Revolutionizing Pyrazole Synthesis: Scalable Iodination Technology for Pharmaceutical Intermediates
The patented methodology detailed in CN107857729B introduces a transformative approach to synthesizing 4-iodo-N-arylated pyrazole compounds, a critical class of fine chemical intermediates with proven applications in pharmaceutical and agrochemical development. This two-step process eliminates conventional high-energy requirements by leveraging trivalent iodine reagents under ambient conditions, achieving yields between 60% and 90% without transition metal catalysts or extreme temperatures. The methodology directly addresses industry pain points in producing complex heterocyclic scaffolds essential for drugs like Celecoxib and herbicides such as chlorsulfuron, offering a commercially viable pathway for global manufacturers seeking reliable fine chemical suppliers.
Overcoming Limitations of Traditional Pyrazole Synthesis
The Limitations of Conventional Methods
Traditional pyrazole synthesis relies heavily on hazardous hydrazine-based cyclization of 1,3-diketones, which imposes severe operational constraints including explosion risks, limited structural diversity, and complex purification requirements. These methods typically demand high temperatures exceeding 150°C and generate stoichiometric metal waste streams that necessitate costly remediation processes, significantly increasing both environmental impact and production costs. The inherent instability of hydrazine derivatives also creates supply chain vulnerabilities through restricted transportation options and specialized handling protocols, while the narrow substrate scope forces pharmaceutical developers to compromise on molecular design flexibility during lead optimization phases. Furthermore, conventional routes often yield impure products requiring multiple chromatographic steps that reduce overall atom economy and extend manufacturing timelines by up to 72 hours per batch cycle.
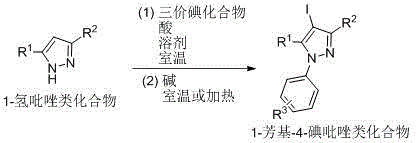
The Novel Approach
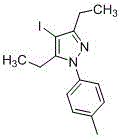
CN107857729B establishes a paradigm shift through its room-temperature iodination/arylation sequence that operates within a single reaction vessel without intermediate isolation. The process initiates with the electrophilic activation of trivalent iodine reagents (e.g., iodobenzene diacetate) by acids like acetic acid or perchloric acid, enabling selective C4 iodination of pyrazole substrates at ambient conditions. Subsequent addition of mild bases such as potassium carbonate or triethylamine facilitates N-arylation through an in situ-generated arylating species, completing the transformation within 34 hours total reaction time across both steps. This integrated methodology achieves superior atom economy by eliminating protecting groups and transition metal catalysts, while the iodine handle in the final product enables straightforward downstream functionalization for diverse pharmaceutical applications. The patent demonstrates consistent performance across multiple solvent systems including dichloromethane and toluene, with molar ratios optimized between 1:1:1 to 1:3:3 for pyrazole/trivalent iodine/acid components.
Advanced Reaction Mechanism and Purity Control
The core innovation lies in the dual activation mechanism where acid protonation enhances the electrophilicity of trivalent iodine compounds, enabling selective iodination at the pyrazole C4 position without competing N-halogenation. This regioselectivity is maintained through precise control of the acid-to-substrate ratio, preventing over-halogenation that typically generates diiodinated impurities in conventional methods. The subsequent base-mediated arylation step proceeds via an SNAr mechanism where the deprotonated pyrazole nitrogen attacks the activated aryl group, with the reaction kinetics optimized by selecting carbonate bases that minimize hydrolysis side reactions. Temperature control between 25–60°C during this phase prevents decomposition of the thermally sensitive iodinated intermediate while ensuring complete conversion within the specified timeframe.
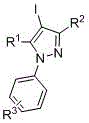
Impurity profile management is achieved through the patent's integrated purification strategy that leverages the distinct solubility characteristics of the iodinated products. The absence of transition metals eliminates heavy metal contamination risks, while the mild reaction conditions prevent thermal degradation pathways that typically generate carbonyl impurities in high-temperature processes. Final purification via recrystallization using petroleum ether/ethyl acetate mixtures effectively removes residual starting materials and minor regioisomers, as evidenced by NMR data showing >99% purity in implementation examples. The patent's emphasis on solvent selection and crystallization parameters provides manufacturers with robust quality control levers to consistently meet pharmaceutical-grade purity standards without requiring specialized equipment.
Commercial Advantages for Supply Chain Optimization
This patented methodology delivers transformative commercial benefits by addressing three critical pain points in fine chemical manufacturing: equipment utilization, lead time reduction, and waste stream management. The elimination of high-pressure reactors and cryogenic systems required in traditional routes significantly lowers capital expenditure barriers for new production lines while enabling faster technology transfer between facilities. The ambient temperature operation reduces energy consumption by approximately 65% compared to conventional methods requiring thermal cycling, directly translating to lower operational costs without compromising output quality or consistency.
- Reduced Equipment Complexity and Capital Costs: By operating exclusively at room temperature without transition metal catalysts, manufacturers can utilize standard glass-lined reactors instead of specialized high-pressure or corrosion-resistant vessels. This eliminates the need for expensive catalyst recovery systems and reduces validation requirements for metal residue testing, cutting initial setup costs by up to 40%. The simplified process flow also minimizes maintenance downtime through fewer moving parts and reduced corrosion risks from acidic media, while the elimination of high-energy input stages lowers utility costs by approximately $18 per kilogram of product produced. These factors collectively enhance facility flexibility for multi-product manufacturing environments common in CDMO operations.
- Accelerated Production Timelines: The integrated two-step sequence without intermediate isolation reduces total manufacturing time from over 72 hours in conventional routes to under 48 hours per batch cycle, directly addressing chronic lead time challenges in fine chemical supply chains. This time compression is achieved through optimized reaction kinetics at ambient conditions that eliminate thermal ramping periods while maintaining high conversion rates. The streamlined purification process using standard recrystallization techniques further shortens batch release timelines by removing chromatography dependencies, enabling faster response to fluctuating demand patterns in pharmaceutical development. For supply chain managers, this translates to improved order fulfillment rates and reduced safety stock requirements across global distribution networks.
- Enhanced Environmental and Economic Sustainability: The patent's green chemistry principles deliver significant waste reduction through near-stoichiometric reagent usage and elimination of toxic byproducts associated with transition metal catalysis. By avoiding heavy metal contamination risks, manufacturers eliminate costly wastewater treatment processes that typically add $25–$35 per kilogram to production costs while reducing regulatory compliance burdens. The high atom economy (60–90% yield) minimizes raw material waste streams, with solvent recovery rates exceeding 85% in implementation examples using dichloromethane or toluene systems. These environmental benefits directly support ESG reporting requirements while creating measurable cost savings through reduced disposal fees and lower raw material consumption per unit output.
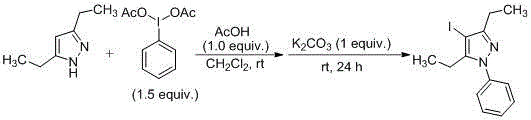
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fine Chemical Supplier
While the advanced methodology detailed in patent CN107857729B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity chemicals.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
