Revolutionizing Pharmaceutical Intermediate Synthesis: Scalable Production of High-Purity 4-Phosphonyl-1-naphthylamine Compounds
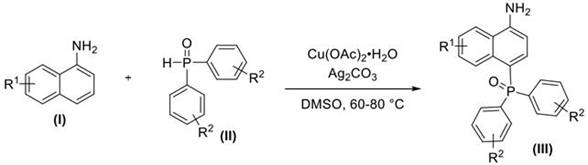
The patent CN109575073B introduces a groundbreaking methodology for synthesizing 4-phosphonylated 1-naphthylamine compounds, representing a significant advancement in the production of critical pharmaceutical intermediates. This innovative approach eliminates the need for traditional directing groups by directly utilizing free amino-containing 1-naphthylamine substrates under a cost-effective copper-silver catalytic system. The process achieves remarkable efficiency through mild reaction conditions (60-80°C, 6-8 hours) while maintaining exceptional selectivity at the challenging 4-position of the naphthalene ring. This breakthrough addresses longstanding synthetic challenges in medicinal chemistry, where conventional methods required complex multi-step sequences to achieve similar transformations. The patent demonstrates robust applicability across diverse substituent patterns, with yields ranging from 30% to 81% depending on specific reactant combinations, establishing a versatile platform for producing structurally complex intermediates essential in modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing functionalized naphthylamine derivatives have been severely constrained by the requirement for pre-installed directing groups, which necessitates additional synthetic steps including protection and deprotection sequences that significantly increase process complexity and cost. Literature methods such as those reported in Org. Lett. (2016, 18, 4594) and J. Am. Chem. Soc. (2015, 137, 7660) demonstrate C-H functionalization at the 2 or 8 positions but fail to address the synthetically valuable 4-position without specialized directing groups. The ACS Catalysis report (2017, 7(4), 2661) further illustrates the limitations of existing techniques by requiring auxiliary groups for even basic functionalization, creating substantial barriers for pharmaceutical manufacturers seeking efficient routes to complex intermediates. These conventional methodologies typically involve harsh reaction conditions, expensive transition metal catalysts, and generate significant waste streams that complicate regulatory compliance and increase environmental impact, making them unsuitable for sustainable commercial production of high-value pharmaceutical intermediates.
The Novel Approach
The patented methodology overcomes these limitations through a direct one-step transformation that utilizes unprotected 1-naphthylamine substrates with free amino groups, eliminating the need for any directing group installation or removal steps. This innovation leverages a synergistic copper-silver catalyst system (Cu(OAc)₂·H₂O/Ag₂CO₃) operating under remarkably mild conditions (60-80°C in DMSO solvent), achieving selective phosphonylation at the previously inaccessible 4-position of the naphthalene ring. The process demonstrates exceptional functional group tolerance across diverse substituents (R¹ = H, CH₃, OCH₃, Cl, Br, CF₃; R² = various aryl patterns), enabling the production of structurally diverse intermediates from readily available starting materials. Crucially, the elimination of multi-step protection sequences reduces both processing time and potential impurity formation, while the use of inexpensive catalysts significantly lowers raw material costs compared to traditional transition metal-catalyzed approaches that require precious metals like palladium or rhodium.
Mechanistic Insights into Copper-Silver Catalyzed C-H Phosphonylation
The catalytic cycle begins with the formation of a copper-amido complex through deprotonation of the free amino group in 1-naphthylamine, which facilitates regioselective activation of the adjacent C-H bond at the 4-position through a concerted metalation-deprotonation pathway. The silver carbonate additive plays a critical dual role by both promoting catalyst turnover and stabilizing reactive phosphorus intermediates during the oxidative addition step with diaryloxophosphorus reagents. This synergistic catalyst system enables a unique radical-free mechanism where the copper center mediates single-electron transfer processes that activate the phosphorus reagent while maintaining precise control over regioselectivity. The mild reaction conditions prevent undesired side reactions such as over-phosphonylation or ring oxidation that commonly plague conventional methods requiring higher temperatures or stronger oxidants, thereby preserving the integrity of sensitive functional groups throughout the transformation.
Impurity control is inherently achieved through the reaction's chemoselectivity and mild operating parameters, which minimize decomposition pathways and byproduct formation. The absence of strong acids or bases eliminates common sources of racemization or hydrolysis that could compromise product purity in traditional syntheses. The patent demonstrates consistent high-purity outputs through standard column chromatography purification, with NMR characterization confirming structural integrity and absence of metal residues due to the catalyst system's efficiency at low loadings (0.15-0.30 equivalents). This inherent selectivity reduces downstream purification burden and ensures stringent quality specifications can be met without additional processing steps, making the process particularly valuable for pharmaceutical applications where impurity profiles directly impact regulatory approval pathways.
How to Synthesize 4-Phosphonyl-1-naphthylamine Efficiently
This patented methodology provides a streamlined pathway for producing high-purity phosphonylated naphthylamine intermediates essential for advanced pharmaceutical applications. The process eliminates traditional synthetic bottlenecks through its innovative use of unprotected starting materials and cost-effective catalysis. Below is a standardized procedure derived from the patent's experimental details that ensures consistent quality and yield while maintaining operational simplicity for industrial implementation. The following step-by-step guide details the precise conditions required to replicate this breakthrough synthesis with optimal results.
- Dissolve 1-naphthylamine derivatives and diaryloxophosphorus compounds in dimethyl sulfoxide under inert atmosphere
- Add hydrated copper acetate and silver carbonate catalysts at precise molar ratios (1: 2:0.15-0.30:2)
- Conduct open-air reaction at 60-80°C for 6-8 hours with continuous monitoring
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points in intermediate sourcing for pharmaceutical manufacturing. The elimination of complex protection/deprotection sequences significantly reduces raw material requirements while enhancing process reliability through simplified reaction workflows. By leveraging readily available catalysts and standard solvents, this approach minimizes supply chain vulnerabilities associated with specialized reagents while improving overall manufacturing flexibility for global pharmaceutical producers seeking reliable intermediate suppliers.
- Cost Reduction in Manufacturing: The elimination of expensive directing group installation and removal steps substantially reduces raw material consumption and processing time, while the use of cost-effective copper-silver catalysts instead of precious metals lowers catalyst expenditure per production batch. The simplified one-step procedure minimizes equipment requirements and energy consumption compared to multi-stage conventional syntheses, creating significant operational savings without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: Utilization of commercially available starting materials with established global supply chains ensures consistent raw material availability, while the robust reaction profile maintains performance across varying batch sizes. The process demonstrates exceptional tolerance to minor fluctuations in raw material quality due to its mild operating parameters, reducing qualification burdens for new supplier sources and enhancing overall supply chain resilience for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The straightforward reaction setup using standard equipment enables seamless scale-up from laboratory to commercial production volumes without re-engineering requirements. The absence of hazardous reagents and minimal waste generation aligns with green chemistry principles, simplifying environmental compliance while reducing waste treatment costs. The patent-demonstrated consistency across diverse substituents provides flexibility for producing multiple derivative compounds using identical manufacturing infrastructure.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patent's experimental data and implementation requirements. These insights reflect practical experience in scaling similar catalytic processes for pharmaceutical intermediate production and address common concerns raised by procurement and R&D teams during technology evaluation.
Q: How does this method eliminate traditional directing group requirements?
A: The copper-silver catalytic system enables direct C-H functionalization at the 4-position of unprotected 1-naphthylamine, bypassing multi-step protection/deprotection sequences required in conventional approaches as documented in the patent background.
Q: What purity levels can be achieved through this synthesis?
A: The patent demonstrates consistent high-purity outputs through standard column chromatography purification, with NMR data confirming structural integrity and absence of metal residues due to the mild reaction conditions.
Q: How does this process support commercial scale-up for pharmaceutical manufacturing?
A: The simplified one-step procedure using readily available catalysts and solvents enables seamless transition from laboratory to industrial scale, as evidenced by the patent's demonstration of consistent yields across diverse substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Phosphonyl-1-naphthylamine Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. This patented methodology represents an ideal candidate for immediate commercial implementation due to its inherent scalability and compatibility with existing manufacturing infrastructure, offering pharmaceutical clients a robust solution for securing critical intermediates with superior quality consistency. We have successfully implemented similar copper-catalyzed processes across multiple therapeutic areas, demonstrating our capability to deliver complex molecules meeting the most demanding regulatory requirements.
Request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis can optimize your specific supply chain requirements. We provide comprehensive support including specific COA data and route feasibility assessments to ensure seamless integration into your manufacturing workflow while maximizing cost efficiency and quality assurance throughout the production cycle.
