Advanced Organocatalytic Strategy for Scalable Beta-Indolaldehyde Pharmaceutical Intermediates Production
Advanced Organocatalytic Strategy for Scalable Beta-Indolaldehyde Pharmaceutical Intermediates Production
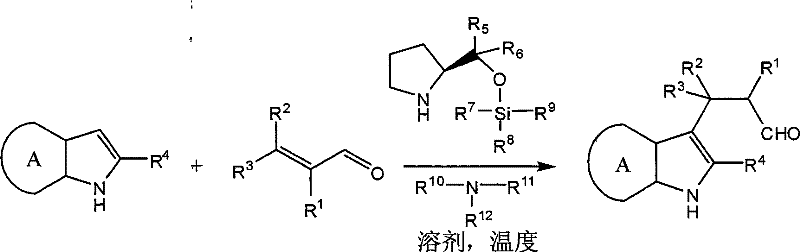
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally sustainable methodologies for constructing complex molecular architectures. A pivotal advancement in this domain is detailed in Chinese Patent CN101492407B, which discloses a novel organocatalytic method for the synthesis of β-indolaldehyde derivatives. This technology represents a significant departure from classical acid-catalyzed pathways, offering a metal-free alternative that achieves high enantioselectivity under remarkably mild conditions. For R&D directors and procurement specialists alike, this patent outlines a pathway to produce high-purity pharmaceutical intermediates while drastically reducing the environmental footprint associated with traditional Friedel-Crafts alkylations. The core innovation lies in the utilization of a prolinol silicon ether catalyst system, which effectively activates α,β-unsaturated aldehydes for conjugate addition with indoles, bypassing the need for corrosive strong acids or toxic heavy metals.
The strategic value of this patent extends beyond mere academic interest; it addresses critical pain points in commercial manufacturing, such as waste minimization and equipment longevity. By shifting the paradigm from stoichiometric acid promotion to sub-stoichiometric organocatalysis, the process inherently lowers the burden on downstream purification and waste treatment facilities. This aligns perfectly with the growing global demand for green chemistry solutions in the supply chain of active pharmaceutical ingredients (APIs). Furthermore, the versatility of the substrate scope, accommodating various substituted indoles and α,β-unsaturated aldehydes, positions this technology as a reliable platform for generating diverse libraries of bioactive 3-substituted indoles, which are ubiquitous motifs in medicinal chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-substituted indoles, particularly β-indole carbonyl derivatives, has relied heavily on the Friedel-Crafts alkylation reaction. Since the initial reports by Szmuszkovicz in 1957, the standard protocol has involved the use of strong protonic acids or Lewis acids to catalyze the addition of indoles to α,β-unsaturated carbonyl compounds. While these traditional methods offer high atom economy, they suffer from severe drawbacks that hinder their applicability in modern, regulated manufacturing environments. The requirement for large quantities of strong acids leads to significant equipment corrosion, necessitating expensive specialized reactors and frequent maintenance. Moreover, the acidic conditions often promote side reactions, reducing selectivity and complicating the isolation of the desired enantiomer. From an environmental perspective, the disposal of acidic waste streams poses a substantial challenge, increasing the overall cost of compliance and waste management for chemical manufacturers.
The Novel Approach
In stark contrast to the harsh conditions of the past, the methodology described in CN101492407B introduces a sophisticated organocatalytic system that operates under neutral to mildly basic conditions. By employing a chiral prolinol silicon ether in conjunction with a tertiary amine cocatalyst, this new approach activates the electrophilic α,β-unsaturated aldehyde without the need for external acid promoters. This shift eliminates the risks associated with equipment corrosion and toxic acid handling. Crucially, the reaction proceeds efficiently in a single solvent system, specifically methyl tert-butyl ether (MTBE), which is preferred over hazardous chlorinated solvents like dichloromethane. The operational simplicity is further enhanced by the fact that the reaction can be conducted in air without stringent anhydrous or oxygen-free requirements, making it exceptionally attractive for cost reduction in pharmaceutical intermediate manufacturing where operational complexity directly correlates with production costs.
Mechanistic Insights into Prolinol Silicon Ether Catalyzed Conjugate Addition
The success of this organocatalytic transformation hinges on the unique ability of the prolinol silicon ether catalyst to form a transient chiral environment around the substrate. Mechanistically, the secondary amine moiety of the prolinol derivative condenses with the α,β-unsaturated aldehyde to form a chiral iminium ion intermediate. This activation mode lowers the LUMO energy of the enal system, thereby rendering the β-carbon more susceptible to nucleophilic attack by the electron-rich indole ring. The bulky diphenyl groups on the catalyst scaffold provide significant steric hindrance, effectively shielding one face of the iminium ion and directing the approach of the indole nucleophile to the opposite face. This precise spatial control is what drives the high enantioselectivity observed in the products, often exceeding 90% ee even at relatively elevated temperatures compared to other organocatalytic systems.
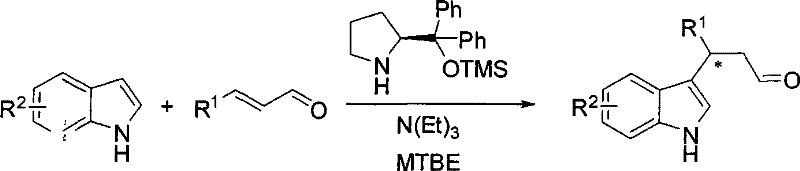
Furthermore, the inclusion of a tertiary amine, such as triethylamine, plays a synergistic role in the catalytic cycle. While the exact mechanistic contribution can vary, the amine likely assists in the deprotonation steps or stabilizes transition states, facilitating the turnover of the catalyst. The patent data indicates that this dual-catalyst system allows the reaction to proceed at temperatures as high as -20°C to 40°C, a significant improvement over previous organocatalytic methods that required cryogenic conditions below -50°C. This thermal robustness is a key indicator of the catalyst's efficiency and suggests a lower activation energy barrier for the rate-determining step. For process chemists, understanding this mechanism is vital for optimizing reaction parameters, ensuring that the delicate balance between catalyst loading, temperature, and reaction time is maintained to maximize both yield and optical purity.
How to Synthesize Beta-Indolaldehyde Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the sequence of reagent addition and temperature control to ensure reproducibility. The patent provides a clear framework for executing the reaction, emphasizing the importance of dissolving the catalyst fully before introducing the reactive substrates. The procedure is designed to be straightforward, avoiding complex inert atmosphere techniques which simplifies the technical barrier for adoption. Operators should note that while the reaction is tolerant to air, maintaining the specified temperature range is critical for achieving the reported high enantioselectivity. The following guide outlines the standardized operational steps derived from the patent examples to assist technical teams in replicating this high-value transformation.
- Dissolve the chiral catalyst, diphenylprolinol trimethylsilyl ether, in methyl tert-butyl ether (MTBE) under stirring.
- Sequentially add the cocatalyst triethylamine, the alpha,beta-unsaturated aldehyde substrate, and the indole derivative to the solution at -20°C.
- Maintain the reaction mixture at -20°C for approximately 24 hours, then concentrate and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this organocatalytic process offers tangible benefits that extend well beyond the reaction flask. The elimination of corrosive acids and toxic chlorinated solvents translates directly into reduced operational expenditures related to safety protocols, waste disposal, and equipment maintenance. By adopting a metal-free and acid-free workflow, manufacturers can significantly streamline their regulatory compliance processes, as the impurity profile of the final product is cleaner and easier to characterize. This reduction in process complexity enhances supply chain reliability, as the reliance on specialized, hazard-classified raw materials is minimized. The use of common, commercially available reagents like triethylamine and MTBE ensures that sourcing remains stable and cost-effective, mitigating the risks associated with supply disruptions of exotic catalysts or reagents.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the simplification of the downstream processing workflow. Since the reaction does not generate acidic waste streams, the need for extensive neutralization and washing steps is removed, leading to substantial savings in utility consumption and labor. Additionally, the organocatalyst, being an organic small molecule, is generally less expensive than precious metal catalysts and can often be recovered or used at low loadings without compromising performance. The avoidance of cryogenic cooling requirements (operating at -20°C rather than -55°C) also results in significant energy savings, as standard industrial chillers can be utilized instead of specialized low-temperature equipment, thereby lowering the overall cost of goods sold for these high-value intermediates.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthetic route ensures consistent production schedules. The tolerance to air and moisture means that reactions are less prone to failure due to minor deviations in atmospheric conditions, which is a common risk factor in large-scale batch processing. The substrates, including various substituted indoles and cinnamaldehyde derivatives, are widely available commodity chemicals, reducing the lead time for raw material procurement. This stability allows supply chain planners to forecast production capacity with greater accuracy, ensuring that downstream API synthesis lines are fed consistently without unexpected delays caused by catalyst sensitivity or reagent scarcity.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this organocatalytic method is inherently designed for scalability. The exotherm of the reaction is manageable under the described conditions, and the use of MTBE, a solvent with favorable physical properties for separation, facilitates efficient product isolation. Environmentally, the process aligns with green chemistry principles by minimizing the E-factor (mass of waste per mass of product). The absence of heavy metals removes the need for costly metal scavenging steps, which are often bottlenecks in GMP manufacturing. This eco-friendly profile not only satisfies internal sustainability goals but also appeals to end-clients who are increasingly demanding greener supply chains for their pharmaceutical products.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patent, we have compiled a set of frequently asked questions based on the specific data and claims within the document. These insights address common concerns regarding reaction conditions, catalyst specificity, and the comparative advantages over legacy technologies. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided below are grounded strictly in the experimental results and descriptions found in the source patent data.
Q: What are the primary advantages of this organocatalytic method over traditional Friedel-Crafts alkylation?
A: Unlike traditional methods requiring strong protonic or Lewis acids which cause equipment corrosion and severe pollution, this organocatalytic approach operates under mild, non-acidic conditions. It eliminates the need for toxic solvents like dichloromethane and avoids the use of hazardous trifluoroacetic acid, resulting in a greener process with high enantioselectivity.
Q: What specific catalyst system is employed to achieve high enantioselectivity?
A: The process utilizes a chiral prolinol silicon ether, specifically diphenylprolinol trimethylsilyl ether, as the primary organocatalyst. This is combined with a tertiary amine cocatalyst, such as triethylamine, to efficiently activate the alpha,beta-unsaturated aldehyde for asymmetric conjugate addition.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method is highly suitable for scale-up. It operates at relatively high temperatures compared to previous organocatalytic methods (e.g., -20°C instead of -55°C), uses a single solvent system (MTBE), and does not require strict anhydrous or oxygen-free conditions, significantly simplifying operational complexity for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Indolaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalytic technologies in modernizing the production of complex pharmaceutical intermediates. Our team of expert process chemists has extensively evaluated the methodology described in CN101492407B and possesses the technical capability to adapt and optimize this route for commercial scale-up. We understand that transitioning from bench-scale discovery to multi-ton production requires rigorous process validation and quality control. Therefore, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to ensure that every batch meets stringent purity specifications. Our state-of-the-art rigorous QC labs are equipped to verify enantiomeric excess and impurity profiles, guaranteeing that the high-purity beta-indolaldehyde intermediates we supply are ready for immediate use in your downstream API synthesis.
We invite you to collaborate with us to explore how this innovative synthetic strategy can enhance your project's efficiency and cost-effectiveness. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. By partnering with us, you gain access not only to high-quality materials but also to deep technical expertise that can accelerate your development timelines. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technological excellence can support your supply chain objectives.
