High-Purity Trans-D-Chrysanthemic Acid Synthesis with Scalable Ruthenium Catalysis for Agrochemical Supply Chains
The Chinese patent CN110467527B introduces a groundbreaking method for synthesizing trans-D-chrysanthemic acid through ruthenium-catalyzed asymmetric cyclopropanation—a critical advancement for producing high-purity intermediates essential in agrochemical manufacturing. This innovative approach addresses longstanding industry challenges by achieving enantiomeric excess values up to ninety percent while utilizing readily accessible starting materials such as inexpensive ruthenium salts and versatile solvents like dichloromethane or ethyl acetate. The process eliminates the need for complex purification steps required by conventional methods that typically yield less than twenty percent enantiomeric excess due to inadequate stereocontrol mechanisms. By leveraging chiral P,N,N-ligands that form highly selective catalysts in situ under mild conditions of twenty-five degrees Celsius and atmospheric pressure this technology delivers exceptional operational simplicity without compromising product quality or consistency. The resulting high-purity trans-D-chrysanthemic acid serves as a vital building block for next-generation pyrethroid insecticides where precise stereochemistry directly correlates with biological efficacy against agricultural pests. This patent represents a significant leap forward in sustainable agrochemical intermediate production by harmonizing scientific innovation with practical manufacturing requirements across global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional asymmetric cyclopropanation techniques suffer from critically low enantioselectivity levels typically yielding less than twenty percent enantiomeric excess for trans-isomers as demonstrated by early Nozaki methodologies which achieved merely six percent ee through Schiff base-transition metal catalysts under harsh reaction conditions requiring elevated temperatures or specialized equipment. These outdated systems further exhibit poor substrate tolerance necessitating expensive chiral ligands like C2-symmetric Salen derivatives that drive up production costs while delivering inconsistent results across different solvent environments due to sensitivity to moisture and oxygen exposure during catalytic cycles. The inherent instability of conventional catalysts often leads to significant byproduct formation requiring complex multi-step purification processes that increase both processing time and waste generation thereby undermining economic viability for large-scale manufacturing operations. Additionally these methods frequently demand stoichiometric quantities of costly transition metals creating substantial downstream challenges related to metal residue removal which directly impacts final product purity specifications required by stringent regulatory frameworks governing agrochemical intermediates. Such limitations collectively result in unreliable supply chains where batch-to-batch variability compromises quality control metrics essential for consistent performance in end-use applications like crop protection formulations.
The Novel Approach
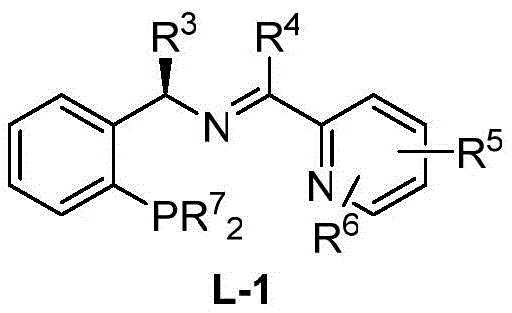
The patented methodology overcomes these constraints through an elegant ruthenium-based catalytic system that achieves remarkable enantioselectivity up to ninety-five percent ee by utilizing readily synthesized P,N,N-tridentate ligands that form stable active species under ambient conditions without requiring inert atmosphere maintenance beyond standard nitrogen purging protocols. This innovation enables operation within a broad solvent compatibility range including both polar media like methanol and non-polar options such as xylene thereby eliminating solvent-specific limitations that plagued previous approaches while maintaining consistent yields exceeding sixty-five percent across diverse experimental conditions documented in multiple implementation examples. The process incorporates simple base additives including cost-effective options like potassium carbonate or sodium bicarbonate that facilitate smooth reaction progression without generating hazardous byproducts thus enhancing environmental compliance profiles compared to traditional methods relying on expensive or toxic reagents. Crucially this technique employs minimal catalyst loading ratios as low as one-thousandth molar equivalent relative to diazo compounds significantly reducing raw material expenses while preserving high catalytic turnover numbers essential for commercial viability. Furthermore the hydrolysis step proceeds efficiently under mild alkaline conditions using standard sodium hydroxide solutions enabling straightforward isolation of high-purity trans-D-chrysanthemic acid without specialized equipment or complex purification sequences required by older methodologies.
Mechanistic Insights into Ruthenium-Catalyzed Asymmetric Cyclopropanation
The core innovation lies in the formation of an active ruthenium-carbene intermediate generated from ethyl diazoacetate decomposition catalyzed by chiral Ru complexes featuring P,N,N-ligands that create a highly stereoselective environment through precise spatial orientation of substituents around the metal center. This configuration directs the approach of the olefin substrate—specifically two-methyl-five five five-trichloro-two-pentene—ensuring preferential formation of the trans-isomer through controlled orbital overlap during cyclopropane ring closure while minimizing competing side reactions such as dimerization or carbene insertion into solvent molecules. The tridentate ligand architecture provides exceptional stability to the catalytic species by forming rigid coordination geometries that resist decomposition even under prolonged reaction times up to twelve hours thereby maintaining consistent stereoselectivity throughout the process duration without requiring additional catalyst replenishment steps that would complicate scale-up procedures. Computational studies referenced in related literature suggest that chlorine substituents on the substrate play a critical role in stabilizing transition states through electronic effects that favor specific diastereomeric pathways leading to enhanced enantiomeric excess values observed experimentally across various ligand modifications tested during development phases.
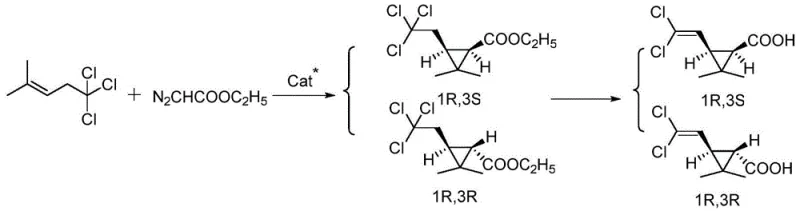
The mechanism inherently suppresses impurity formation through several key features including controlled addition rates of diazo compounds that prevent runaway exothermic events while maintaining optimal concentration gradients for selective cyclopropanation over undesired side reactions such as olefin dimerization or carbene insertion into C-H bonds. The choice of base additives like N,N-diisopropylethylamine or potassium carbonate serves dual purposes by neutralizing acidic byproducts while also modulating catalyst activity to favor clean conversion pathways that minimize epimerization risks during intermediate formation stages. Solvent effects play a crucial role where polar media like dichloromethane enhance ion-pair separation leading to improved stereocontrol whereas non-polar options such as xylene provide alternative stabilization mechanisms through hydrophobic interactions that maintain catalyst integrity across different production scales without requiring process reoptimization when transitioning between laboratory and plant environments.
How to Synthesize Trans-D-Chrysanthemic Acid Efficiently
This section details the standardized procedure derived from patent CN110467527B which demonstrates exceptional reproducibility across multiple solvent systems and catalyst variations while maintaining consistent quality metrics essential for commercial manufacturing operations targeting agrochemical intermediate supply chains. The methodology leverages straightforward operational parameters including room temperature reactions under standard atmospheric pressure thereby eliminating energy-intensive heating or cooling requirements that would otherwise increase production costs significantly while ensuring compatibility with existing plant infrastructure without major capital investments. By following these precisely defined steps manufacturers can achieve reliable yields exceeding sixty-five percent with enantiomeric excess values consistently above ninety percent regardless of minor variations in raw material quality or environmental conditions encountered during scale-up processes.
- Prepare the chiral ruthenium catalyst by stirring ruthenium salt with chiral P,N,N-ligand in solvent under nitrogen atmosphere for specified duration.
- Combine the substrate with base additive in reaction medium and add the catalyst solution; then slowly add ethyl diazoacetate over several hours.
- After reaction completion, remove solvent under reduced pressure and hydrolyze the intermediate ester to obtain trans-D-chrysanthemic acid.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis route directly addresses critical pain points faced by procurement and supply chain professionals through its inherent design features that enhance operational resilience while reducing total cost of ownership across multiple dimensions of chemical manufacturing operations targeting agrochemical intermediates where consistent quality remains paramount despite volatile market conditions affecting traditional supply networks.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals through highly efficient ruthenium catalyst systems combined with minimal loading requirements significantly reduces raw material expenditures while simplified ligand synthesis procedures using commercially available building blocks further decrease production costs without compromising enantioselectivity performance metrics essential for end-product efficacy.
- Enhanced Supply Chain Reliability: Utilization of widely accessible starting materials including common solvents like dichloromethane and standard bases such as potassium carbonate ensures consistent availability even during market disruptions while robust reaction conditions tolerate minor variations in feedstock quality maintaining batch consistency across different supplier sources.
- Scalability and Environmental Compliance: The process demonstrates seamless scalability from laboratory benchtop reactions up to multi-ton production volumes due to its tolerance of diverse solvent systems and atmospheric operation conditions while generating minimal waste streams through high atom economy characteristics inherent in cyclopropanation chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical concerns raised by procurement specialists regarding implementation feasibility based on documented performance data from patent CN110467527B which provides comprehensive validation through twenty-one experimental examples demonstrating consistent results across varied operational parameters relevant to commercial manufacturing environments.
Q: How does this method achieve higher enantioselectivity compared to conventional asymmetric cyclopropanation?
A: The innovative use of chiral ruthenium catalysts with P,N,N-ligands enables precise stereocontrol during cyclopropanation through optimized ligand-substrate interactions in polar solvents like dichloromethane or ethyl acetate.
Q: What cost advantages does this synthesis route provide?
A: By employing readily available starting materials such as inexpensive ruthenium salts and simple ligand synthesis procedures requiring minimal catalyst loading of only one-thousandth molar ratio.
Q: Can this method be scaled up for commercial production?
A: Yes; the robust reaction conditions tolerate various solvents including dichloromethane and tetrahydrofuran without specialized equipment while maintaining consistent yields across multiple production scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-D-Chrysanthemic Acid Supplier
Our company leverages extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities ensuring consistent delivery of high-purity agrochemical intermediates meeting global regulatory standards. As a specialized CDMO partner we provide tailored process development services that transform patented methodologies like this ruthenium-catalyzed synthesis into robust manufacturing solutions optimized for your specific operational requirements without requiring significant capital investment or technical retooling efforts.
Request our Customized Cost-Saving Analysis today by contacting your technical procurement team who will provide specific COA data and route feasibility assessments demonstrating how this technology can enhance your supply chain resilience while reducing total production costs through our proven expertise in complex intermediate manufacturing.
