Advanced Oxygen-Promoted Synthesis of Beta-Hydroxy-Difluoro Sulfonyl Esters for Commercial Scale-Up
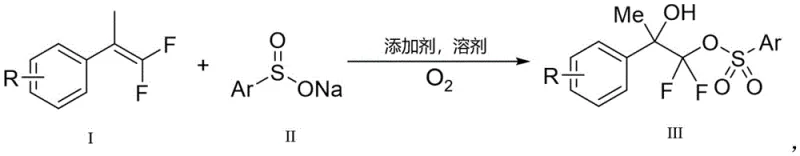
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex fluorinated scaffolds, which are critical motifs in modern drug design. Patent CN113527146B introduces a groundbreaking approach for the preparation of beta-hydroxy-difluoro sulfonyl ester compounds by promoting the hydroxylation sulfonyl esterification reaction of difluoroalkenes using molecular oxygen. This technology represents a significant leap forward in organic synthesis, addressing the long-standing challenge of functionalizing electron-deficient olefins efficiently. By utilizing non-toxic molecular oxygen as the terminal oxidant and readily available sodium sulfinates, this method offers an environmentally benign pathway to valuable intermediates. The reaction proceeds through a radical difunctionalization mechanism that tolerates a wide range of substrates, delivering moderate to good yields under mild conditions. For R&D teams focused on fluorinated medicinal chemistry, this patent provides a reliable blueprint for accessing diverse beta-hydroxy sulfone derivatives that serve as versatile building blocks for further elaboration into bioactive molecules.
Traditional methods for synthesizing beta-hydroxy sulfones often rely on stoichiometric oxidants or transition metal catalysts that pose significant economic and environmental burdens. Conventional approaches, such as those involving palladium, copper, or nickel catalysis, frequently require rigorous exclusion of air and moisture, adding complexity to the operational protocol. Furthermore, existing literature indicates that electron-deficient olefins, particularly gem-difluoroalkenes, are notoriously difficult substrates for standard radical difunctionalization reactions due to their low electron density. Previous attempts using iodine catalysis or electrochemical oxidation have shown promise but often suffer from limited substrate scope or the need for specialized equipment. The reliance on expensive metal catalysts also introduces the risk of heavy metal contamination in the final product, necessitating costly purification steps to meet stringent pharmaceutical purity specifications. These limitations hinder the rapid scale-up and cost-effective manufacturing of fluorinated intermediates required for large-scale drug production.
The novel approach disclosed in CN113527146B overcomes these hurdles by leveraging the unique reactivity of molecular oxygen to generate sulfonyloxy radicals directly from sodium sulfinates. Instead of forming simple sulfonyl radicals, the oxygen-promoted system facilitates the formation of a reactive species that effectively attacks the electron-deficient double bond of the gem-difluoroalkene. This strategy eliminates the need for transition metal catalysts entirely, thereby removing the associated costs of catalyst procurement and the downstream burden of metal scavenging. The reaction conditions are remarkably mild, typically operating at temperatures around 50°C in common solvents like 1,2-dichloroethane (DCE) with trifluoroacetic acid (TFA) as an additive. This simplicity translates directly into operational ease, allowing for straightforward implementation in standard glassware without the need for inert atmosphere gloveboxes or electrochemical cells. The broad substrate tolerance demonstrated in the patent examples suggests that this methodology is robust enough to handle various aryl and heteroaryl substitutions, making it a versatile tool for library synthesis.
Mechanistic Insights into Molecular Oxygen-Promoted Radical Difunctionalization
The core of this innovation lies in the mechanistic pathway where molecular oxygen acts not merely as a passive oxidant but as an active participant in generating the key radical intermediates. The proposed mechanism involves the oxidation of the sodium sulfinate salt by oxygen to produce a sulfonyloxy radical species, which is distinct from the traditional sulfonyl radical generated in metal-catalyzed systems. This sulfonyloxy radical adds to the gem-difluoroalkene substrate, initiating a cascade that ultimately installs both the sulfonyl ester group and the hydroxyl functionality across the double bond. The presence of the two fluorine atoms on the alkene significantly influences the electronic properties of the intermediate carbon-centered radical, stabilizing it sufficiently to allow for the subsequent trapping by oxygen or water equivalents to form the beta-hydroxy motif. Understanding this radical trajectory is crucial for R&D directors aiming to optimize reaction parameters or adapt the chemistry to novel substrates. The regioselectivity observed, where the sulfonyl group and hydroxyl group add in a specific orientation, is driven by the stability of the radical intermediates and the steric environment of the substituents on the alkene.
Impurity control in this radical process is inherently managed by the selectivity of the oxygen-mediated oxidation. Unlike harsh chemical oxidants that might over-oxidize sensitive functional groups on the aromatic rings, molecular oxygen offers a level of chemoselectivity that preserves other moieties such as halides, methoxy groups, or nitriles. The patent data demonstrates that substrates bearing electron-withdrawing groups like cyano or halogen atoms on the sulfinate ring are well-tolerated, yielding the desired products without significant side reactions. This high level of functional group compatibility is essential for late-stage functionalization in drug discovery, where complex molecules often contain multiple sensitive sites. Furthermore, the absence of metal catalysts means that the impurity profile is cleaner, lacking the trace metal residues that often complicate regulatory filings for new drug applications. The reaction byproducts are minimal, primarily consisting of benign salts or water, which simplifies the workup procedure and enhances the overall mass balance of the process.
How to Synthesize Beta-Hydroxy-Difluoro Sulfonyl Esters Efficiently
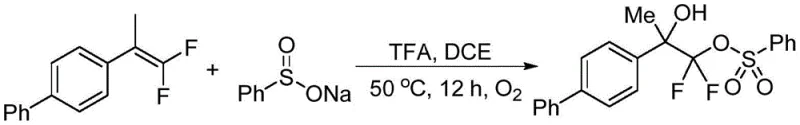
Implementing this synthesis route requires careful attention to the molar ratios and reaction atmosphere to maximize yield and reproducibility. The patent outlines a preferred embodiment where the gem-difluoroolefin and sodium benzenesulfinate are mixed in a molar ratio of approximately 1:2, ensuring an excess of the sulfinate source to drive the reaction to completion. The choice of solvent is critical, with halogenated hydrocarbons like DCE showing optimal performance, although other polar aprotic solvents may also be viable depending on substrate solubility. The addition of a protonic acid additive, specifically trifluoroacetic acid, is a key parameter that likely assists in protonating intermediates or stabilizing the radical species during the transformation. Operators should maintain a steady flow of oxygen or an oxygen-rich atmosphere throughout the 12-hour reaction period at 50°C to ensure continuous regeneration of the active oxidizing species. Following the reaction, the crude mixture can be concentrated and purified using standard flash column chromatography techniques to isolate the high-purity beta-hydroxy-difluoro sulfonyl ester product.
- Mix gem-difluoroolefin (Compound I) and sodium benzenesulfinate (Compound II) in a reaction vessel equipped with a stirrer.
- Add the protonic acid additive, preferably trifluoroacetic acid (TFA), and the solvent, optimally 1,2-dichloroethane (DCE).
- Heat the mixture to 50°C and stir for 12 hours under an oxygen atmosphere, then isolate the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this oxygen-promoted methodology offers substantial strategic advantages over traditional metal-catalyzed routes. The elimination of precious metal catalysts such as palladium or rhodium removes a significant variable cost component and mitigates the supply risk associated with fluctuating metal prices. Sodium sulfinates are commodity chemicals that are widely available from multiple global suppliers, ensuring a stable and competitive supply chain for the primary sulfur source. Furthermore, the use of molecular oxygen, which can be sourced from ambient air or industrial gas suppliers at low cost, drastically reduces the expense of oxidants compared to stoichiometric reagents like peroxides or hypervalent iodine compounds. This shift towards abundant and inexpensive reagents aligns perfectly with initiatives for cost reduction in fine chemical manufacturing, allowing companies to improve their gross margins on key intermediates. The simplified reaction setup also reduces the capital expenditure required for specialized reactor equipment, as standard stainless steel or glass-lined reactors capable of handling mild pressure and temperature are sufficient.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the use of air-stable reagents lead to a direct decrease in raw material costs. Without the need for complex ligand systems or anhydrous conditions, the operational expenditure related to solvent drying and inert gas purging is also significantly lowered. The simplified purification process, which avoids heavy metal scavenging resins, further contributes to overall cost efficiency by reducing consumable usage and waste disposal fees. These cumulative savings make the process economically attractive for large-scale production runs where even marginal cost improvements translate into substantial financial benefits.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like sodium sulfinates and oxygen ensures that the supply chain is not vulnerable to the geopolitical or logistical bottlenecks often associated with specialty catalysts. The robustness of the reaction conditions means that manufacturing can be easily transferred between different sites or contract manufacturing organizations without requiring extensive re-validation of sensitive parameters. This flexibility enhances supply continuity, a critical factor for maintaining uninterrupted production of downstream pharmaceutical APIs. Additionally, the stability of the starting materials allows for longer shelf-life and easier inventory management, reducing the risk of raw material spoilage.
- Scalability and Environmental Compliance: The green nature of using molecular oxygen as the oxidant aligns with increasingly strict environmental regulations regarding waste generation and hazardous chemical usage. The process generates minimal hazardous waste, primarily aqueous waste streams that are easier to treat than those containing heavy metals or toxic oxidants. This environmental profile facilitates smoother regulatory approvals and reduces the liability associated with hazardous waste disposal. The mild reaction temperatures and pressures also enhance process safety, making the scale-up from laboratory to pilot and commercial plant safer and more predictable, thereby reducing lead time for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxygen-promoted synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances helps in making informed decisions about process integration and resource allocation.
Q: What are the advantages of using molecular oxygen in this sulfonyl esterification?
A: Using molecular oxygen as the oxidant eliminates the need for expensive and toxic transition metal catalysts or harsh chemical oxidants. This significantly reduces raw material costs and simplifies the post-reaction purification process by avoiding heavy metal residue removal, making it highly suitable for green manufacturing of pharmaceutical intermediates.
Q: Can this method accommodate electron-deficient olefins like gem-difluoroalkenes?
A: Yes, unlike many conventional radical difunctionalization methods that struggle with electron-deficient substrates, this patented protocol specifically targets gem-difluoroolefins. The radical mechanism facilitated by oxygen allows for efficient addition across the electron-poor double bond, expanding the substrate scope for fluorinated drug discovery.
Q: What are the typical reaction conditions and purification methods described?
A: The reaction typically proceeds at moderate temperatures around 50°C for 12 hours in solvents like DCE with TFA as an additive. The resulting beta-hydroxy-difluoro sulfonyl esters can be isolated through standard workup procedures such as spin-drying followed by flash column chromatography or solvent washing, ensuring high purity without complex distillation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Hydroxy-Difluoro Sulfonyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the oxygen-promoted hydroxylation sulfonyl esterification method disclosed in CN113527146B for the production of high-value fluorinated intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of radical chemistry and oxygen-mediated reactions safely and efficiently. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of beta-hydroxy-difluoro sulfonyl ester meets the highest quality standards required by the global pharmaceutical industry. Our commitment to excellence ensures that you receive a reliable pharmaceutical intermediate supplier partnership that prioritizes both quality and consistency.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target cost structures. By partnering with NINGBO INNO PHARMCHEM, you gain access to our deep expertise in process optimization and supply chain management. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your development timeline and secure a competitive advantage in the market with our superior manufacturing capabilities and dedication to customer success.
