Advanced Palladium-Catalyzed Carbonylation for Scalable 3-Benzylidene-2,3-Dihydroquinolone Manufacturing
Introduction to Novel Dihydroquinolone Synthesis
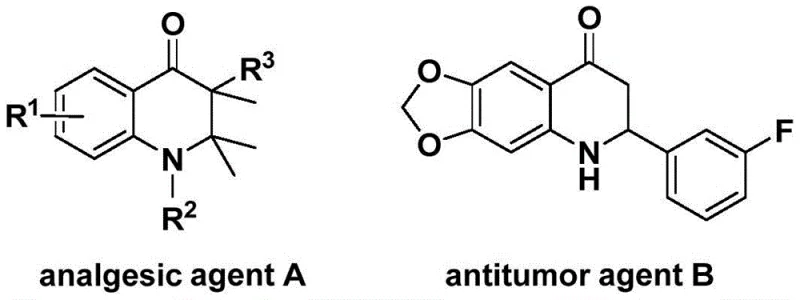
The landscape of heterocyclic chemistry is continually evolving, driven by the demand for efficient routes to bioactive scaffolds. Patent CN113735826A introduces a significant advancement in the preparation of 3-benzylidene-2,3-dihydroquinolone compounds, a structural motif prevalent in molecules exhibiting potent analgesic and antitumor activities. As illustrated in the biological context, these nitrogen-containing six-membered rings serve as critical cores for drug discovery, yet their synthesis has often been plagued by complex multi-step sequences or harsh reaction conditions. This new methodology leverages a palladium-catalyzed carbonylation strategy that not only streamlines the synthetic pathway but also utilizes readily available starting materials like N-pyridinesulfonyl-o-iodoaniline and allenes. By replacing toxic gaseous carbon monoxide with a solid surrogate, the process addresses key safety concerns while maintaining high atom economy, positioning it as a viable candidate for industrial scale-up in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3-dihydroquinolone skeleton has relied on classical cyclization reactions that often suffer from significant drawbacks regarding operational complexity and environmental impact. Traditional routes frequently require pre-functionalized substrates that are expensive to procure or synthesize, leading to inflated raw material costs that burden the supply chain. Furthermore, many existing protocols necessitate the use of stoichiometric amounts of hazardous reagents or extreme temperatures that compromise safety profiles in a manufacturing setting. The reliance on gaseous carbon monoxide for carbonylation steps introduces severe logistical challenges, including the need for specialized high-pressure equipment and rigorous safety monitoring, which can drastically increase capital expenditure for production facilities. Additionally, conventional methods often exhibit poor functional group tolerance, limiting the diversity of derivatives that can be accessed without extensive protection-deprotection strategies, thereby reducing overall process efficiency.
The Novel Approach
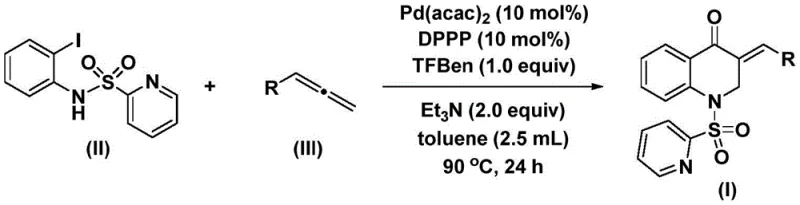
In stark contrast, the methodology disclosed in the patent offers a transformative solution by employing a catalytic system that operates under mild and controlled conditions. The core innovation lies in the use of 1,3,5-trimesic acid phenyl ester as a safe and effective carbon monoxide substitute, which eliminates the risks associated with handling toxic CO gas while ensuring a steady release of the carbonyl source during the reaction. This approach utilizes a robust palladium catalyst system paired with the DPPP ligand, facilitating a smooth coupling between the iodoaniline derivative and various allenes to form the target exocyclic double bond with high stereoselectivity. The reaction proceeds efficiently in toluene at moderate temperatures around 90°C, demonstrating exceptional compatibility with electron-donating and electron-withdrawing groups on the aromatic ring. This versatility allows for the rapid generation of diverse compound libraries, as evidenced by the successful synthesis of derivatives bearing methyl, methoxy, and halogen substituents with isolated yields reaching up to 93%, showcasing the method's superiority for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The success of this synthetic route is underpinned by a well-defined catalytic cycle that ensures high turnover and minimal byproduct formation, a critical factor for R&D directors focused on impurity profiles. The mechanism initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridinesulfonyl-o-iodoaniline, generating a reactive aryl-palladium intermediate. This step is crucial as it activates the aromatic ring for subsequent transformations. Following this activation, the carbon monoxide released from the trimesic acid derivative inserts into the palladium-carbon bond, forming an acyl-palladium species. This carbonyl insertion is the key step that builds the lactam carbonyl functionality inherent to the quinolone core. Subsequently, the allene substrate coordinates to the metal center and undergoes migratory insertion, creating a new carbon-carbon bond and establishing the exocyclic alkene geometry. The cycle concludes with a reductive elimination step that releases the final 3-benzylidene-2,3-dihydroquinolone product and regenerates the active palladium catalyst, allowing the cycle to continue with high efficiency.
From a quality control perspective, the choice of the pyridine sulfonyl protecting group plays a pivotal role in directing the regioselectivity and stabilizing the intermediates throughout the reaction pathway. The electron-withdrawing nature of the sulfonyl group enhances the electrophilicity of the nitrogen, facilitating the initial oxidative addition and preventing unwanted side reactions such as homocoupling of the aryl iodide. Furthermore, the use of triethylamine as an additive helps to neutralize any acidic byproducts generated during the decomposition of the CO surrogate, maintaining a neutral pH environment that preserves the integrity of the catalyst. The mechanistic clarity provided by this pathway allows for precise tuning of reaction parameters, ensuring that impurities are kept to a minimum and that the final product meets stringent purity specifications required for downstream applications in drug development. This level of control is essential for scaling up complex organic syntheses where reproducibility is paramount.
How to Synthesize 3-Benzylidene-2,3-Dihydroquinolone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to maximize yield and safety. The process begins with the careful weighing of the palladium catalyst and ligand to ensure the correct molar ratios, typically 10 mol% each relative to the substrate, which balances cost with catalytic activity. The reaction mixture is assembled in an inert atmosphere to prevent catalyst deactivation by oxygen, and the temperature is ramped to 90°C to initiate the carbonylation sequence. Monitoring the reaction progress via TLC or HPLC is recommended to determine the optimal endpoint, usually achieved within 24 hours for most substrates. Upon completion, the workup procedure is notably straightforward, involving simple filtration to remove insoluble palladium residues followed by standard chromatographic purification. For detailed operational parameters and specific stoichiometric ratios tailored to different allene substrates, please refer to the standardized synthesis guide below.
- Combine bis(acetylacetone)palladium, DPPP ligand, triethylamine, 1,3,5-trimesic acid phenyl ester, N-pyridinesulfonyl-o-iodoaniline, and the specific allene substrate in toluene within a Schlenk tube.
- Heat the reaction mixture to a temperature range of 80-100°C (optimally 90°C) and maintain stirring for 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the crude residue with silica gel, and purify the final product via column chromatography to isolate the target dihydroquinolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend beyond mere chemical yield, impacting the overall cost structure and reliability of the supply chain. The elimination of high-pressure carbon monoxide gas removes a significant bottleneck in facility licensing and safety compliance, effectively reducing the barrier to entry for manufacturers who may lack specialized high-pressure infrastructure. This simplification translates directly into lower capital expenditure and reduced operational overhead, making the production of these valuable intermediates more economically viable. Furthermore, the use of commercially available and inexpensive starting materials, such as substituted allenes and iodoanilines, ensures a stable supply base that is less susceptible to market volatility compared to exotic reagents. The robustness of the reaction conditions also implies a wider processing window, reducing the risk of batch failures and ensuring consistent delivery schedules for downstream clients.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substitution of hazardous gaseous reagents with stable solid surrogates and the use of earth-abundant palladium catalysts that can be potentially recovered. By avoiding the need for specialized high-pressure reactors and complex safety systems, manufacturers can significantly lower their fixed costs and maintenance expenses. Additionally, the high atom economy of the carbonylation reaction minimizes waste generation, leading to reduced costs associated with waste disposal and environmental compliance. The streamlined one-pot nature of the synthesis reduces labor hours and solvent consumption compared to multi-step alternatives, further contributing to substantial cost savings in the overall manufacturing budget without compromising on product quality.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals for the starting materials ensures that the supply chain remains resilient against disruptions. Unlike processes dependent on custom-synthesized precursors with long lead times, the reagents for this method can be sourced from multiple global suppliers, mitigating the risk of single-source dependency. The mild reaction conditions also allow for flexible scheduling and easier integration into existing production lines without requiring extensive retrofitting. This flexibility enables manufacturers to respond more agilely to fluctuations in demand, ensuring that critical pharmaceutical intermediates are available when needed to support continuous drug production pipelines and reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram levels is facilitated by the homogeneous nature of the catalytic system and the absence of exothermic hazards associated with gas handling. The use of toluene, a common industrial solvent, simplifies solvent recovery and recycling processes, aligning with green chemistry principles and reducing the environmental footprint of the operation. The straightforward workup procedure, which avoids complex extractions or crystallizations, minimizes solvent waste and energy consumption during isolation. These factors collectively enhance the sustainability profile of the manufacturing process, helping companies meet increasingly stringent environmental regulations while maintaining high production throughput and operational efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed carbonylation technology. Understanding these nuances is vital for process chemists aiming to adapt this method for specific project needs. The answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing a reliable foundation for decision-making. Whether concerning catalyst loading, substrate scope, or purification strategies, these insights aim to clarify the practical aspects of adopting this innovative synthetic route.
Q: What is the role of 1,3,5-trimesic acid phenyl ester in this reaction?
A: It serves as a solid carbon monoxide (CO) surrogate, releasing CO in situ to facilitate the carbonylation step without requiring hazardous high-pressure CO gas cylinders, thereby enhancing operational safety.
Q: Can this method tolerate diverse functional groups on the allene substrate?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating various substituents such as methyl, methoxy, tert-butyl, and halogens (F, Cl, Br) at ortho, meta, and para positions with high yields.
Q: What are the optimal reaction conditions for maximum yield?
A: The patent specifies using toluene as the solvent at 90°C for 24 hours with 10 mol% Pd(acac)2 and 10 mol% DPPP ligand, which consistently provides yields ranging from 69% to 93% across different substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Benzylidene-2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet rigorous quality standards, utilizing state-of-the-art rigorous QC labs to verify every batch. Our capability to implement advanced catalytic technologies, such as the palladium-catalyzed carbonylation described here, allows us to offer competitive pricing and reliable supply continuity for complex heterocyclic building blocks essential for modern medicinal chemistry.
We invite you to collaborate with us to leverage these technological advancements for your specific projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage potential partners to reach out for specific COA data and route feasibility assessments to understand how our optimized processes can enhance your supply chain efficiency. Let us be your trusted partner in navigating the complexities of fine chemical synthesis, delivering value through innovation and operational excellence.
