Advanced Palladium-Catalyzed Synthesis of Polysubstituted Benzodihydrofuran Heterocycles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds that serve as the backbone for bioactive molecules. Patent CN111205279B, published in August 2021, introduces a significant advancement in this domain by disclosing a novel preparation method for polysubstituted benzodihydrofuran heterocyclic compounds. These structural motifs are not merely academic curiosities; they are critical components found in high-value therapeutic agents. For instance, the chroman-heterocyclic derivatives illustrated in the patent background are known for their outstanding biological activities, ranging from high-affinity 5-HT4 receptor agonists like Prucalopride to IMPDH inhibitors such as CellCept. The ability to efficiently synthesize these cores is paramount for drug discovery pipelines. This patent addresses the urgent need for efficient synthetic routes by leveraging a palladium-catalyzed strategy that merges simplicity with high step economy.
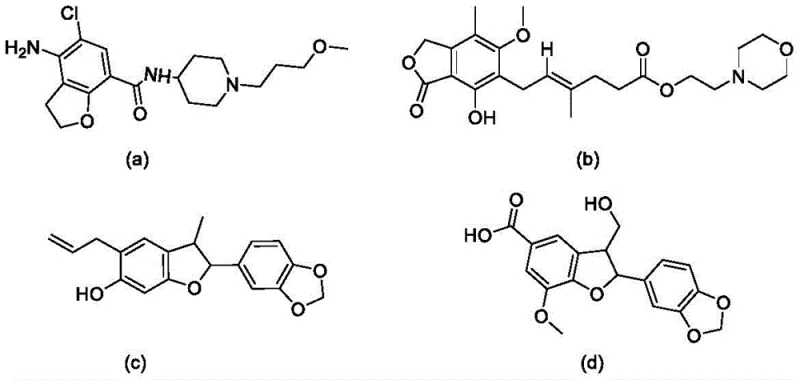
The strategic importance of this technology lies in its potential to streamline the supply chain for reliable pharmaceutical intermediate suppliers. By utilizing a catalytic system that tolerates a wide array of functional groups, manufacturers can access diverse chemical space without the need for extensive protecting group strategies. The patent explicitly highlights the synthesis of a series of multi-substituted benzodihydrofuranoheterocyclic compounds using simple and readily available acetylenone oxime ethers and o-iodophenyl alkenyl ethers. This approach contrasts sharply with older methodologies that often suffered from limited substrate scope and苛刻 reaction conditions. For R&D directors focusing on purity and impurity profiles, the mild nature of this reaction suggests a cleaner crude product profile, reducing the burden on downstream purification processes and ultimately enhancing the overall yield of high-purity OLED material or API precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of dihydrobenzofuran and chroman frameworks has relied on a variety of transition metal-catalyzed strategies, including nucleophilic substitutions, hydroalkylations, and intramolecular Heck cyclizations. While these methods have served the industry for decades, they are increasingly viewed as suboptimal for modern green chemistry standards. A primary limitation is the requirement for harsh reaction conditions, often involving excessive temperatures that can degrade sensitive functional groups or lead to polymerization of reactive intermediates. Furthermore, many conventional protocols exhibit a narrow substrate range, failing to accommodate electron-rich olefinic ethers or bulky substituents without significant loss in conversion efficiency. This lack of versatility forces process chemists to develop custom, non-scalable routes for each new analog, drastically increasing development time and cost. Additionally, the reliance on stoichiometric amounts of toxic reagents or difficult-to-remove metal catalysts complicates the purification workflow, posing challenges for meeting stringent regulatory limits on residual metals in final drug substances.
The Novel Approach
The methodology described in CN111205279B represents a paradigm shift by employing a palladium-catalyzed intermolecular eneyne cross-coupling reaction that operates under remarkably mild conditions. The core innovation involves the reaction of an alkynone oxime ether with an o-iodophenyl alkenyl ether in the presence of a specific catalytic system. As depicted in the general reaction scheme, this process facilitates the formation of the benzodihydrofuran ring through a concerted mechanism that avoids the pitfalls of earlier techniques. The reaction proceeds smoothly at temperatures between 50°C and 70°C, which is significantly lower than the reflux conditions often required for traditional cyclizations. This thermal gentleness preserves the integrity of sensitive moieties such as esters, halides, and heteroaromatic rings. Moreover, the use of electron-rich olefinic ethers, which have historically been neglected in favor of simpler alkenes, expands the chemical diversity accessible to medicinal chemists. This novel approach effectively bridges the gap between academic methodology and industrial practicality, offering a pathway for cost reduction in heterocyclic compound manufacturing.

Mechanistic Insights into Pd-Catalyzed Intermolecular Eneyne Cross-Coupling
To fully appreciate the technical robustness of this synthesis, one must delve into the mechanistic underpinnings of the palladium-catalyzed cycle. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the o-iodophenyl alkenyl ether substrate. This generates a reactive aryl-palladium(II) intermediate, which is the cornerstone of the subsequent transformation. In the presence of the oxidant, specifically copper chloride as detailed in the patent examples, the catalytic cycle is sustained, preventing the premature precipitation of palladium black which often deactivates such reactions. The aryl-palladium species then undergoes a migratory insertion into the alkene moiety of the second molecule, the alkenyl ether. This step is critical as it establishes the carbon-carbon bond that will eventually close the ring. The presence of additives like tetrabutylammonium bromide plays a subtle yet vital role, likely stabilizing the cationic palladium species or facilitating ligand exchange to maintain catalytic turnover. Finally, the cycle concludes with an oxidation and addition sequence that triggers mass decomposition and ring closure, yielding the polysubstituted benzodihydrofuran heterocyclic compound. This intricate dance of organometallic steps ensures high regioselectivity and minimizes the formation of unwanted byproducts.
From an impurity control perspective, the mechanism offers distinct advantages. The specificity of the oxidative addition step means that only the iodinated substrate participates in the initial activation, leaving other halogenated functionalities (like chlorides or bromides on the aromatic rings) untouched. This chemoselectivity is invaluable for late-stage functionalization where multiple halogen handles might be present on a complex drug candidate. Furthermore, the use of oxime ethers as directing groups or participants helps to steer the reaction trajectory, reducing the likelihood of random radical polymerization which is a common side reaction in free-radical mediated cyclizations. The patent data indicates that the reaction tolerates a wide variety of R1 and R2 groups, including phenyl, naphthyl, thiophene, and even long alkyl chains. This broad tolerance suggests that the transition state is not overly sterically hindered, allowing for the successful coupling of bulky substrates. For quality control teams, this translates to a more predictable impurity profile, simplifying the validation of analytical methods for high-purity benzodihydrofuran derivatives.
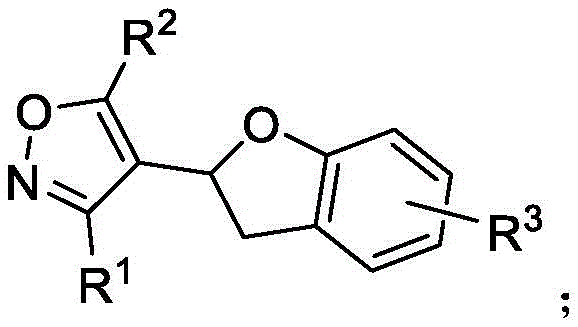
How to Synthesize Polysubstituted Benzodihydrofuran Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it accessible for both laboratory scale optimization and pilot plant operations. The protocol begins with the precise charging of reagents into a standard reactor, emphasizing the importance of stoichiometry in achieving optimal yields. The patent specifies a molar ratio of palladium acetate catalyst to substrate between 0.10 and 0.20 to 1, which strikes a balance between catalytic efficiency and cost. The oxidant, copper chloride, is used in a slight excess (2.0 to 3.0 equivalents) to ensure complete turnover of the palladium cycle. The reaction is conducted in tetrahydrofuran (THF), a ubiquitous solvent that offers excellent solubility for both organic substrates and inorganic salts. Maintaining the temperature strictly within the 50-70°C window is crucial; deviations could lead to incomplete conversion or decomposition of the oxime ether functionality. Following the reaction period of 10 to 16 hours, the workup involves a standard aqueous quench and extraction with ethyl acetate. The crude product is then purified, typically via thin layer chromatography or column chromatography using petroleum ether and ethyl acetate mixtures. Detailed standardized synthesis steps are provided in the guide below.
- Combine palladium acetate catalyst, copper chloride oxidant, tetrabutylammonium bromide additive, and potassium carbonate base in a reactor.
- Add alkynone oxime ether and o-iodophenyl alkenyl ether substrates dissolved in tetrahydrofuran solvent to the mixture.
- Stir the reaction at 50-70°C for 10-16 hours, then perform extraction and chromatographic purification to isolate the target heterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the significant simplification of the raw material supply chain. The starting materials, alkynone oxime ethers and o-iodophenyl alkenyl ethers, are described as simple and readily available. This accessibility reduces the risk of supply bottlenecks that often plague projects relying on exotic or custom-synthesized building blocks. Furthermore, the reaction conditions are inherently safer and more energy-efficient. Operating at 50-70°C eliminates the need for high-energy heating systems or cryogenic cooling, directly contributing to cost reduction in heterocyclic compound manufacturing. The absence of extreme pressures or temperatures also lowers the capital expenditure required for specialized reactor equipment, allowing production to be scaled up in standard glass-lined or stainless steel vessels commonly found in multipurpose chemical plants.
- Cost Reduction in Manufacturing: The economic viability of this process is bolstered by the elimination of expensive and toxic reagents often found in alternative routes. By utilizing a palladium catalyst that operates at low loading (10-20 mol%) and a cheap copper oxidant, the direct material costs are kept in check. More importantly, the high step economy means fewer unit operations are required to reach the final target. Traditional routes might require separate steps for ring closure and functional group installation, whereas this one-pot cascade achieves both simultaneously. This consolidation of steps drastically reduces solvent consumption, labor hours, and waste disposal costs. The streamlined purification process, which relies on standard chromatography rather than complex crystallization or distillation sequences, further enhances the overall cost efficiency, making the final product more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the robustness of the manufacturing process. The broad substrate scope of this reaction means that if a specific R-group substituent becomes unavailable due to geopolitical or logistical issues, the synthesis can often be adapted to use a structurally similar analog without re-optimizing the entire process. This flexibility is a critical risk mitigation strategy for long-term supply agreements. Additionally, the mild reaction conditions reduce the likelihood of batch failures due to thermal runaways or equipment malfunctions. Consistent batch-to-batch reproducibility is easier to achieve when the process window is forgiving, ensuring that delivery schedules are met reliably. The use of common solvents like THF and ethyl acetate also ensures that solvent supply remains stable, avoiding shortages associated with niche specialty chemicals.
- Scalability and Environmental Compliance: Scaling chemical processes from grams to tons often reveals hidden inefficiencies, but this methodology appears well-suited for commercial scale-up of complex heterocycles. The reaction does not generate hazardous gaseous byproducts, simplifying off-gas treatment requirements. The waste stream primarily consists of aqueous salt solutions and organic solvents, which are manageable through standard wastewater treatment and solvent recovery systems. This aligns with increasingly strict environmental regulations, reducing the compliance burden on manufacturing sites. The ability to run the reaction at near-atmospheric pressure and moderate temperatures also enhances operator safety, a key metric for modern chemical manufacturing. Consequently, this process supports sustainable growth, allowing companies to increase production volumes to meet market demand without proportionally increasing their environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production portfolios. The responses cover aspects ranging from substrate compatibility to purification strategies, offering a comprehensive overview for technical stakeholders.
Q: What are the key advantages of this palladium-catalyzed method over traditional synthesis?
A: This method operates under significantly milder conditions (50-70°C) compared to harsh traditional protocols, offers excellent functional group tolerance, and utilizes readily available starting materials like alkynone oxime ethers.
Q: What is the substrate scope for R1 and R2 groups in this reaction?
A: The reaction demonstrates broad applicability, accommodating diverse substituents including phenyl, naphthyl, chlorophenyl, methoxyphenyl, thiophene, and long-chain alkyl groups, ensuring versatility for various drug scaffold designs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common reagents and standard organic solvents like THF, with a straightforward workup procedure involving extraction and chromatography, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzodihydrofuran Supplier
The technological potential of polysubstituted benzodihydrofuran heterocycles is immense, serving as a gateway to next-generation therapeutics and functional materials. However, translating a patent into a commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this transition, offering comprehensive CDMO services tailored to the unique challenges of heterocyclic synthesis. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from clinical trials to full-scale market launch. We understand that consistency is key; therefore, our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch meets the highest international standards. Whether you require custom synthesis of novel analogs or reliable supply of established intermediates, our infrastructure is designed to support your long-term goals.
We invite you to explore how this advanced palladium-catalyzed technology can optimize your supply chain and reduce your overall cost of goods. Our technical procurement team is ready to engage in a detailed discussion about your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain insights into how switching to this methodology might impact your bottom line. We encourage you to contact us to obtain specific COA data for related compounds and to discuss route feasibility assessments for your target molecules. Let us collaborate to bring high-quality benzodihydrofuran derivatives to the market faster and more efficiently.
