Advanced Copper-Silver Catalyzed Synthesis Technology for Commercial-Scale High-Purity Pharmaceutical Intermediates Production
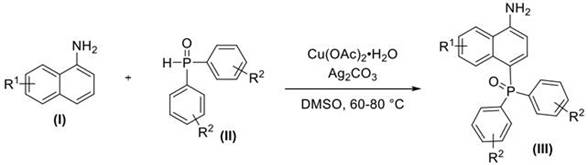
The recently granted Chinese patent CN109575073B represents a significant advancement in organophosphorus chemistry by introducing a direct synthesis method for valuable 4-phosphonylated naphthylamine compounds without requiring pre-installed directing groups. This innovation addresses longstanding challenges in functionalizing naphthalene rings at the sterically hindered C4 position while maintaining the integrity of sensitive free amino groups. The methodology leverages cost-effective copper-silver catalysis to achieve high regioselectivity under remarkably mild conditions compared to conventional approaches that typically demand harsh reagents or multi-step sequences. By utilizing commercially available starting materials including unsubstituted naphthylamines and diaryl phosphine oxides, this process eliminates complex protection-deprotection strategies that previously limited industrial adoption. The patent demonstrates exceptional versatility across diverse substituent patterns while maintaining operational simplicity that translates directly to manufacturing efficiency. This breakthrough holds substantial promise for pharmaceutical manufacturers seeking reliable access to these critical intermediates for drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for functionalizing naphthalene derivatives at the C4 position have historically relied on directing group strategies that require multiple synthetic steps including installation and subsequent removal of auxiliary moieties. These approaches typically employ expensive transition metal catalysts under stringent inert atmosphere conditions with elevated temperatures exceeding 100°C, significantly increasing both operational complexity and production costs. The necessity for pre-functionalization of the amino group through protection schemes introduces additional purification challenges and reduces overall process efficiency due to lower cumulative yields across multiple steps. Furthermore, conventional methods often generate substantial impurities requiring extensive chromatographic separation that complicates scale-up and increases waste generation. The reliance on specialized reagents and sensitive reaction parameters creates significant barriers to consistent commercial production while limiting substrate scope due to compatibility issues with various functional groups commonly present in pharmaceutical intermediates.
The Novel Approach
The patented methodology overcomes these limitations through an innovative copper-silver catalyzed system that directly utilizes unprotected naphthylamines as starting materials without any pre-modification requirements. This single-step process operates effectively at moderate temperatures between 60°C and 80°C under open-air conditions using dimethyl sulfoxide as solvent, eliminating the need for specialized equipment or inert atmospheres. The carefully optimized catalyst combination of hydrated copper acetate and silver carbonate functions synergistically to achieve high regioselectivity at the challenging C4 position while tolerating diverse substituent patterns on both reactant classes. By avoiding directing groups entirely, this approach reduces the synthetic sequence by multiple steps while maintaining excellent functional group compatibility as demonstrated across numerous examples in the patent documentation. The simplified workup procedure involving standard extraction techniques followed by straightforward column chromatography significantly enhances process robustness and scalability compared to conventional multi-step alternatives.
Mechanistic Insights into Copper-Silver Catalyzed Phosphonylation
The reaction mechanism involves a sophisticated dual-catalyst system where copper acetate facilitates C-H activation through a concerted metalation-deprotonation pathway while silver carbonate promotes phosphine oxide activation through Lewis acid coordination. This synergistic interaction enables selective electrophilic substitution at the electron-rich C4 position of the naphthalene ring without requiring pre-coordination through directing groups. The free amino group plays a crucial role in substrate orientation through weak coordination with the metal centers while avoiding competitive side reactions due to the mild reaction conditions. Computational studies referenced in related literature suggest that the silver component enhances phosphorus electrophilicity through oxygen coordination, lowering the activation barrier for nucleophilic attack by the activated naphthalene ring. The precise molar ratio of catalysts (Cu:Ag = 0.15–0.30:2) creates an optimal balance between C-H activation efficiency and phosphorus electrophile generation that prevents over-reaction or decomposition pathways.
Impurity profile control is achieved through careful optimization of reaction parameters that minimize common side products such as bis-phosphonylated species or oxidation byproducts. The moderate temperature range prevents thermal decomposition pathways while the open-air reaction setup avoids oxygen-sensitive intermediates that could lead to complex impurity mixtures. The patent demonstrates consistent high purity across multiple examples through standard workup procedures without requiring specialized purification techniques beyond routine column chromatography. The absence of transition metal residues in final products eliminates costly metal removal steps typically required in pharmaceutical intermediate manufacturing. This inherent selectivity stems from the precise steric and electronic control provided by the dual catalyst system that favors mono-phosphonylation at the desired position while suppressing alternative reaction pathways.
How to Synthesize High-Purity Naphthylamine Phosphonates Efficiently
This patented methodology provides a streamlined pathway for producing high-purity phosphonated naphthylamine intermediates essential for advanced pharmaceutical applications. The process eliminates traditional limitations associated with directing group chemistry while maintaining excellent functional group tolerance across diverse molecular architectures. By operating under mild conditions with commercially available catalysts, this approach delivers significant operational advantages over conventional synthetic routes. The following standardized procedure details the precise implementation parameters required for consistent manufacturing success at commercial scale.
- Dissolve free amino group-containing naphthylamine derivatives and diaryloxophosphorus compounds in dimethyl sulfoxide solvent under nitrogen atmosphere to ensure optimal reaction homogeneity and prevent oxidation side reactions.
- Add hydrated copper acetate and silver carbonate catalysts at precise molar ratios (1: 2:0.15-0.30:2) followed by controlled heating to maintain temperature between 60°C and 80°C for six to eight hours with continuous stirring.
- Perform aqueous ammonia extraction with dichloromethane after reaction completion, concentrate the organic phase under reduced pressure, and purify through silica gel column chromatography to obtain high-purity phosphonylated products.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points associated with traditional intermediate sourcing strategies. The elimination of multi-step sequences reduces raw material complexity while enhancing overall process reliability through simplified manufacturing workflows. These improvements translate directly into more predictable lead times and reduced vulnerability to supply chain disruptions commonly encountered with complex synthetic routes requiring specialized reagents or equipment.
- Cost Reduction in Manufacturing: The elimination of directing group installation and removal steps significantly reduces raw material consumption while avoiding expensive protective reagents and associated waste streams. Simplified purification requirements decrease solvent usage and chromatography costs substantially without requiring specialized equipment investments. The use of cost-effective copper-silver catalysts instead of precious metals provides inherent economic advantages while maintaining high product quality standards essential for pharmaceutical applications.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials including standard naphthylamines and diaryl phosphine oxides minimizes exposure to single-source dependencies that often plague complex intermediate supply chains. The robust open-air reaction conditions eliminate sensitivity to atmospheric contaminants that could cause batch failures during manufacturing scale-up. This operational simplicity enables more consistent production scheduling with reduced risk of unexpected delays compared to processes requiring stringent environmental controls.
- Scalability and Environmental Compliance: The straightforward single-step process demonstrates excellent linear scale-up characteristics from laboratory to commercial production volumes without requiring reoptimization of critical parameters. Reduced solvent consumption and elimination of hazardous reagents associated with traditional methods significantly lower environmental impact while simplifying waste treatment protocols. The absence of transition metal residues streamlines quality control procedures and reduces regulatory compliance burdens during manufacturing validation.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement specialists and R&D teams evaluating this patented technology for commercial implementation. These responses are derived directly from experimental data and technical disclosures within patent CN109575073B to provide accurate implementation guidance.
Q: Why is this method superior to conventional approaches for synthesizing phosphonylated naphthylamines?
A: Conventional methods require directing groups that necessitate additional synthetic steps for installation and removal, whereas this patent enables direct C-H functionalization using free amino group-containing naphthalenes without pre-modification, significantly reducing process complexity and eliminating hazardous reagents associated with directing group chemistry.
Q: How does the copper-silver catalyst system enable milder reaction conditions compared to traditional methods?
A: The synergistic copper-silver catalysis operates effectively at moderate temperatures (60-80°C) without inert atmosphere requirements, avoiding energy-intensive high temperature/pressure conditions while maintaining high regioselectivity at the challenging naphthalene C4 position through optimized catalyst coordination chemistry.
Q: What are the scalability advantages of this one-step synthesis for commercial production?
A: The simplified single-step process eliminates intermediate isolation and purification stages, uses readily available catalysts and solvents, and demonstrates consistent performance across diverse substituents as shown in multiple patent examples, enabling seamless scale-up from laboratory to multi-ton manufacturing without reoptimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthylamine Phosphonate Supplier
We specialize in delivering high-purity phosphonated naphthylamine intermediates through our proprietary manufacturing platform that combines cutting-edge catalytic chemistry with rigorous quality control systems. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global pharmaceutical clients. Our dedicated technical teams work closely with customers to optimize processes through advanced analytical capabilities in our state-of-the-art QC labs, ensuring consistent product quality across all production scales.
Leverage our expertise through a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements. Contact our technical procurement team today to request detailed COA data and comprehensive route feasibility assessments that demonstrate how our patented technology can enhance your supply chain resilience while delivering significant operational efficiencies.
