advanced Synthesis of 1,2-Diiodo Olefins: A Cost-Effective Route for Pharmaceutical Intermediates
Advanced Synthesis of 1,2-Diiodo Olefins: A Cost-Effective Route for Pharmaceutical Intermediates
The landscape of organic synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign methodologies for constructing complex molecular architectures. A significant breakthrough in this domain is detailed in patent CN107501024B, which discloses a novel synthesis method for 1,2-diiodo olefin compounds. These compounds serve as critical synthons in the construction of carbon-carbon, carbon-oxygen, and carbon-nitrogen bonds, making them indispensable intermediates in the production of active pharmaceutical ingredients (APIs) and advanced agrochemicals. The patented process utilizes a unique combination of zinc diiodide and tert-butyl nitrite (TBN) to facilitate the direct iodination of phenylacetylene derivatives, offering a robust alternative to traditional halogenation strategies that often suffer from high costs and operational complexity.
For R&D directors and process chemists, the ability to access high-purity 1,2-diiodo olefins through a streamlined protocol represents a substantial opportunity to optimize synthetic routes. The method described in the patent not only achieves high yields but also demonstrates exceptional tolerance for a wide variety of functional groups, thereby expanding the chemical space available for drug discovery and development. By leveraging this technology, manufacturers can significantly reduce the lead time for high-purity pharmaceutical intermediates, ensuring a more agile response to market demands while maintaining rigorous quality standards essential for regulatory compliance in the global pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2-diiodo olefin compounds has relied heavily on direct iodination strategies using reagents such as N-iodosuccinimide (NIS), elemental iodine, or various iodide salts coupled with strong oxidizing agents. While these methods are well-documented in academic literature, they present significant challenges when translated to industrial-scale manufacturing. The use of NIS, for instance, introduces succinimide byproducts that can be difficult to remove, complicating the purification process and potentially impacting the purity profile of the final API. Furthermore, reactions involving elemental iodine often require harsh conditions or stoichiometric amounts of oxidants, leading to poor atom economy and generating substantial quantities of hazardous waste that necessitate costly disposal procedures.
From a supply chain perspective, the reliance on expensive specialty reagents like NIS creates vulnerability in the procurement process, as price volatility for such chemicals can erode profit margins. Additionally, the operational difficulties associated with controlling exothermic iodination reactions and managing corrosive byproducts pose safety risks that require specialized equipment and rigorous safety protocols. These factors collectively contribute to higher manufacturing costs and longer production cycles, making conventional methods less attractive for the commercial scale-up of complex pharmaceutical intermediates where efficiency and cost control are paramount.
The Novel Approach
The innovative methodology outlined in patent CN107501024B addresses these痛点 by employing a catalytic system based on zinc diiodide and tert-butyl nitrite. This approach facilitates the direct conversion of phenylacetylene substrates into the corresponding 1,2-diiodo olefins under remarkably mild conditions, often at room temperature or slightly elevated temperatures up to 100°C. The use of zinc diiodide, a commercially abundant and inexpensive Lewis acid, combined with TBN as a radical initiator or oxidant source, creates a highly efficient reaction environment that minimizes side reactions and maximizes product yield.
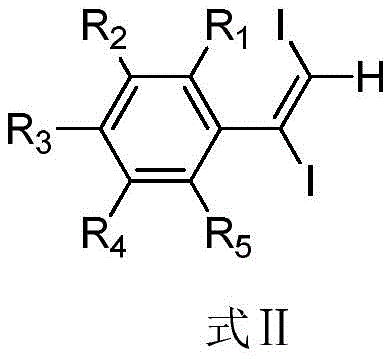
This novel route eliminates the need for expensive iodinating agents like NIS, replacing them with cost-effective commodity chemicals that are readily available in bulk quantities. The reaction proceeds smoothly in common organic solvents such as dichloromethane or chloroform, and the workup procedure is straightforward, involving a simple aqueous quench and extraction. For procurement managers, this translates to a drastic simplification of the raw material list and a significant reduction in the cost of goods sold (COGS). Moreover, the mild reaction conditions enhance process safety, reducing the energy consumption associated with heating or cooling and lowering the overall environmental footprint of the manufacturing process.
Mechanistic Insights into ZnI2/TBN-Mediated Iodination
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to implement this technology effectively. The reaction likely proceeds through a radical-mediated pathway initiated by the decomposition of tert-butyl nitrite, which generates reactive nitrogen oxide species and tert-butoxy radicals. These radicals interact with the zinc diiodide species to generate electrophilic iodine equivalents in situ, which then attack the electron-rich triple bond of the phenylacetylene substrate. The presence of zinc ions may also serve to activate the alkyne through coordination, increasing its susceptibility to nucleophilic or electrophilic attack depending on the specific electronic nature of the substituents.
The regioselectivity and stereoselectivity observed in the formation of the 1,2-diiodo olefin suggest a highly controlled addition process. The steric and electronic properties of the substituents on the phenyl ring (R1-R5) play a pivotal role in directing the outcome of the reaction. Electron-donating groups such as methoxy or alkyl chains can stabilize intermediate carbocations or radical species, potentially accelerating the reaction rate, while electron-withdrawing groups like halogens or nitro groups may require slightly elevated temperatures to achieve full conversion, as evidenced by the examples in the patent where reactions were run at 100°C for substrates like 4-fluorophenylacetylene.
From an impurity control standpoint, the mechanism favors the formation of the desired diiodo product with minimal formation of mono-iodinated or over-oxidized byproducts. The use of a stoichiometric excess of TBN ensures complete conversion of the alkyne, while the specific reactivity of the ZnI2/TBN system suppresses competing pathways that often plague traditional iodination methods. This high level of chemoselectivity is particularly valuable for complex molecules containing sensitive functional groups, as it allows for late-stage functionalization without the need for extensive protecting group strategies, thereby streamlining the overall synthetic sequence.
How to Synthesize 1,2-Diiodo Olefin Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The general procedure involves dissolving the reagents in a dry solvent under an inert atmosphere to prevent moisture interference, followed by the addition of the substrate and stirring until completion. The simplicity of the protocol means that it can be adapted to existing reactor setups with minimal modification, facilitating rapid technology transfer. Detailed standardized synthesis steps are provided below to guide process engineers in optimizing the parameters for specific substrates.
- Dissolve zinc diiodide and tert-butyl nitrite in a dry solvent such as trichloromethane under a nitrogen or argon atmosphere.
- Add the phenylacetylene reaction substrate to the solution and stir at a temperature between 25°C and 100°C until TLC indicates completion.
- Quench the reaction with sodium thiosulfate solution, extract with dichloromethane, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ZnI2/TBN-mediated synthesis offers compelling economic and logistical benefits. The primary advantage lies in the substitution of high-cost specialty reagents with inexpensive, commodity-grade chemicals. Zinc diiodide and tert-butyl nitrite are widely produced and available from multiple global suppliers, mitigating the risk of supply disruptions and price spikes that are common with niche reagents like N-iodosuccinimide. This shift in raw material strategy directly contributes to cost reduction in pharmaceutical intermediate manufacturing, allowing companies to maintain competitive pricing in a crowded market.
- Cost Reduction in Manufacturing: The elimination of expensive iodinating agents and the reduction in waste disposal costs associated with hazardous oxidants lead to substantial savings. The simplified workup procedure, which avoids complex chromatographic separations often required to remove succinimide byproducts, further reduces solvent consumption and labor hours. By streamlining the production process, manufacturers can achieve a lower cost per kilogram of the final product, enhancing overall profitability and enabling more aggressive pricing strategies for downstream customers.
- Enhanced Supply Chain Reliability: The use of readily available reagents ensures a stable and continuous supply of raw materials, which is critical for meeting tight production schedules. The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvents, provides flexibility in sourcing and logistics. This reliability reduces the lead time for high-purity pharmaceutical intermediates, allowing supply chain managers to respond more quickly to fluctuations in demand and minimize inventory holding costs.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of common solvents make this process highly scalable from gram to multi-ton quantities without significant re-engineering. Furthermore, the improved atom economy and reduced generation of hazardous waste align with increasingly stringent environmental regulations. This compliance not only avoids potential fines but also enhances the company's reputation as a sustainable manufacturer, a factor that is becoming increasingly important for multinational corporations seeking responsible partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 1,2-diiodo olefins using this patented method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals evaluating this technology for their own processes.
Q: What are the optimal reaction conditions for this synthesis?
A: The reaction is typically conducted in a dry solvent like trichloromethane under an inert nitrogen atmosphere, with temperatures ranging from room temperature to 100°C depending on the substrate reactivity.
Q: What is the substrate scope for this iodination method?
A: The method supports a wide range of phenylacetylene substrates, including those substituted with alkyl, alkoxy, halogen, nitro, or aryl groups on the benzene ring.
Q: How is the product purified after the reaction?
A: After quenching with sodium thiosulfate and extraction, the crude product is purified using standard silica gel column chromatography to obtain high-purity 1,2-diiodo olefin compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Diiodo Olefin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team of expert chemists has extensively evaluated the synthesis method described in CN107501024B and has successfully integrated it into our manufacturing portfolio. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of 1,2-diiodo olefin meets the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecular targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for maximum efficiency and cost-effectiveness.
