Advanced Catalytic Hydrogenation for Scalable Cariprazine Intermediate Production
Advanced Catalytic Hydrogenation for Scalable Cariprazine Intermediate Production
The pharmaceutical landscape for treating schizophrenia is evolving rapidly, with third-generation antipsychotics like Cariprazine setting new standards for efficacy and patient compliance. However, the commercial viability of these advanced therapies often hinges on the availability of high-quality, cost-effective intermediates. Patent CN115806510B introduces a groundbreaking synthesis method for 3-(trans)-4-(2-hydroxyethyl)cyclohexyl-1,1-dimethylurea, a critical fragment of the Cariprazine molecule. This innovation addresses long-standing bottlenecks in API manufacturing by replacing expensive, multi-step routes with a streamlined, two-step catalytic process. By leveraging a specialized platinum-based hydrogenation system, this technology not only enhances stereochemical control but also significantly lowers the barrier to entry for generic and branded manufacturers alike.
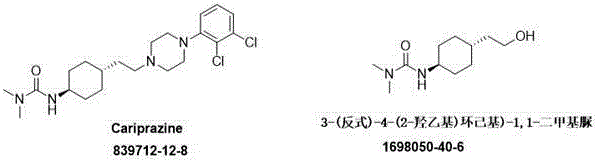
For R&D directors and process chemists, the structural integrity of the intermediate is paramount. As illustrated in the molecular comparison, the target urea derivative serves as the foundational scaffold for the final active pharmaceutical ingredient. The ability to synthesize this core structure with high trans-selectivity directly impacts the purity profile of the final drug substance, reducing the burden on downstream purification and ensuring consistent therapeutic performance. This patent represents a strategic shift towards more efficient, atom-economical pathways in psychotropic drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cariprazine intermediates has been plagued by inefficiencies that drive up costs and complicate supply chains. One prevalent literature method initiates with p-nitrophenylacetic acid, requiring a sequence of Raney nickel hydrogenation, esterification, protection, reduction, and substitution. This convoluted pathway suffers from abysmal yields, with patents reporting as low as 32% over the first two steps alone. Such poor efficiency renders the process economically unviable for large-scale production, creating a fragile supply chain susceptible to raw material fluctuations. Another common approach utilizes 4-amino Boc cyclohexanone, which involves Wittig-Horner reactions and subsequent hydrogenation. While chemically feasible, this route relies on prohibitively expensive starting materials and generates a roughly 1:1.7 mixture of cis and trans isomers. Separating these isomers requires energy-intensive recrystallization steps, further eroding profit margins and extending lead times for procurement teams.
The Novel Approach
In stark contrast, the methodology disclosed in CN115806510B offers a paradigm shift by utilizing p-nitrophenethyl alcohol as the starting material. This abundant and inexpensive feedstock undergoes a direct transformation into the target urea via just two high-efficiency reactions: catalytic hydrogenation followed by acylation. The brilliance of this approach lies in its simplicity and stereocontrol. By optimizing the hydrogenation conditions, the process achieves a superior trans-to-cis ratio, effectively bypassing the difficult separation steps required by older methods. This streamlined workflow not only accelerates the time-to-market for the intermediate but also aligns perfectly with the principles of green chemistry by minimizing waste and solvent usage. For a reliable pharmaceutical intermediate supplier, adopting this route translates to a more robust and competitive product portfolio.
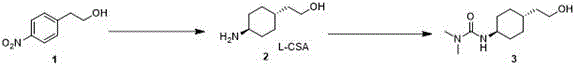
Mechanistic Insights into Platinum-Catalyzed Asymmetric Hydrogenation
The core innovation of this synthesis lies in the sophisticated manipulation of the hydrogenation step. Platinum, a Group 10 transition metal, is renowned for its ability to activate hydrogen and reduce aromatic systems. However, standard platinum catalysts often lack the necessary stereoselectivity for complex pharmaceutical intermediates. The patent reveals that by employing Platinum Dioxide (PtO2) in conjunction with a chiral acid additive, specifically L-Camphorsulfonic Acid (L-CSA), the reaction environment is fundamentally altered. The L-CSA acts as a chiral modifier, adsorbing onto the catalyst surface and creating a sterically hindered environment. This steric bulk directs the approach of the substrate, energetically favoring the formation of the thermodynamically stable trans-isomer over the cis-isomer. Experimental data within the patent indicates that this specific catalyst-additive combination can achieve a trans/cis ratio of up to 4.2/1, a significant improvement over non-modified systems which often yield near-equimolar mixtures.
Furthermore, the choice of solvent plays a critical role in modulating this selectivity. The patent data demonstrates that alcoholic solvents such as ethanol, methanol, and isopropanol maintain high conversion rates while preserving the favorable stereoselectivity induced by the L-CSA. In contrast, acidic solvents like acetic acid were found to diminish the trans-selectivity. This mechanistic understanding allows process engineers to fine-tune reaction parameters—such as temperature (75-80°C) and hydrogen pressure (0.8-2.5 MPa)—to maximize yield and purity. The subsequent acylation step is equally robust, utilizing dimethylcarbamoyl chloride and potassium carbonate in a biphasic system to ensure complete conversion without compromising the sensitive hydroxyl group or the newly formed stereocenters.
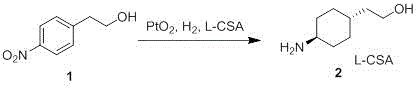
How to Synthesize 3-(trans)-4-(2-hydroxyethyl)cyclohexyl-1,1-dimethylurea Efficiently
Implementing this synthesis route requires precise adherence to the optimized conditions outlined in the patent to ensure reproducibility and high quality. The process is divided into two distinct stages: the stereoselective hydrogenation of the nitro-aromatic precursor and the subsequent urea formation. The first stage is critical, as it sets the stereochemical foundation for the entire molecule. Operators must carefully control the hydrogen pressure and temperature to balance reaction rate with selectivity. The second stage is a straightforward nucleophilic substitution that benefits from the high purity of the amine intermediate generated in step one. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below.
- Perform hydrogenation of p-nitrophenethyl alcohol using Platinum Dioxide and L-Camphorsulfonic acid in ethanol at 75-80°C under 2.0 MPa hydrogen pressure to obtain the trans-amine intermediate.
- React the resulting amine intermediate with dimethylcarbamoyl chloride in a biphasic system of dichloromethane and water, utilizing potassium carbonate as a base at 0-10°C.
- Purify the final urea product through extraction and concentration, achieving high purity suitable for subsequent coupling reactions in Cariprazine synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond mere chemical elegance. The shift from complex, low-yielding multi-step processes to a concise two-step protocol fundamentally alters the cost structure of the intermediate. By eliminating the need for expensive protecting groups like Boc and avoiding low-yield reduction steps, the overall material throughput is significantly increased. This efficiency gain directly correlates to a substantial reduction in the cost of goods sold (COGS), allowing for more competitive pricing in the global API market. Furthermore, the reliance on commodity chemicals like p-nitrophenethyl alcohol ensures a stable and secure supply of raw materials, mitigating the risks associated with sourcing specialized, high-cost precursors.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials such as 4-amino Boc cyclohexanone represents a major financial advantage. Traditional routes incur high costs not only from the raw materials themselves but also from the reagents required for protection and deprotection steps. By streamlining the synthesis to just hydrogenation and acylation, the consumption of auxiliary chemicals is drastically minimized. Additionally, the high yield of the hydrogenation step reduces the volume of waste generated per kilogram of product, lowering disposal costs and improving the overall environmental footprint of the manufacturing process.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the complexity of the manufacturing route. Long, multi-step syntheses introduce multiple points of failure, where a delay or quality issue in one step can halt the entire production line. This novel two-step approach significantly reduces the cycle time and simplifies the logistical chain. The robustness of the platinum-catalyzed hydrogenation ensures consistent output, while the use of common solvents like ethanol and dichloromethane simplifies procurement and inventory management. This reliability is crucial for maintaining continuous supply to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden challenges, but this method utilizes unit operations that are standard in the fine chemical industry. High-pressure hydrogenation and biphasic acylation are well-understood technologies that can be easily scaled from pilot plants to multi-ton reactors. Moreover, the improved atom economy and reduced solvent usage align with increasingly stringent environmental regulations. The ability to produce high-purity intermediates with less waste generation facilitates easier regulatory approval and supports sustainable manufacturing goals, making it an attractive option for environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production frameworks. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: How does the new catalytic system improve the cis-trans ratio compared to traditional methods?
A: The patented method utilizes a specific combination of Platinum Dioxide (PtO2) and L-Camphorsulfonic Acid (L-CSA). This chiral acid additive creates steric hindrance during the hydrogenation of the aromatic ring, significantly favoring the formation of the trans-isomer over the cis-isomer, achieving a ratio of up to 4.2/1, which simplifies downstream purification.
Q: What are the primary cost drivers eliminated in this synthesis route?
A: Traditional routes often rely on expensive starting materials like 4-amino Boc cyclohexanone or require low-yielding multi-step sequences. This method starts from cheap and readily available p-nitrophenethyl alcohol and reduces the total step count to just two high-yielding reactions, drastically lowering raw material and operational costs.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability. It employs standard industrial unit operations such as high-pressure hydrogenation and biphasic acylation. The use of common solvents like ethanol and dichloromethane, along with robust catalysts, ensures that the technology can be seamlessly transferred from laboratory to commercial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(trans)-4-(2-hydroxyethyl)cyclohexyl-1,1-dimethylurea Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex antipsychotic medications depends on the unwavering quality and availability of key intermediates. Our technical team has thoroughly analyzed the pathway described in CN115806510B and possesses the expertise to execute this platinum-catalyzed hydrogenation with precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical cis-trans isomer ratios required for high-performance API synthesis.
We invite you to collaborate with us to leverage this advanced synthesis technology for your Cariprazine projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and comprehensive route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and efficiency in your supply chain.
