Advanced Photochemical Synthesis of Polysubstituted Indanone Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for synthesizing complex molecular scaffolds, particularly those with significant biological activity. Patent CN111807942B introduces a groundbreaking preparation method for polysubstituted indanone derivatives, a class of compounds renowned for their diverse pharmacological properties including anti-inflammatory, antiviral, and anticancer activities. This innovation represents a paradigm shift from traditional transition metal-catalyzed processes to a more environmentally benign photochemical approach. By leveraging ultraviolet illumination to drive the cyclization of aryl aldehydes and alkyne derivatives, this technology addresses critical pain points in modern organic synthesis, such as high catalyst costs and complex purification requirements. For R&D directors and procurement managers alike, understanding the implications of this patent is crucial for optimizing supply chains and reducing the overall cost of goods sold for high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indanone derivatives has relied heavily on the use of precious transition metals such as palladium, rhodium, and ruthenium to facilitate carbon-hydrogen activation or carbonyl insertion reactions. While effective, these conventional methods suffer from significant drawbacks that hinder their commercial viability on a large scale. The reliance on expensive noble metals not only inflates the raw material costs but also introduces stringent regulatory hurdles regarding heavy metal residues in final pharmaceutical products. Furthermore, these catalytic systems often require harsh reaction conditions, including high pressures or extreme temperatures, which can compromise safety and increase energy consumption. The complexity of removing trace metal contaminants from the final product necessitates additional purification steps, thereby extending production lead times and reducing overall process efficiency. These factors collectively contribute to a higher cost base and a less sustainable manufacturing footprint for companies relying on legacy synthetic routes.
The Novel Approach
In stark contrast, the method disclosed in patent CN111807942B utilizes a metal-free photochemical strategy that dramatically simplifies the synthetic landscape. By employing ultraviolet light as the primary energy source to drive the reaction between aryl aldehydes and alkyne derivatives, this novel approach eliminates the need for costly transition metal catalysts entirely. The reaction proceeds under relatively mild conditions, typically utilizing common organic solvents like acetonitrile or toluene, which are readily available and easy to handle in an industrial setting. This shift not only reduces the direct material costs associated with catalyst procurement but also streamlines the downstream processing by removing the necessity for specialized metal scavenging resins or complex extraction protocols. The result is a cleaner reaction profile with high regioselectivity, enabling the production of high-purity intermediates with minimal environmental impact.
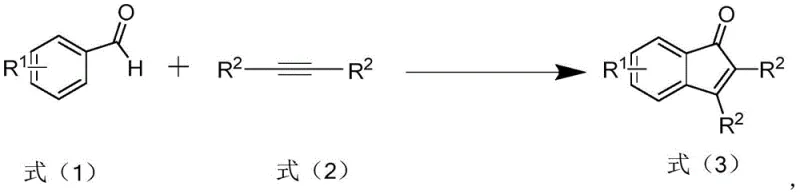
Mechanistic Insights into Photochemical Cyclization
The core of this technological advancement lies in its unique mechanistic pathway, which bypasses the traditional organometallic cycles in favor of a direct photo-induced cyclization. Under ultraviolet illumination, the aryl aldehyde and alkyne derivative undergo excitation, facilitating a radical or pericyclic process that leads to the formation of the indanone core. This mechanism is highly dependent on the wavelength and power of the light source, with the patent specifying optimal ranges between 250W and 300W to maximize efficiency. The absence of metal coordination spheres allows for greater flexibility in substrate scope, accommodating various substituents on the aromatic rings without the steric or electronic constraints often imposed by bulky ligands in metal catalysis. This robustness ensures that the process can be adapted to synthesize a wide array of analogues, providing R&D teams with the versatility needed to explore structure-activity relationships effectively.
Furthermore, the impurity profile generated by this photochemical route is significantly cleaner compared to metal-catalyzed counterparts. Without the introduction of metal salts, the risk of metal-induced side reactions or decomposition pathways is minimized, leading to a more predictable and controllable reaction outcome. The high regioselectivity observed in this process ensures that the desired polysubstituted indanone is formed predominantly, reducing the burden on purification teams to separate closely related isomers. This level of control is paramount for maintaining consistent quality in pharmaceutical intermediates, where even minor impurities can have significant implications for drug safety and efficacy. The ability to achieve purities exceeding 99% directly from the reaction workup underscores the precision of this photochemical transformation.
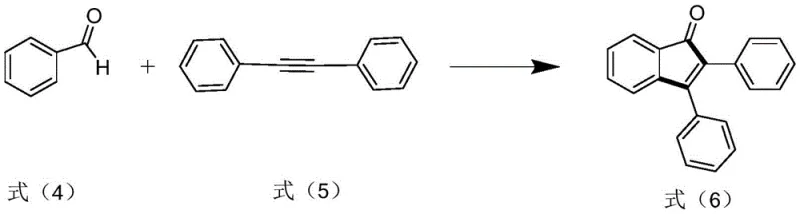
How to Synthesize Polysubstituted Indanone Derivative Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to ensure optimal yield and purity. The process begins with the precise mixing of aryl aldehyde and alkyne derivatives in a suitable solvent, followed by exposure to controlled ultraviolet radiation. Detailed standard operating procedures regarding light intensity, temperature control, and reaction duration are critical for reproducibility. The patent outlines specific molar ratios and solvent choices that have been empirically validated to provide the best results, serving as a foundational guide for process chemists looking to adopt this technology. For a comprehensive breakdown of the exact experimental conditions and step-by-step instructions, please refer to the standardized guide below.
- Prepare the reaction mixture by combining aryl aldehyde and alkyne derivative in a solvent such as acetonitrile.
- Subject the mixture to ultraviolet illumination (250W-300W) at temperatures between 130°C and 140°C.
- Perform post-treatment including cooling, extraction, drying, and chromatography to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this photochemical synthesis method offers substantial strategic advantages for procurement and supply chain management. The elimination of precious metal catalysts translates directly into significant cost savings, as the volatility of prices for metals like palladium and rhodium is removed from the cost equation. Additionally, the simplified workup procedure reduces the consumption of auxiliary materials and solvents, further driving down the variable costs associated with manufacturing. For supply chain heads, the use of widely available starting materials and standard equipment enhances supply security, mitigating the risks associated with sourcing specialized reagents. This resilience is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts fundamentally alters the cost structure of producing polysubstituted indanone derivatives. Without the need for palladium or rhodium complexes, the raw material expenditure is drastically lowered, allowing for more competitive pricing strategies. Moreover, the simplified purification process reduces the operational expenses related to waste treatment and solvent recovery. This economic efficiency enables manufacturers to offer high-quality intermediates at a lower price point, enhancing the overall value proposition for downstream customers seeking cost-effective solutions for their drug development pipelines.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as aryl aldehydes and alkynes, coupled with common solvents like acetonitrile, ensures a robust and stable supply chain. Unlike specialized catalysts that may have limited suppliers or long lead times, the key inputs for this process are readily accessible from multiple global sources. This diversification of supply reduces the risk of bottlenecks and ensures that production can be scaled up rapidly to meet surges in demand. For procurement managers, this reliability translates into greater confidence in securing long-term contracts and maintaining inventory levels without fear of disruption.
- Scalability and Environmental Compliance: The photochemical nature of this reaction is inherently scalable, as light sources can be arranged in parallel or series configurations to accommodate larger batch sizes. The mild reaction conditions and absence of toxic heavy metals align perfectly with increasingly stringent environmental regulations, reducing the regulatory burden on manufacturing facilities. This compliance not only minimizes the risk of fines or shutdowns but also enhances the corporate sustainability profile of the manufacturer. For supply chain leaders, partnering with a supplier that prioritizes green chemistry initiatives ensures alignment with corporate social responsibility goals and future-proofs the supply chain against evolving environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical feasibility and commercial implications of this synthesis method. These answers are derived directly from the technical disclosures within the patent documentation, providing accurate and reliable information for decision-makers. Understanding these details is essential for evaluating the potential integration of this technology into existing manufacturing workflows. For further technical clarification or custom feasibility studies, our team is available to provide detailed support.
Q: What are the advantages of this photochemical method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive transition metals like palladium or rhodium, significantly reducing raw material costs and simplifying purification by avoiding metal residue removal.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process uses simple operation conditions, common solvents like acetonitrile, and avoids harsh reagents, making it highly scalable for industrial manufacturing of pharmaceutical intermediates.
Q: What is the typical purity and yield achievable with this synthesis route?
A: Optimized conditions in the patent demonstrate yields up to 85% with purity reaching 99.5%, ensuring high quality for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Indanone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photochemical synthesis method described in patent CN111807942B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative chemistry from the laboratory to the marketplace. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of polysubstituted indanone derivative meets the highest industry standards. We are committed to leveraging this technology to deliver superior quality products that accelerate our clients' drug development timelines.
We invite you to collaborate with us to explore how this cost-effective and sustainable synthesis route can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic advantages of switching to this metal-free process. Please contact us today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for the next generation of pharmaceutical innovations.
