Advanced Synthesis of Fused Ring 3,3'-Biindole Derivatives for Pharmaceutical Applications
Advanced Synthesis of Fused Ring 3,3'-Biindole Derivatives for Pharmaceutical Applications
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for more efficient, environmentally benign, and cost-effective methodologies. A significant breakthrough in this domain is documented in patent CN114349759A, which discloses a novel synthetic route for fused ring 3,3'-biindole derivatives. These complex molecular scaffolds are of immense interest in medicinal chemistry, particularly for their potential applications in treating protein folding disorders such as Huntington's disease, Parkinson's disease, and Alzheimer's disease. The patented method represents a paradigm shift from traditional transition-metal catalyzed processes to a more streamlined acid-catalyzed cyclization. By leveraging specific Lewis or Brønsted acid catalysts, this technology enables the construction of the fused biindole core under remarkably mild conditions. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this technology is crucial for securing high-quality building blocks for next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,3'-biindole derivatives has relied heavily on the use of precious transition metal catalysts such as palladium, gold, or copper, often in conjunction with chiral phosphoric acids or strong oxidants. While these methods have yielded valuable compounds, they are fraught with significant industrial drawbacks. Conventional protocols frequently necessitate prolonged reaction times, sometimes extending over many hours or even days, which severely impacts throughput and energy efficiency. Furthermore, the requirement for excess oxidants or bases introduces additional complexity to the workup procedure, generating substantial chemical waste that complicates environmental compliance. The reliance on expensive noble metals not only inflates the raw material costs but also poses risks of metal contamination in the final product, a critical concern for high-purity pharmaceutical intermediates intended for clinical use. These factors collectively create a bottleneck in the commercial scale-up of complex pharmaceutical intermediates, limiting the availability of these vital scaffolds for drug discovery programs.
The Novel Approach
In stark contrast to the cumbersome traditional routes, the methodology described in CN114349759A offers a refined and efficient alternative. This novel approach utilizes an acidic catalyst to promote the cycloisomerization of readily available indole precursors. The reaction proceeds with exceptional speed, often reaching completion within mere minutes, thereby drastically enhancing process efficiency. By operating at mild temperatures, typically around 50°C, the method minimizes thermal stress on sensitive functional groups and reduces energy consumption. The elimination of heavy metal catalysts and harsh oxidants simplifies the purification process, leading to cleaner product profiles and reduced waste generation. This streamlined protocol not only addresses the economic inefficiencies of older methods but also aligns with modern green chemistry principles, making it an attractive option for cost reduction in API manufacturing. The versatility of this system allows for the introduction of diverse substituents, facilitating the rapid generation of structural analogs for structure-activity relationship (SAR) studies.
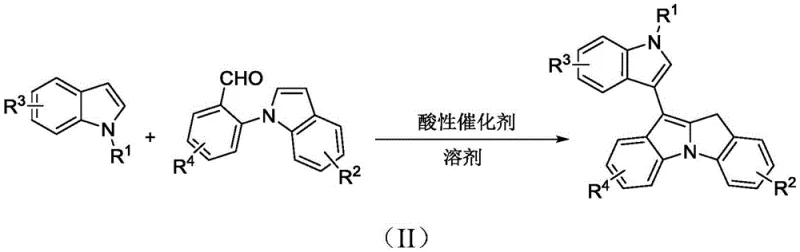
Mechanistic Insights into Acid-Catalyzed Cyclization
The core of this technological advancement lies in the precise activation of the substrate by the acidic catalyst. Whether utilizing a Lewis acid like silver triflate (AgOTf) or zinc iodide (ZnI2), or a Brønsted acid, the catalyst functions by activating the electrophilic center of the N-2-formylphenyl substituent on the second indole compound. This activation facilitates a nucleophilic attack by the C3 position of the first indole compound, initiating the cascade that leads to the formation of the new carbon-carbon bond and the subsequent ring closure. The mechanistic pathway is designed to be highly selective, favoring the formation of the desired fused ring system while suppressing side reactions such as polymerization or oligomerization, which are common pitfalls in indole chemistry. The mild nature of the catalysis ensures that the reaction trajectory remains controlled, preventing the degradation of sensitive moieties that might be present on the aromatic rings. This level of control is paramount for maintaining the integrity of the molecular architecture, ensuring that the final product possesses the requisite structural fidelity for biological evaluation.
Furthermore, the choice of catalyst and solvent system plays a pivotal role in managing the impurity profile of the reaction. The use of solvents like 1,2-dichloroethane (DCE) provides an optimal medium for solubilizing the organic substrates while supporting the catalytic cycle. The specific interaction between the silver or zinc species and the carbonyl oxygen enhances the electrophilicity without inducing unwanted redox processes. This subtle balance allows for the tolerance of a wide range of functional groups, including esters, halogens, and alkyl chains, as demonstrated in the various embodiments of the patent. For quality assurance teams, this implies a more predictable impurity spectrum, which simplifies the development of analytical methods and validation protocols. The ability to achieve high conversion rates with minimal byproduct formation translates directly into higher isolated yields and reduced downstream processing costs, a key metric for any reliable agrochemical intermediate supplier or pharma partner looking to optimize their supply chain.
How to Synthesize Fused Ring 3,3'-Biindole Derivatives Efficiently
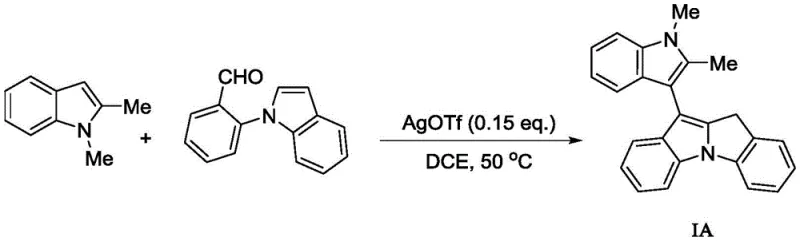
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The general procedure involves a sequential addition strategy where the catalyst is first dispersed in the solvent with the first indole component to ensure proper activation before the introduction of the aldehyde-containing partner. This order of addition is critical for controlling the exotherm and ensuring uniform mixing. The reaction is then maintained at a constant moderate temperature, avoiding the fluctuations that can lead to inconsistent results. Detailed standard operating procedures (SOPs) would specify the exact stoichiometric ratios, typically favoring a slight excess of the aldehyde component to drive the equilibrium towards the product. The following visual guide illustrates a representative example of this transformation, highlighting the specific reagents and conditions used to generate a methyl-substituted derivative.
- Prepare the reaction mixture by combining the first indole compound, solvent (e.g., 1,2-dichloroethane), and the acidic catalyst (e.g., AgOTf or ZnI2) in a reaction vessel.
- Stir the initial mixture at a moderate temperature (preferably 50°C) for a brief period (5-10 minutes) to ensure catalyst activation and substrate solubilization.
- Add the second indole compound containing the N-2-formylphenyl substituent to the mixture and continue stirring at the same temperature until the reaction is complete, typically within 5 to 30 minutes.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple chemical transformation. The shift away from precious metal catalysts represents a significant opportunity for cost reduction in electronic chemical manufacturing and pharmaceutical sectors alike. By removing the dependency on volatile commodities like palladium or gold, manufacturers can stabilize their raw material costs and mitigate the risk of supply disruptions associated with these critical metals. Additionally, the simplified workup procedure reduces the consumption of auxiliary chemicals and solvents required for purification, further driving down the overall cost of goods sold (COGS). The shortened reaction time also implies a higher throughput capacity for existing reactor infrastructure, allowing facilities to produce more material in less time without requiring capital expenditure on new equipment. These efficiencies contribute to a more resilient and responsive supply chain capable of meeting fluctuating market demands.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in reaction time directly lower the variable costs associated with production. The process avoids the need for specialized ligands or rigorous inert atmosphere conditions often required for sensitive metal catalysis, further reducing operational overhead. Consequently, the overall manufacturing economics are improved, allowing for more competitive pricing structures in the global market. This cost efficiency is achieved without compromising the quality of the final product, ensuring that budget optimizations do not come at the expense of purity or performance.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, primarily substituted indoles and formylphenyl derivatives, are commodity chemicals that are widely available from multiple global sources. This abundance reduces the risk of single-source dependency and ensures a steady flow of raw materials. The robustness of the reaction conditions means that production is less susceptible to minor variations in utility supplies or environmental factors, leading to more consistent batch-to-batch quality. For supply chain planners, this reliability translates into more accurate forecasting and the ability to maintain lower safety stock levels while still meeting delivery commitments.
- Scalability and Environmental Compliance: The mild thermal profile and the use of standard organic solvents make this process inherently scalable from gram to tonne quantities. The absence of hazardous oxidants simplifies waste treatment protocols, reducing the environmental footprint of the manufacturing process. This alignment with green chemistry principles facilitates easier regulatory approval and permits for expansion. As regulatory scrutiny on chemical manufacturing intensifies, having a process that naturally generates less hazardous waste provides a distinct competitive advantage and ensures long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these fused ring derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for integrating this technology into existing development pipelines and for making informed sourcing decisions.
Q: What are the primary advantages of this acid-catalyzed method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and potentially toxic transition metals like palladium or gold. It operates under significantly milder conditions (50°C vs. higher temperatures) and achieves completion in minutes rather than hours, reducing energy consumption and simplifying downstream purification.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process is highly scalable. The use of common solvents like 1,2-dichloroethane, mild thermal requirements, and short reaction times make it ideal for commercial scale-up without the safety hazards associated with high-pressure or high-temperature reactors.
Q: What types of substituents are tolerated on the indole rings?
A: The methodology demonstrates excellent functional group tolerance. It accommodates various substituents including alkyl groups, aryl groups, esters, cyano groups, and halogens at multiple positions on the indole scaffold, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fused Ring 3,3'-Biindole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN114349759A and is fully equipped to leverage these insights for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to manufacturing is seamless. Our state-of-the-art facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that employ the latest analytical techniques to guarantee product integrity. Whether you require custom synthesis of novel analogs or bulk supply of established intermediates, our infrastructure is ready to support your goals.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic impact of switching to this efficient route for your specific volume needs. We encourage you to contact us today to request specific COA data and route feasibility assessments for your target molecules. Let us help you optimize your supply chain and accelerate your path to market with our reliable and high-quality chemical solutions.
